
ĻõĖįŃĪŹÜČČ·Ö½āµÄ²śĪļÓėĻõĖįŃĪµÄ×é³ÉŅŌ¼°ŹÜČȵÄĪĀ¶ČµČŅņĖŲÓŠ¹Ų£®
(1)Ņ»¶ØĢõ¼žĻĀĻõĖįļ§ŹÜČČ·Ö½āµÄ»Æѧ·“Ó¦·½³ĢŹ½ĪŖ£ŗ
NH4NO3£HNO3£«N2£«H2O(Ī“ÅäĘ½)
·“Ó¦ÖŠÉś³ÉµÄŃõ»Æ²śĪļŹĒ__________£»
ŌŚ·“Ó¦ÖŠ±»Ńõ»ÆÓė±»»¹ŌµÄµŖŌ×ÓŹżÖ®±ČĪŖ__________£»
·“Ó¦ÖŠČō²śÉś0.2 mol””N2ĘųĢ壬Ōņ×ŖŅʵē×ÓµÄĪļÖŹµÄĮæŹĒ________mol£®
(2)ijĻõĖįŃĪŹÜČČ·Ö½āµÄ²śĪļĪŖ²»ŗ¬µŖŌŖĖŲµÄ¹ĢĢåĪļÖŹŗĶNO2”¢O2ĘųĢ壬Čō²śÉśNO2ŗĶO2µÄĪļÖŹµÄĮæÖ®±ČĪŖ8”Ć1£¬Ōņ½šŹōŌŖĖŲµÄ¼ŪĢ¬ŌŚ·“Ó¦¹ż³ĢÖŠ________(Ģī”°Éżøß”±”¢”°½µµĶ”±”¢”°²»±ä”±)£®

| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
£Ø1£©Ņ»¶ØĢõ¼žĻĀĻõĖįļ§ŹÜČČ·Ö½āµÄĪ“ÅäĘ½»Æѧ·“Ó¦·½³ĢŹ½ĪŖ£ŗNH4NO3”ŖHNO3+N2+H2OŌŚ·“Ó¦ÖŠ±»Ńõ»ÆÓė±»»¹ŌµÄµŖŌ×ÓŹżÖ®±ČĪŖ_________”£
£Ø2£©Ä³ĻõĖįŃĪŹÜČČ·Ö½āµÄ²śĪļĪŖ²»ŗ¬µŖŌŖĖŲµÄ¹ĢĢåĪļÖŹŗĶNO2”¢O2ĘųĢ壬Čō²śÉśNO2ŗĶO2µÄĪļÖŹµÄĮæÖ®±ČĪŖ8”Ć1£¬Ōņ½šŹōŌŖĖŲµÄ¼ŪĢ¬ŌŚ·“Ó¦¹ż³ĢÖŠ_________£ØĢī”°Éżøß”±”°½µµĶ”±”°²»±ä”±£©”£ČōøĆĻõĖįŃĪµÄ»ÆѧŹ½±ķŹ¾ĪŖM£ØNO3£©2£¬ŌņĘäŹÜČČ·Ö½āµÄ»Æѧ·½³ĢŹ½ĪŖ______________________________”£
£Ø3£©2 mol Cu(NO3)2ŗĶAgNO3»ģŗĻĪļ£¬¼ÓČČŹ¹Ęä·Ö½ā£¬µĆµ½NO2”¢O2Į½ÖÖĘųĢåŗĶCuO”¢Ag¶žÖÖ¹ĢĢ壬½«²śÉśµÄNO2”¢O2»ģŗĻĘųĢå¾Ė®³ä·ÖĪüŹÕŗó£¬Ź£ÓąĘųĢåŹĒ____________________£¬ĘäĪļÖŹµÄĮæ£Ømol£©·¶Ī§ŹĒ_______________________________________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2012-2013ѧğ¼ŖĮÖŹ”¼ŖĮÖŅ»ÖŠøßŅ»11ŌĀŌĀæ¼»ÆѧŹŌ¾ķ£Ø“ų½āĪö£© ĢāŠĶ£ŗ¼ĘĖćĢā
ijĄąĻõĖįŃĪŹÜČČ·Ö½āµÄ²śĪļĪŖ²»ŗ¬µŖŌŖĖŲµÄ¹ĢĢåĪļÖŹŗĶNO2”¢O2ĘųĢå£ŗ
(1)ijÖÖĻõĖįŃĪŹÜČČ·Ö½āŗó£¬Čō²śÉśNO2ŗĶO2µÄĪļÖŹµÄĮæÖ®±ČĪŖ6©U1£¬Ōņ½šŹōŌŖĖŲµÄ¼ŪĢ¬ŌŚ·“Ó¦¹ż³ĢÖŠ_______(Ģī”°Éżøß”±”¢”°½µµĶ”±”¢”°²»±ä”±)
(2) ĻÖČ”mg Cu(NO3)2ĪŽĖ®¾§Ģ壬ĒæČČŹ¹Ęä·Ö½ā£¬µĆµ½NO2”¢O2ŗĶng¹ĢĢ唣½«ĘųĢåÓĆĖ®³ä·ÖĪüŹÕŗ󣬽į¹ū»¹ÓŠĘųĢåŹ£Óą£¬Ķ¬Ź±µĆµ½100mLČÜŅŗ”£Ōņ£ŗ²ŠĮō¹ĢĢå³É·ÖæÉÄÜŹĒ_____________ _______________(ÓĆ»ÆѧŹ½±ķŹ¾)£»ĖłµĆČÜŅŗµÄĪļÖŹµÄĮæÅØ¶ČŹĒ__________________£ØÓĆÖ»ŗ¬mµÄ“śŹżŹ½±ķŹ¾£©£»
