
°±ŗĶĮŖ°±(N2H4)ŹĒµŖµÄĮ½ÖÖ³£¼ū»ÆŗĻĪļ£¬ŌŚæĘѧ¼¼ŹõŗĶÉś²śÖŠÓŠÖŲŅŖµÄÓ¦ÓĆ”£øł¾Ż
ĢāŅāĶź³ÉĻĀĮŠ¼ĘĖć£ŗ
ĮŖ°±ÓĆŃĒĻõĖįŃõ»ÆÉś³ÉµŖµÄĮķŅ»ÖÖĒā»ÆĪļ£¬øĆĒā»ÆĪļµÄĻą¶Ō·Ö×ÓÖŹĮæĪŖ43.0£¬ĘäÖŠµŖ
Ō×ÓµÄÖŹĮæ·ÖŹżĪŖ0.977£¬¼ĘĖćČ·¶ØøĆĒā»ÆĪļµÄ·Ö×ÓŹ½ĪŖ ”£øĆĒā»ÆĪļŹÜײ»÷ŌņĶźČ«·Ö½āĪŖµŖĘųŗĶĒāĘų”£4.30 gøĆĒā»ÆĪļŹÜײ»÷ŗó²śÉśµÄĘųĢåŌŚ±ź×¼×“æöĻĀµÄĢå»ż L”£
ĮŖ°±ŗĶĖÄŃõ»Æ¶žµŖæÉÓĆ×÷»š¼żĶĘ½ų¼Į£¬ĮŖ°±ŹĒČ¼ĮĻ£¬ĖÄŃõ»Æ¶žµŖ×÷Ńõ»Æ¼Į£¬·“Ó¦²śĪļ
ŹĒµŖĘųŗĶĖ®”£ÓÉĮŖ°±ŗĶĖÄŃõ»Æ¶žµŖ×é³ÉµÄ»š¼żĶĘ½ų¼ĮĒ”ŗĆĶźČ«·“Ӧɜ³É72.0 kgĖ®£¬ĶĘ½ų¼ĮÖŠĮŖ°±µÄÖŹĮæĪŖ kg”£
°±µÄĖ®ČÜŅŗæÉÓĆÓŚĪüŹÕNOÓėNO2»ģŗĻĘųĢ壬æÉĻū³żµŖŃõ»ÆŗĻĪļ¶Ō»·¾³µÄĪŪČ¾”£Ēė·Ö
±šŠ“³öÓŠ¹ŲµÄ·“Ó¦·½³ĢŹ½ĪŖ£ŗ £» ”£
£Ø1£©HN3»ņN3H (1·Ö) 4.48(1·Ö)
£Ø2£©64 kg (2·Ö)
£Ø3£©4NH3+6NO=5N2+6H2O (2·Ö) 8NH3+6NO2=7N2+12H2O (2·Ö)£ØŠ“³ÉNH3”¤ H2OŅ²øų·Ö£©
½āĪöŹŌĢā·ÖĪö£ŗ£Ø1£©N(N)=43.0”Į0.977”Ā14=3£¬N(H)=(43.0-14”Į3)”Ā1=1£¬·Ö×ÓŹ½ĪŖ HN3£»
n(HN3)=4.30”Ā43=0.1mol£¬øł¾ŻŌ×ÓŹŲŗćµĆµ½n(H2)=0.05mol£¬n(N2)=0.15mol
±źæöĻĀĢå»żĪŖ£Ø0.05+0.15£©”Į22.4=4.48L
£Ø2£©øĆ·“Ó¦·½³ĢŹ½ĪŖ2N2H4+N2O4=3N2+4H2O£¬øł¾Ż·½³ĢŹ½¼ĘĖćæɵĆm(N2H4)=64kg
£Ø3£©4NH3+6NO=5N2+6H2O 8NH3+6NO2=7N2+12H2O
æ¼µć£ŗæ¼²éŌŖĖŲ»ÆŗĻĪļ¼°·“Ó¦ČČµÄ¼ĘĖćÓŠ¹ŲĪŹĢā”£



| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
ŅŃÖŖA”¢B”¢C”¢D”¢E”¢FŹĒŗ¬ÓŠĶ¬Ņ»ÖÖŌŖĖŲµÄ»ÆŗĻĪļ£¬ĘäÖŠFÄÜŹ¹ŗģÉ«ŹŖČóŹÆČļŹŌÖ½±äĄ¶É«£¬ĖüĆĒÖ®¼äÄÜ·¢ÉśČēĻĀ·“Ó¦£ŗ
