
��14�֣�A��B��C��D��E��F �����ֶ����ڵ�����Ԫ�أ����ǵ�ԭ���������ε�����AԪ��������Ԫ�ؾ�����ͬһ���ڣ�CԪ����B��DԪ��ͬ�������ڣ�CԪ�ص�����������Ӧˮ������CԪ�ص���̬�⻯��X�����Ӧ������Y��D��FԪ��ͬ�壬E�Ƕ�����������ԭ�Ӱ뾶����Ԫ�ء�������������⣺
��1��Dλ��Ԫ�����ڱ���__________����__________�塣
��2��Eԭ�ӵĽṹʾ��ͼΪ__________��
��3��D��F��Ԫ����Ƚϣ��ǽ����Խ�ǿ����__________��Ԫ�����ƣ���������֤�ý��۵���ʵ��__________________________________________________��ֻдһ�����ɣ���
��4��A��D��E����Ԫ����ɵĻ�����������Ļ�ѧ��������____________________��
��5������X���Լ�����ֽ��__________��
��6����A��C��D����Ԫ����ɵ�����ǿ����ʼ��ң����ǵ�ˮ��Һ�������ԡ���������ˮ�ĵ��룬��Ļ�ѧʽΪ__________ �����Ҵٽ�ˮ�ĵ��룬���ҵ�ˮ��Һ�����Ե�ԭ���ǣ������ӷ���ʽ��ʾ��______________________________________________��
��7��д��D��E��Ԫ���γɵ�ԭ�Ӹ�����Ϊ1��1�Ļ�������ˮ��Ӧ�Ļ�ѧ����ʽ��
____________________________________________________________ ��
��8��A��D����γɸ�һ��˫ԭ�������ӣ���10�����ӣ�д���������������ᷴӦ�����ӷ���ʽ__________________________________________________��
��9����֪0.4mol Һ̬C2 A4��Һ̬˫��ˮ��Ӧ������C2��Һ̬ˮ���ų�327.2 kJ���������÷�Ӧ���Ȼ�ѧ����ʽΪ__________________________________________________��
��10��D��E��F�����Ӱ뾶�ɴ�С��˳��Ϊ��______________________________�������ӷ��ű�ʾ����
��11��д��A��D����Ԫ���γɵĻ������һ����;______________________________��
��1��2 ��A����1�֣�
��2��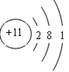 ��1�֣�
��1�֣�
��3������1�֣���̬�⻯���ȶ��ԣ�H2O>H2S(1��)�������𰸺���Ҳ���֣�
��4�����Ӽ������ۼ�����1�֣�
��5����ɫʯ����ֽ���ɫʯ����Һ��1�֣��������𰸺���Ҳ���֣�
��6��HNO3��1�֣� NH4++H2O NH3��H2O+H+(1��)
NH3��H2O+H+(1��)
��7��2Na2O2+2H2O===4NaOH+O2����1�֣�
��8��H��+OH����H2O��1�֣�
��9��N2H4(l)+2H2O2(l)===N2(g)+4H2O(l)����H����818kJ��mol��1��2�֣�������������Ҳ���֣�
��10��S2����O2����Na+��1�֣�
��11��H2O���ܼ�����H2O2����ɫ����������1�֣�������������Ҳ���֣�
����



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| ������ | X | Y | Z |
| �۵�/K | 1266 | 1534 | 183 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 [��ѧ/ѡ��/���ʽṹ������]A��B��C��D��E���ֶ�����Ԫ�أ�ԭ��������������Ԫ�ض�Ӧ�ĵ��ʾ�Ϊ���壮A��C��E��Ԫ�ص�ԭ�Ӻ����ֻ��2��δ�ɶԵ��ӣ�B��EԪ�ص�ԭ������֮�͵���C��DԪ�ص�ԭ������֮�ͣ�
[��ѧ/ѡ��/���ʽṹ������]A��B��C��D��E���ֶ�����Ԫ�أ�ԭ��������������Ԫ�ض�Ӧ�ĵ��ʾ�Ϊ���壮A��C��E��Ԫ�ص�ԭ�Ӻ����ֻ��2��δ�ɶԵ��ӣ�B��EԪ�ص�ԭ������֮�͵���C��DԪ�ص�ԭ������֮�ͣ��鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| 1 |
| 2 |
| 1 |
| 2 |
| 3 |
| 2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com