

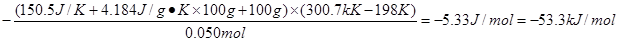 ��
��


| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A�������� | B����������ľ̿���� | C���ƾ�����ˮ | D��HCl��������ˮ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 O2(g)��H2O(l)����H����285.8 kJ��mol-1
O2(g)��H2O(l)����H����285.8 kJ��mol-1 O2(g)��H2O(g)����H����241.8 kJ��mol-1
O2(g)��H2O(g)����H����241.8 kJ��mol-1 O2(g)��CO(g)����H����110.5 kJ��mol-1
O2(g)��CO(g)����H����110.5 kJ��mol-1�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��ϡ������ϡ����������Һ�����кͷ�Ӧ���Ȼ�ѧ����ʽΪ CH3COOH (aq) + NaOH (aq) = CH3COONa(aq) + H2O(l) ��H =  57.3kJ/mol 57.3kJ/mol |
| B����101kPaʱ1mol����ȼ��ʱ�ķ�Ӧ�Ƚ��������ʵ�ȼ���� |
| C��δ��������Ϊ����Դ������һ���ŵ��dz��ȼ�յIJ��ﲻ��Ⱦ���� |
| D�������¶Ȼ������������Ըı仯ѧ��Ӧ�ķ�Ӧ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
| ��ѧ�� | Si��O | Si��Cl | H��H | H��Cl | Si��Si | Si��C |
| ����/kJ��mol?1 | 460 | 360 | 436 | 431 | 176 | 347 |

 Si(s)��4HCl(g)���÷�Ӧ�ķ�Ӧ��DH��
Si(s)��4HCl(g)���÷�Ӧ�ķ�Ӧ��DH���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��ˮ��ﵽƽ��ʱ(������)�����Ȼ����ﱥ����Һ����Һ�����Ի���ǿ |
| B��Ũ��Ϊ5 mol/L��0.5 mol/L������FeCl3��Һ������������ͬʱ��Fe3����ˮ��̶�ǰ�߱Ⱥ���С |
| C����50���20���ͬŨ�ȵ�����FeCl3ϡ��Һ������������ͬʱ��Fe3����ˮ��̶�ǰ�߱Ⱥ���С |
| D��Ϊ����Fe3����ˮ�⣬�Ϻõر���FeCl3��Һ��Ӧ���������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
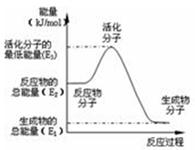
| A��H2ȼ���ܷų��������ȣ���CH3OHת���H2�Ĺ��̱����������� |
| B����Ӧ���е������仯����ͼ��ʾ�����H2= E1�� E2 |
| C�����ݢ���֪��CH3OH��ȼ���ȡ�H����192.9 kJ��mol -1 |
| D������ͼ�У�������ʵĴ�������ʹE3���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��N5+����34��������� |
| B��N5+�е�ԭ�Ӽ������Ӽ���� |
| C��������N5ASF6��AS�Ļ��ϼ�Ϊ+1�� |
| D��������N5ASF6Ϊ���ӻ����� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com