(3)(2)ÖŠm = 3.76£¬n = 1.52”£ŹŌĶعż¼ĘĖćČ·¶Ø²ŠĮō¹ĢĢåµÄ³É·Ö¼°ĘäÖŹĮæ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2012-2013ѧğÉĻŗ£ŹŠ±¦É½ĒųøßČżÉĻѧʌʌĩ½Ģѧ֏Įæµ÷ŃŠ»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗĢīæÕĢā
ĻõĖįŃĪŹÜČČ·Ö½āµÄ²śĪļÓėĻõĖįŃĪµÄ×é³ÉŅŌ¼°ŹÜČȵÄĪĀ¶ČµČŅņĖŲÓŠ¹Ų”£
¢ń£®Ņ»¶ØĢõ¼žĻĀĻõĖįļ§ŹÜČČ·Ö½āµÄ»Æѧ·“Ó¦·½³ĢŹ½ĪŖ£ŗNH4NO3”śHNO3+N2 +H2O£ØĪ“ÅäĘ½£©
£Ø1£©øĆ·“Ó¦ÖŠ·¢Éś»¹Ō·“Ó¦µÄ¹ż³ĢŹĒ_______”ś________”£
£Ø2£©·“Ó¦ÖŠ±»Ńõ»ÆÓė±»»¹ŌµÄµŖŌ×ÓŹżÖ®±ČĪŖ__________£»ĻÖÓŠ1mol NH4NO3·Ö½ā£¬Éś³ÉµÄŃõ»Æ²śĪļµÄĪļÖŹµÄĮæŹĒ mol”£
¢ņ£®ĻõĖįļ§ŹÜײ»÷»ņøßĪĀ·¢Éś±¬ÕصĻÆѧ·“Ó¦·½³ĢŹ½ĪŖ£ŗm NH4NO3”śn O2+p N2+q H2O
£Ø3£©ĻĀĮŠ¹ŲÓŚøĆ·½³ĢŹ½ÖŠ»Æѧ¼ĘĮæŹżµÄĖµ·ØÕżČ·µÄŹĒ_________£ØŃ”Ģī×ÖÄø±ąŗÅ£©”£
a£®m£½1 b£®n£½2 c£®p£½3 d£®q£½4
£Ø4£©Čō·“Ó¦¹ż³ĢÖŠ¹²×ŖŅĘ3£®01”Į1023øöµē×Ó£¬Ōņ·“Ó¦æɲśÉśĘųĢåµÄĢå»żĪŖ_____£Ø±ź×¼×“æö£©”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2015½ģ¼ŖĮÖŹ”øßŅ»11ŌĀŌĀæ¼»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗ¼ĘĖćĢā
ijĄąĻõĖįŃĪŹÜČČ·Ö½āµÄ²śĪļĪŖ²»ŗ¬µŖŌŖĖŲµÄ¹ĢĢåĪļÖŹŗĶNO2”¢O2ĘųĢå£ŗ
(1)ijÖÖĻõĖįŃĪŹÜČČ·Ö½āŗó£¬Čō²śÉśNO2ŗĶO2µÄĪļÖŹµÄĮæÖ®±ČĪŖ6©U1£¬Ōņ½šŹōŌŖĖŲµÄ¼ŪĢ¬ŌŚ·“Ó¦¹ż³ĢÖŠ_______(Ģī”°Éżøß”±”¢”°½µµĶ”±”¢”°²»±ä”±)
(2) ĻÖČ”mg Cu(NO3)2ĪŽĖ®¾§Ģ壬ĒæČČŹ¹Ęä·Ö½ā£¬µĆµ½NO2”¢O2ŗĶng¹ĢĢ唣½«ĘųĢåÓĆĖ®³ä·ÖĪüŹÕŗ󣬽į¹ū»¹ÓŠĘųĢåŹ£Óą£¬Ķ¬Ź±µĆµ½100mLČÜŅŗ”£Ōņ£ŗ²ŠĮō¹ĢĢå³É·ÖæÉÄÜŹĒ_____________ _______________(ÓĆ»ÆѧŹ½±ķŹ¾)£»ĖłµĆČÜŅŗµÄĪļÖŹµÄĮæÅØ¶ČŹĒ__________________£ØÓĆÖ»ŗ¬mµÄ“śŹżŹ½±ķŹ¾£©£»
(3)(2)ÖŠm = 3.76£¬n = 1.52”£ŹŌĶعż¼ĘĖćČ·¶Ø²ŠĮō¹ĢĢåµÄ³É·Ö¼°ĘäÖŹĮæ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ×ØĻīĢā ĢāŠĶ£ŗŹµŃéĢā
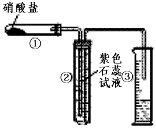
 2Ag2O£«4NO2”ü£«O2”ü£¬ (¢ņ) 2AgNO3
2Ag2O£«4NO2”ü£«O2”ü£¬ (¢ņ) 2AgNO3 2Ag£«2NO2”ü£«O2”ü”£
2Ag£«2NO2”ü£«O2”ü”£ ²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com