¢Ł A+H2O ”ś B+C ¢Ś C+F ”ś D ¢Ū D+NaOH ”ś F+E+H2O
£Ø1£©Š“³öĖüĆĒµÄ»ÆѧŹ½£ŗD_______________£¬F_______________”£
£Ø2£©Š“³ö¢Ł·“Ó¦µÄ»Æѧ·½³ĢŹ½£ŗ_______________________________
£Ø3£©Š“³ö·“Ó¦¢ŪµÄĄė×Ó·½³ĢŹ½£ŗ___________________________”£
£Ø4£©¹¤ŅµÉś²śCµÄ¹ż³ĢÖŠÓŠČēĻĀŅ»²½·“Ó¦£ŗ¼“F¾“ß»ÆŃõ»ÆÉś³ÉBŗĶH2O£¬Š“³öøĆ²½·“Ó¦µÄ»Æѧ·½³ĢŹ½£ŗ________________________________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
ĄūÓĆŹÆ»ŅČéŗĶĻõĖį¹¤ŅµµÄĪ²Ęų(ŗ¬NO”¢NO2)·“Ó¦£¬¼ČÄܾ»»ÆĪ²Ęų£¬ÓÖÄÜ»ńµĆÓ¦ÓĆ¹ć·ŗµÄCa(NO2)2£¬Ęä²æ·Ö¹¤ŅÕĮ÷³ĢČēĻĀ£ŗ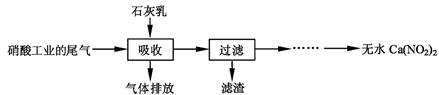
Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©Ņ»¶ØĢõ¼žĻĀ£¬NOÓėNO2“ęŌŚĻĀĮŠ·“Ó¦£ŗNO(g)£«NO2(g)  N2O3(g)£¬ĘäĘ½ŗā³£Źż±ķ“ļŹ½ĪŖK= ”£
N2O3(g)£¬ĘäĘ½ŗā³£Źż±ķ“ļŹ½ĪŖK= ”£
£Ø2£©ÉĻŹö¹¤ŅÕÖŠ²ÉÓĆĘųŅŗÄęĮ÷½Ó“„ĪüŹÕ(Ī²Ęų“ÓĪüŹÕĖžµ×²æ½ųČė£¬ŹÆ»ŅČé“ÓĪüŹÕĖž¶„²æÅēĮÜ)£¬ĘäÄæµÄŹĒ £»ĀĖŌüæÉŃ»·ĄūÓĆ£¬ĀĖŌüµÄÖ÷ŅŖ³É·ÖŹĒ (Ģī»ÆѧŹ½)”£
£Ø3£©øĆ¹¤ŅÕŠčæŲÖĘNOŗĶNO2ĪļÖŹµÄĮæÖ®±Č½Ó½ü1©U1”£Čōn(NO)£ŗn(NO2)£¾1©U1,Ōņ»įµ¼ÖĀ £»Čōn(NO)£ŗn(NO2)£¼1©U1,Ōņ»įµ¼ÖĀ ”£
£Ø4£©Éś²śÖŠČÜŅŗŠč±£³ÖČõ¼īŠŌ£¬ŌŚĖįŠŌČÜŅŗÖŠCa(NO2)2»į·¢Éś·Ö½ā£¬²śĪļÖ®Ņ»ŹĒNO£¬Ęä·“Ó¦µÄĄė×Ó·½³ĢŹ½ ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
ĮņĖįŹĒÓĆĶ¾¹ć·ŗµÄ»Æ¹¤ŌĮĻ£¬æÉ×÷ĶŃĖ®¼Į”¢ĪüĖ®¼Į”¢Ńõ»Æ¾£ŗĶ“߻ƼĮµČ”£
