
ij����С��ֱ�����ͼ��ʾװ�ö�ԭ��غ͵���ԭ������ʵ��̽����

ͼ1 ͼ2
��ش�:
��.��ͼ1��ʾװ�ý��е�һ��ʵ�顣
(1)�ڱ�֤�缫��Ӧ����������,�������Cu���缫����������(����ĸ���)��
| A���� | B��ʯī | C���� | D���� |
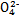 ������������(��������ҡ�������������)�ƶ�;��ֽ���ܹ۲쵽����������������������������������������������
������������(��������ҡ�������������)�ƶ�;��ֽ���ܹ۲쵽���������������������������������������������� 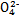 )����Һ�г��Ϻ�ɫ��
)����Һ�г��Ϻ�ɫ�� Fe
Fe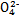 +4H2O����������������
+4H2O����������������  Fe2O3+ZnO+2K2ZnO2
Fe2O3+ZnO+2K2ZnO2 ������״Ԫ���Ծ�ϵ�д�
������״Ԫ���Ծ�ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���������л�ʵ�����г��õ���Ʒ������������ԭ��Ӧ֪ʶ�͵�ѧ֪ʶ���Լ��������һ��ԭ��ء�����д���пհף�
��1��ʵ��ԭ����Fe��2H��=Fe2����H2����
��2��ʵ����Ʒ���缫________��________��ϡ���ᡢ________����д��ȱ��ʵ����Ʒ�������������ߵ����ƣ���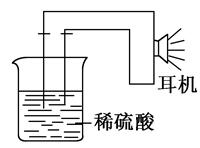
��3��ʵ��װ�ã�
��4��ԭ�����Ƽ�ע������⡣
�ٰ���ͼ��ʾ���Ӻ�ʵ��������ע��۲죨�������������Ƿ����������������û�У��ɽ�ԭ��ص������缫�е�һ���Ӵ�������ͷ�ϵ�һ����ע�⣺�Ӵ���ͬʱ��������һ������������ԭ��ص���һ���缫�ϵģ�����ʱ�����������������������ꡱ��������ԭ���ǣ���ԭ����У��ɻ�ѧ��ת��Ϊ________��
�������װ���еĶ�����Ϊ�����ƣ�������Ӧ�ýӵ����Ƶ�______
�����缫��Ӧ��________________����һ��Ӧ�ýӵ����Ƶ�________�����缫������________��Ӧ������������ԭ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�������ǿ������������ԭ�ֶΣ��ڻ���������������Ҫ��Ӧ�á���ش��������⣺
��1����ͭΪ��������ʯīΪ��������NaCl��Һ�����Һ���е�⣬�õ��뵼�����Cu2O��һ�������Դ����������ӦʽΪ___________________________��������ӦʽΪ________��
��2��ijͬѧ�����ͼ��ʾ��װ��̽�������ĸ�ʴ����������жϺ�������____________������ţ���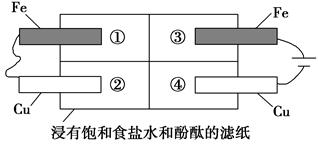
a������ͭƬ�������ݲ���
b��������Ƭ�ĵ缫��ӦʽΪ2Cl����2e��=Cl2��
c�����ȹ۲쵽��ɺ�ɫ�������Ǣ���
d�������͢�����ͭƬ���������������仯
��3�������о����֣��ø�Ĥ��ⷨ������Ũ����ȩ��ˮ�Ĺ��վ������̼��ܺĽϵ͵��ŵ㣬��ԭ����ʹ��ȩ�ֱ�����������������Ӧ�����Ҵ������ᣬ�ܷ�ӦʽΪ2CH3CHO��H2O CH3CH2OH��CH3COOH
CH3CH2OH��CH3COOH
ʵ�����У���һ��Ũ�ȵ���ȩ��Na2SO4��ҺΪ�������Һ��ģ����ȩ��ˮ�Ĵ������̣���װ����ͼ��ʾ��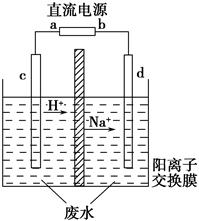
�����Լ������ȼ�ϵ��Ϊֱ����Դ����ȼ�ϵ����b��Ӧͨ��__________���ѧʽ�����缫��ӦʽΪ____________________���������У�������Na2SO4�����ʵ���________�����������С�����䡱����
����ʵ�ʹ��մ����У���������ȩ��ȥ���ʿɴ�60%�������������ֱ�ע��1 m3��ȩ����Ϊ3 000 mg��L��1�ķ�ˮ���ɵõ��Ҵ�________kg������������С�����һλ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��14�֣���ҵ����ij����������Cu2O��Al2O3��Fe2O3��SiO2����ȡͭ�IJ����������£�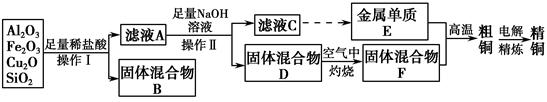
��֪��Cu2O��2H��=Cu��Cu2����H2O
��1��ʵ������������Ϊ________���ڿ��������չ�������Dʱ���õ����ֹ������ʵ������������������ƾ��ơ��������⣬����________�����������ƣ���
��2����ҺA����Ԫ�صĴ�����ʽΪ________�������ӷ��ţ������ɸ����ӵ����ӷ���ʽΪ_______________________________________________
________��������ҺA�д��ڸ����ӵ��Լ�Ϊ________�����Լ����ƣ���
��3����������E���������F������ijһ��Ӧ�����ں��Ӹֹ죬�÷�Ӧ�Ļ�ѧ����ʽΪ_____________________________________________________��
��4�������£���pH��NaAlO2��NaOH������Һ�У���ˮ�������c��OH����ǰ��Ϊ���ߵ�108������������Һ��pH��________��
��5�������õ�ⷨ���д�ͭ����ʱ������������ȷ����________������ţ���
a������ȫ��ת��Ϊ��ѧ��
b����ͭ�ӵ�Դ����������������Ӧ
c����ͭ��������������Һ��Cu2��Ũ�ȼ�С
d����ͭ����ʱͨ���ĵ�������������ͭ������ȷ����ϵ
�ڴ�Ũ���ᡢŨ���ᡢ����ˮ��ѡ�ú��ʵ��Լ����ⶨ��ͭ��Ʒ�н���ͭ�������������漰����Ҫ���裺��ȡһ����������Ʒ��______________�����ˡ�ϴ�ӡ����������ʣ�����ͭ������������ȱ�ٵIJ������裬���������������̵�ϸ�ڣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��.������ʳ����ȷ��ҩ�������彡������Ҫ��֤��
����������������Aʳ�Σ�BС�մ�Cƻ��֭��D�����ǣ�E��ù�أ��밴����Ҫ�����(�����)��
����ά����C���� ����ֱ�ӽ���ѪҺ�������������� ��Ӧ����㷺�Ŀ�����֮һ���� ���ȿ���Ϊ���ɼ����ֿ�����θ�������� ��ʳ�ù��������Ѫѹ���ߡ���������� ��
��.������������ʹ�����������������������һ����Ҫ��־��
��1��д����ҵ���ó�������������Ҫ��ѧ��Ӧ����ʽ�� ��
��2�������˵�����δ��ʱϴ������Һ�к�NaCl��,�ڶ�������ʴ���ֺ��ɫ��ߡ��Իش�
�������ĸ�ʴ��Ҫ���� ���ѧ��绯ѧ����ʴ��ɵġ��γɵ��������Ҫ�ɷ��� ��
��Ϊ��ֹ�ִ��Ĵ����ں�ˮ�и�ʴ��һ���ڴ������� ���п�顱��ͭ�顱����
��.������������������ͷ�չ����Ҫ���ʻ���������ʹ�ò��Ͽ��Ը������ǵ����
��1���������ݽ���������������ϡ����в��ϲ����ڹ����β��ϵ��� ������ĸ����
A��ʯ��ʯ B��ˮ�� C������
��2�������в����У��������ǽ������ϵ��� (����ĸ)������������Ʒ���� ��
A�����ڡ������� B��������ϩ���ϡ���C���������մ� D��������
��3�������йغϽ����ʵ�˵����ȷ���� ������ĸ����
A���Ͻ���۵�һ������ijɷֽ�����
B���Ͻ��Ӳ��һ������ijɷֽ�����
C����ɺϽ��Ԫ��������ͬ���Ͻ�����ܾ�һ����ͬ
D���Ͻ�����ɷֽ�����ȣ�����������������������ѧ���е����
��4���ϳ����ϡ��ϳ��� �dz�˵������ϳɲ��ϡ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����ͼ��ʾ���ձ��ж�ʢ��ϡ���ᡣ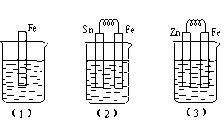
��д����3���еĵ缫��Ӧ��Fe�� �� Zn�� ��
�ڱȽϣ�1������2������3���д�������ʴ�������ɿ쵽����˳���� ������ţ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�Ի�ͭ��Ϊԭ�ϣ���ȡ����ͭ�Ĺ���������ʾ��
����ͭ����Ҫ�ɷ�ΪCuFeS2����������CaO��MgO��Al2O3�����顣
������ͼ��ʾװ�ý��е绯ѧ����ʵ�顣����ѡ��ͭ��ۼ�����۵������������ٽ��裬ʹ����ܽ⡣��������ͨ������������������������
��һ��ʱ���ȡ��������Һ�������м����л���ȡ��(RH)������Ӧ��2RH(�л���)+Cu2+(ˮ��) R2Cu(�л���)+2H��(ˮ��)������л��࣬�����м���һ��Ũ�ȵ����ᣬʹCu2+���������� �����������ͭ��Һ�Ƶý���ͭ��
R2Cu(�л���)+2H��(ˮ��)������л��࣬�����м���һ��Ũ�ȵ����ᣬʹCu2+���������� �����������ͭ��Һ�Ƶý���ͭ��
��1����ͭ��ۼ��������������ἰ��������Ҫ�������·�Ӧ��CuFeS2 + 4H+��Cu2+ + Fe2+ + 2H2S�� 2Fe3+ + H2S��2Fe2+ + S��+ 2H+ ������������������Ҫ������ ����2�����������缫�Ͽ�ʼʱ�д������ݲ��������к�ɫ����������һ��ʱ����ɫ�����ܽ⡣д��������ɫ����ķ�Ӧ����ʽ ��
��3������ʵ���ҽ��в�������л����ˮ�����Ҫʵ�������� ��
��4����������л����м���һ��Ũ�ȵ����ᣬCu2+����������ԭ���� ����5��������������200mL0.5 mol/L��CuSO4��Һ������ͭ3.2 g����ʱ��Һ������Ũ���ɴ�С��˳���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��������ҵ���������ɡ���������ȡ������ұ�������ļӹ��Ȼ��ڹ��ɡ���ش��������⣺
��1����ҵ�ϲ��õ���������ͱ���ʯ(Na3AlF6)������ķ���ұ���õ���������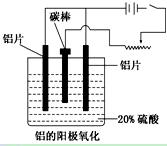
2Al2O3  4Al��3O2��
4Al��3O2��
�������ʯ�����ã�________________________________________________��
��2��������������������������������Fe��Si�����ʣ����õ�ⷽ����һ���ᴿ���õ����������ĵ缫��ӦʽΪ________________�����п����������ϵ���__________��
A������ B��ʯī C��Ǧ�� D������
��3������������ʹ���������������ܵ�����Ĥ����ϡ����Ϊ���Һ�������������ĵ缫��ӦʽΪ_____________________________________________________________��
��4�������������������У���Ҫ���ϵص�����ѹ��������_________________��
��5������˵����ȷ����__________________��
A������������Ӧ��ԭ���ԭ�����н������ϱ��洦���ļ���
B������������������ǿ������ľ�Ե����
C������������������߽���������Ͻ����ʴ�ԣ�����ĥ���½�
D��������������Ĥ���ж���ԣ����к�ǿ���������ܣ�������Ⱦ�϶��ʸ�����ɫ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��Ҫ����գ�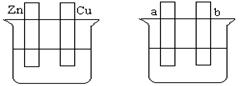
A B
��1����Aͼ�У�ϡ����Ϊ�������Һ���õ������Ӻ�ͭƬ�缫��Ӧʽ ��
��2����Bͼ�����ֱ����Դ����Ҫ��a����ͭ�����Ա�Ҫ�����Ӻ�װ�ý� ��b���缫��Ӧʽ ��
��3����Bͼ�����ֱ����Դ�����缫Ϊ���Ե缫���������Һ��CuSO4��Һ��������������ܷ�Ӧ���ӷ���ʽΪ ����������3.2 g���������Ϸų��������ڱ�״���µ������_____L������һ������ ���ѧʽ������Һ�ָܻ�������ǰ��ȫһ�¡�
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com