¹¤ŅµÖĘĮņĖįĶµÄ·½·Øŗܶą”£
£Ø1£©·½·ØŅ»”¢ÓĆÅØĮņĖįŗĶĶÖĘČ”ĮņĖįĶ”£øĆ·“Ó¦µÄ»Æѧ·½³ĢŹ½ŹĒ_____________________,“Ė·ØµÄ×ī“óȱµćŹĒ____________________________”£
£Ø2£©·½·Ø¶ž”¢ÓĆĻ”ĮņĖį”¢ĶŗĶŃõ»ÆĢśÖĘČ”ĮņĖįĶ£¬Éś²śµÄÖ÷ŅŖ¹ż³ĢČēĻĀĶ¼ĖłŹ¾£ŗ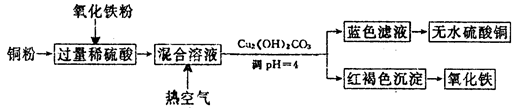
¢ŁĻ”ĮņĖį”¢ĶŗĶŃõ»ÆĢś·“Ó¦µÄĄė×Ó·½³ĢŹ½ŹĒ__________________”¢________________£»
Ļņ»ģŗĻČÜŅŗÖŠĶØČėČČæÕĘųµÄ·“Ó¦µÄĄė×Ó·½³ĢŹ½ŹĒ_______________________________”£
¢ŚĒėĖµ³öµ÷ÕūPHĪŖ4µÄÄæµÄŹĒ_______________________£»ÓÉĀĖŅŗµĆµ½ĪŽĖ®ĮņĖįĶµÄŹµŃé²Ł×÷ŹĒ______________________________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
Šķ¶ąŗ¬ĀČĪļÖŹÓėÉś»īĆÜĒŠĻą¹Ų,ČēHC10”¢C102”¢NaClO2µČ¶¼ŹĒÖŲŅŖµÄɱ¾śĻū¶¾¼ĮŗĶĘÆ°×¼Į”£ĻĀĮŠŹĒÖŲŅŖµÄŗ¬ĀČĘÆ°×¼ĮNaClO2µÄ¹¤ŅµŗĻ³ÉĮ÷³ĢĶ¼”£
ŅŃÖŖ£ŗ“æClO2Ņ×·¢Éś±¬ÕØ”£Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
(1) ŌŚ¼īŠŌČÜŅŗÖŠNaClO2±Č½ĻĪȶØ,ĖłŅŌĪüŹÕĖžÖŠÓ¦Ī¬³ÖNaOHÉŌ¹żĮæ,ÅŠ¶ĻNaOHŹĒ·ń¹żĮæµÄ¼ņµ„ŹµŃé·½·ØŹĒ____________”£
(2)ĪüŹÕĖžÖŠĪŖ·ĄÖ¹NaClO2±»»¹Ō³ÉNaCl,ĖłÓĆ»¹Ō¼ĮµÄ»¹ŌŠŌÓ¦ŹŹÖŠ,³żÓĆH2O2Ķā£¬»¹æÉŅŌŃ”ŌńµÄ»¹Ō¼ĮŹĒ______(Ģī“śŗÅ£©”£
a£®Na2O2 b£®Na2S c£®FeCl2 d£®Ģś·Ū
(3)NaClO2ČÜŅŗÓėFeCl2ČÜŅŗĻąÓö£¬ÓŠ“óĮæŗģŗÖÉ«³Įµķ²śÉś,øĆ·“Ó¦µÄĄė×Ó·½³ĢŹ½ĪŖ______
(4)ĪŖĮĖ²ā¶ØNaClO2 ? 3H2OµÄ“æ¶Č,Č”ÉĻŹöŗĻ³É²śĘ·10 gČÜÓŚĖ®Åä³É500 mLČÜŅŗ,Č”³ö10 mLČÜŅŗӌ׶ŠĪĘæÖŠ,ŌŁ¼ÓČĖ×ćĮæĖį»ÆµÄKIČÜŅŗ£¬³ä·Ö·“Ó¦ŗó¼ÓČĖ2 ~3µĪµķ·ŪČÜŅŗ£¬ÓĆ0£®264 mol/L Na2S2O3±ź×¼ŅŗµĪ¶Ø,׶ŠĪĘæÖŠČÜŅŗ______(ĢīŃÕÉ«±ä»Æ£©£¬ĒŅ°ė·ÖÖÓÄŚ²»·¢Éś±ä»Æ,ĖµĆ÷µĪ¶Ø“ļÖÕµć£¬ÓĆČ„±ź×¼Ņŗ20£®OOmL,ŹŌŃł“æ¶ČŹĒ·ńŗĻøń______(Ģī”°ŗĻøń”±»ņ”°²»ŗĻøń”±,ŗĻøń“æ¶ČŌŚ90%ŅŌÉĻ£©”£ĢįŹ¾£ŗ ”£
ӣ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
ĒåĻ“ŗĶÖĘČŽŹĒ¹č¾§Ę¬ÖĘ×÷µÄÖŲŅŖ²½ÖčÖ®Ņ»£¬¹čʬ»ÆѧĒåĻ“µÄÖ÷ŅŖÄæµÄŹĒ³żČ„¹čʬ±ķĆęŌÓÖŹ£ØČēijŠ©ÓŠ»śĪļ£¬ĪŽ»śŃĪ£¬½šŹō”¢Si”¢SiO2·Ū³¾µČ£©”£³£ÓƵĻÆѧĒåĻ“¼ĮÓŠøß“æĖ®”¢ÓŠ»śČܼĮ”¢Ė«ŃõĖ®”¢ÅØĖį”¢Ēæ¼īµČ”£ĘäÖŠČ„³ż¹čµÄŃõ»ÆĪļ£¬Ķس£ÓĆŅ»¶ØÅØ¶ČµÄHFČÜŅŗ£¬ŹŅĪĀĢõ¼žĻĀ½«¹čʬ½žÅŻ1ÖĮŹż·ÖÖÓ”£ÖĘČŽŹĒŌŚ¹čʬ±ķĆęŠĪ³É½š×ÖĖžŠĪµÄČŽĆę£¬Ōö¼Ó¹č¶ŌĢ«Ńō¹āµÄĪüŹÕ”£µ„¾§ÖĘČŽĶس£ÓĆNaOH£¬Na2SiO3µČ»ģŗĻČÜŅŗŌŚ75”«90”ę·“Ó¦25”«35 min£¬Š§¹ūĮ¼ŗĆ”£
»Ų“šĻĀĮŠĪŹĢā
£Ø1£©ÄÜ·ńÓĆ²£Į§ŹŌ¼ĮĘæĄ“Ź¢HFČÜŅŗ£¬ĪŖŹ²Ć“?ÓĆ»Æѧ·½³ĢŹ½¼ÓŅŌ½āŹĶ £»
£Ø2£©Š“³ö¾§Ę¬ÖĘČŽ·“Ó¦µÄĄė×Ó·½³ĢŹ½ £¬¶Ōµ„¾§ÖĘČŽ1990Äź»Æѧ¼ŅSeidelĢį³öĮĖŅ»Öֵĵē»ÆŃ§Ä£ŠĶ£¬ĖūÖø³öSiÓėNaOHČÜŅŗµÄ·“Ó¦£¬Ź×ĻČŹĒSiÓėOHŅ»·“Ó¦£¬Éś³ÉSiO44Ņ»£¬Č»ŗóSiO44Ņ»ŃøĖŁĖ®½āÉś³ÉH4SiO4”£»łÓŚ“ĖŌĄķ·ÖĪö·“Ó¦ÖŠŃõ»Æ¼ĮĪŖ ”£
£Ø3£©±¾Š£»ÆѧŠĖȤŠ”×éĶ¬Ń§£¬ĪŖŃéÖ¤SeidelµÄĄķĀŪŹĒ·ńÕżČ·£¬Ķź³ÉŅŌĻĀŹµŃé£ŗ
| | ŹµŃéŹĀŹµ |
| ŹĀŹµŅ» | Ė®ÕōĘūŌŚ600”ꏱæÉŹ¹·Ūĩד¹č»ŗĀżŃõ»Æ²¢·Å³öĒāĘų”£ |
| ŹĀŹµ¶ž | Ź¢·ÅÓŚ²¬»ņŹÆÓ¢Ę÷ĆóÖŠµÄ“æĖ®³¤Ź±¼ä¶Ō·Ūĩד»¹Ō¹čĪŽøÆŹ“×÷ÓĆ”£ |
| ŹĀŹµČż | ĘÕĶز£Į§Ę÷ĆóÖŠµÄĖ®½öŅņŗ¬ÓŠ“Ó²£Į§ÖŠČܳöµÄĪ¢ĮæµÄ¼ī±ćæÉŹ¹·Ūĩד¹čŌŚĘäÖŠ»ŗĀżČܽā”£ |
| ŹĀŹµĖÄ | ŌŚŅ°Ķā»·¾³Ąļ£¬ÓĆ½Ļøß°Ł·Ö±ČµÄ¹čĢś·ŪÓėøÉŌļµÄCa(OH)2ŗĶNaOH£¬µć×ÅŗóģĖÉÕ£¬æɾēĮŅ·Å³öH2”£ |
| ŹĀŹµĪå | 1g£Ø0.036mo1£©SiŗĶ20mLŗ¬ÓŠlgNaOH£Ø0.025mol£©µÄČÜŅŗ£¬Š”ŠÄ¼ÓČČ£ØÉŌĪ¢Ō¤ČČ£©£¬ŹÕ¼Æµ½Ō¼1700mL H2£¬ŗܽӽüĄķĀŪÖµ£Ø1600mL£©”£ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
£Ø13·Ö£©Ö±½ÓÅŷŵŖŃõ»ÆĪļ»įŠĪ³ÉĖįÓź”¢Īķö²£¬“߻ƻ¹Ō·ØŗĶŃõ»ÆĪüŹÕ·ØŹ¦³£ÓƵē¦Ąķ·½·Ø”£
£Ø1£©ÓĆ»Æѧ·½³ĢŹ½±ķŹ¾NO2ŠĪ³ÉĖįÓźµÄ¹ż³Ģ____________________________
£Ø2£©ĄūÓĆNH3ŗĶCH4µČĘųĢå³żČ„ŃĢĘųÖŠµÄµŖŃõ»ÆĪļ”£
¢ŁNH3µÄµē×ÓŹ½
¢ŚČČĪČ¶ØŠŌ NH3 CH4£ØĢī”°£¾”±=”°<”±£©
¢ŪŅŃÖŖ£ŗCH4(g)+2O2(g)=CO2(g)+2H20(l) ”÷H1=akJ/mol£» Óū¼ĘĖć·“Ó¦CH4(g)+4NO(g)=CO2(g)+2H20(l)+2N2(g)µÄģŹ±ä ”÷H2Ōņ»¹ŠčŅŖ²éŃÆij·“Ó¦µÄģŹ±ä”÷H3£¬µ±·“Ó¦ÖŠø÷ĪļÖŹµÄ»Æѧ¼ĘĮæŹżÖ®±ČĪŖ×ī¼ņÕūŹż±ČŹ±£¬”÷H3=bkJ/mol£¬øĆ·“Ó¦µÄČČ»Æѧ·½³ĢŹ½ŹĒ__________________________¾Ż“Ė¼ĘĖć³ö”÷H2= kJ/mol £ØÓĆŗ¬a”¢bµÄŹ½×Ó±ķŹ¾£©”£
£Ø3£©ĄūÓĆŃõ»Æ·Ø³żČ„ŃĢĘųÖŠµÄµŖŃõ»ÆĪļ”£Ź×ĻČĻņµŖŃõ»ÆĪļÖŠ²¹³äŃõĘų£¬Č»ŗ󽫻ģŗĻĘųĢåĶØČėŹÆ»ŅČéÖŠ£¬Ź¹Ö®×Ŗ»ÆĪŖĻõĖįøĘ”£ŅŃÖŖijµŖŃõ»ÆĪļÓÉNOŗĶNO2×é³É£¬ĒŅn(NO)£ŗn(NO2)=1£ŗ3.Š“³öŃõ»ÆĪüŹÕ·Ø³żČ„µŖŃõ»ÆĪļµÄ»Æѧ·½³ĢŹ½______________________________________ĻÖÓŠ±źæöĻĀµÄO233.6ml£¬ĄķĀŪÉĻæÉŅŌ“¦ĄķøƵŖŃõ»ÆĪļ______ml£Ø±ź×¼×“æöĻĀ£©”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĪŹ“šĢā
£Ø15·Ö£©
Ä³Ń§Ļ°Š”×éŅĄ¾ŻSO2¾ßÓŠ»¹ŌŠŌ£¬ĶĘ²āSO2Äܱ»Cl2Ńõ»ÆÉś³ÉSO2Cl2”£
²éŌÄ׏ĮĻ£ŗSO2Cl2³£ĪĀĻĀĪŖĪŽÉ«ŅŗĢ壬¼«Ņ×Ė®½ā£¬Óö³±ŹŖæÕĘų»į²śÉś°×Īķ”£
¢ń£®»ÆŗĻĪļSO2Cl2ÖŠSŌŖĖŲµÄ»ÆŗĻ¼ŪŹĒ ”£
¢ņ£®ÓƶžŃõ»ÆĆĢŗĶÅØŃĪĖįÖĘĀČĘųµÄ»Æѧ·½³ĢŹ½ŹĒ ”£
¢ó£®ŌŚŹÕ¼ÆĀČĘųĒ°£¬Ó¦ŅĄ“ĪĶعżŹ¢ÓŠ±„ŗĶŹ³ŃĪĖ®ŗĶ µÄĻ“ĘųĘ攣
¢ō£®ÓĆČēĶ¼ĖłŹ¾×°ÖĆŹÕ¼ÆĀśCl2£¬ŌŁĶØČėSO2£¬¼ÆĘųĘæÖŠĮ¢¼“²śÉśĪŽÉ«ŅŗĢ壬
³ä·Ö·“Ó¦ŗ󣬽«ŅŗĢåŗĶŹ£ÓąĘųĢå·ÖĄė£¬½ųŠŠČēĻĀŃŠ¾æ”£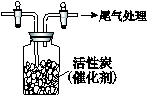
£Ø1£©ŃŠ¾æ·“Ó¦µÄ²śĪļ”£ĻņĖłµĆŅŗĢåÖŠ¼ÓĖ®£¬³öĻÖ°×Īķ£¬Õńµ“”¢¾²ÖƵƵ½ĪŽÉ«ČÜŅŗ”£¾¼ģŃéøĆČÜŅŗÖŠµÄŅõĄė×ӣسżOH£Ķā£©Ö»ÓŠSO42£”¢Cl£ £¬Ö¤Ć÷ĪŽÉ«ŅŗĢåŹĒSO2Cl2”£
¢Ł Š“³öSO2Cl2ÓėH2O·“Ó¦µÄ»Æѧ·½³ĢŹ½ ”£
¢Ś ¼ģŃéøĆČÜŅŗÖŠCl£µÄ·½·ØŹĒ ”£
£Ø2£©¼ĢŠųŃŠ¾æ·“Ó¦½ųŠŠµÄ³Ģ¶Č”£ÓĆNaOHČÜŅŗĪüŹÕ·ÖĄė³öµÄĘųĢ壬ÓĆĻ”ŃĪĖįĖį»Æŗó£¬ŌŁµĪ¼ÓBaCl2ČÜŅŗ£¬²śÉś°×É«³Įµķ”£
¢Ł øĆ°×É«³ĮµķµÄ³É·ÖŹĒ ”£
¢Ś Š“³öSO2ÓėCl2·“Ó¦µÄ»Æѧ·½³ĢŹ½£¬²¢²ūŹöĄķÓÉ______”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŹµŃéĢā
£Ø16·Ö£©»šĮ¦·¢µēŌŚĪŅ¹śµÄÄÜŌ“ĄūÓĆÖŠÕ¼½Ļ“ó±ČÖŲ£¬µ«ŹĒÅŷųöµÄSO2»įŌģ³ÉŅ»ĻµĮŠ»·¾³ŗĶÉśĢ¬ĪŹĢā£¬Ö±½ÓÅÅ·Åŗ¬SO2µÄŃĢĘų»įŠĪ³ÉĖįÓź£¬Ī£ŗ¦»·¾³”£
£Ø1£©ÓĆ»Æѧ·½³ĢŹ½±ķŹ¾SO2ŠĪ³ÉĮņĖįŠĶĖįÓźµÄ·“Ó¦£ŗ ”£2·Ö
£Ø2£©¹¤ŅµÉĻÓĆNa2SO3ČÜŅŗĪüŹÕŃĢĘųÖŠµÄSO2”£½«ŃĢĘųĶØČė1.0 mol”¤L-1µÄNa2SO3ČÜŅŗ£¬ČÜŅŗpH²»¶Ļ¼õŠ””£µ±ČÜŅŗpHŌ¼ĪŖ6Ź±£¬ĪüŹÕSO2µÄÄÜĮ¦ĻŌÖųĻĀ½µ£¬Ó¦øü»»ĪüŹÕ¼Į”£
¢Ł “ĖŹ±ČÜŅŗÖŠc(SO32ØC)µÄÅØ¶ČŹĒ0.2 mol”¤L-1£¬ŌņČÜŅŗÖŠc(HSO3ØC)ŹĒ_______mol?L-1”£
¢Ś ĻņpHŌ¼ĪŖ6µÄĪüŹÕ¼ĮÖŠĶØČė×ćĮæµÄO2£¬æɽ«ĘäÖŠµÄNaHSO3×Ŗ»ÆĪŖĮ½ÖÖĪļÖŹ£¬·“Ó¦µÄ»Æѧ·½³ĢŹ½ŹĒ ”£2·Ö
¢Ū Ä³ŃŠ¾æŠ”×éĪŖĢ½¾æĢįøßŗ¬ĮņŃĢĘųÖŠSO2µÄĪüŹÕŠ§ĀŹµÄ“ėŹ©£¬Ä£ÄāŹµŃéĪüŹÕŗ¬ĮņŃĢĘų£¬ŹµŃé½į¹ūČēĶ¼ĖłŹ¾”£Ōņ£ŗ £¬ÓŠĄūÓŚĢįøßSO2µÄĪüŹÕŠ§ĀŹ”£2·Ö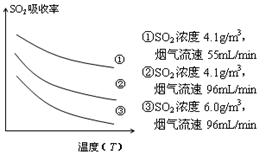
£Ø3£©¹¤³§²Öæā“ę·ÅµÄNa2SO3Ņ©Ę·ŅŃ²æ·Ö±»æÕĘųŃõ»Æ£¬øĆ»ÆѧŠ”×éĻėÓĆŅŃÖŖÅØ¶ČµÄĖįŠŌKMnO4ČÜŅŗĄ“Č·¶ØĘäŗ¬Į棬¾ßĢå²½ÖčČēĻĀ£ŗ
²½Öči””³ĘȔѳʷ1.000 g”£
²½Öčii””½«ŃłĘ·Čܽāŗó£¬ĶźČ«×ŖŅʵ½250 mLČŻĮæĘæÖŠ£¬¶ØČŻ£¬³ä·ÖŅ”ŌČ”£
²½Öčiii””ŅĘČ”25.00 mLѳʷČÜŅŗÓŚ250 mL׶ŠĪĘæÖŠ£¬ÓĆ0.01000 mol”¤L£1 KMnO4±ź×¼ČÜŅŗµĪ¶ØÖĮÖÕµć”£
°“ÉĻŹö²Ł×÷·½·ØŌŁÖŲø“2“Ī”£
¢Ł Š“³ö²½ÖčiiiĖł·¢Éś·“Ó¦µÄĄė×Ó·½³ĢŹ½_________________________________£»
¢Ś ŌŚÅäÖĘ0.01000 mol”¤L£1 KMnO4ČÜŅŗŹ±ČōŃöŹÓ¶ØČŻ£¬Ōņ×īÖÕ²āµĆŅ©Ę·ÖŠNa2SO3µÄŗ¬Įæ________(Ģī”°Ę«“ó”±”¢”°Ę«Š””±»ņ”°ĪŽÓ°Ļģ”±)”£
¢Ū ijĶ¬Ń§Éč¼ĘÓĆĻĀĮŠŅĒĘ÷½ųŠŠµĪ¶ØŹµŃé(¼Š³Ö²æ·ÖĀŌČ„)£¬×īŗĻĄķµÄ×éŗĻŹĒ (Ģī×ÖÄø)”£

A B C D E
¢Ü µĪ¶Ø½į¹ūČēĻĀ±ķĖłŹ¾£ŗ
| µĪ¶Ø“ĪŹż | “ż²āČÜŅŗ µÄĢå»ż/mL | ±ź×¼ČÜŅŗµÄĢå»ż | |
| µĪ¶ØĒ°æĢ¶Č/mL | µĪ¶ØŗóæĢ¶Č/mL | ||
| 1 | 25.00 | 1.02 | 21.03 |
| 2 | 25.00 | 2.00 | 21.99 |
| 3 | 25.00 | 2.20 | 20.20 |
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com