
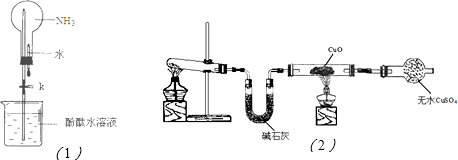
ÓūÓĆĪŽĖ®
CuSO4·ŪÄ©ÅäÖĘ500 mL””0.1 mol”¤L£1””CuSO4ČÜŅŗ£®øł¾ŻĢāŅāĢīæÕ£ŗ(1)ČēĶ¼ĖłŹ¾ŅĒĘ÷£¬ÅäÖĘÉĻŹöČÜŅŗæĻ¶Ø²»ŠčÓƵ½µÄŹĒ________(Ģī×ÖÄø)ÅäÖĘÉĻŹöČÜŅŗ»¹ŠčŅŖµÄ²£Į§ŅĒĘ÷ÓŠ________(ĢīŅĒĘ÷Ćū³Ę)£®

(2)ÅäÖĘøĆČÜŅŗӦєÓĆ________mLČŻĮæĘ棬Ź¹ÓĆČŻĮæĘæÖ®Ē°±ŲŠė½ųŠŠ________£®
(3)ÅäÖĘøĆČÜŅŗÓ¦ÓĆĶŠÅĢĢģĘ½³ĘČ”________gĪŽĖ®
CuSO4£®(4)Ź¹ÓĆČŻĮæĘæÅäÖĘČÜŅŗŹ±£¬ÓÉÓŚ²Ł×÷²»µ±£¬»įŅżĘšĪó²ī£¬ĻĀĮŠĒéæö»įŹ¹ĖłÅäČÜŅŗÅضČĘ«µĶµÄŹĒ________(Ģī±ąŗÅ)£®
¢ŁÓĆĢģĘ½(Ź¹ÓĆÓĪĀė)³ĘĮæŹ±£¬±»³ĘĮæĪļÓėķĄĀėµÄĪ»ÖĆ·Åµßµ¹ĮĖ
¢ŚĻņČŻĮæĘæÖŠ×ŖŅĘČÜŅŗŹ±²»É÷½«ŅŗµĪČ÷ŌŚČŻĮæĘæĶāĆę
¢ŪČÜŅŗ×ŖŅʵ½ČŻĮæĘæŗó£¬ÉÕ±¼°²£Į§°ōĪ“ÓĆÕōĮóĖ®Ļ“µÓ
¢Ü×ŖŅĘČÜŅŗĒ°ČŻĮæĘæÄŚÓŠÉŁĮæÕōĮóĖ®
¢Ż¶ØČŻŹ±£¬ŃöŹÓČŻĮæĘæµÄæĢ¶ČĻß
¢Ž¶ØČŻŗóŅ”ŌČ£¬·¢ĻÖŅŗĆę½µµĶ£¬ÓÖ²¹¼ÓÉŁĮæĖ®£¬ÖŲŠĀ“ļµ½æĢ¶ČĻß
 Ņ»æĪŅ»Į·æĪŹ±“ļ±źĻµĮŠ“š°ø
Ņ»æĪŅ»Į·æĪŹ±“ļ±źĻµĮŠ“š°ø ĘŚÄ©±¦µäµ„ŌŖ¼ģ²ā·ÖĄąø“Ļ°¾ķĻµĮŠ“š°ø
ĘŚÄ©±¦µäµ„ŌŖ¼ģ²ā·ÖĄąø“Ļ°¾ķĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŌĶĮĄķ½ā
| ||
| ||
| ||
| ||
 £»
£» £»
£»| H+ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
£Ø9·Ö£©ÓūÓĆĪŽĖ®CuSO4·ŪÄ©ÅäÖĘ500mL0.1mol”¤L”Ŗ1 CuSO4ČÜŅŗ”£øł¾ŻĢāŅāĢīæÕ£ŗ
£Ø1£©ČēĶ¼ĖłŹ¾ŅĒĘ÷£¬ÅäÖĘÉĻŹöČÜŅŗæĻ¶Ø²»ŠčÓƵ½µÄŹĒ £ØĢī×ÖÄø£©ÅäÖĘÉĻŹöČÜŅŗ»¹ŠčŅŖµÄ²£Į§ŅĒĘ÷ÓŠ £ØĢīŅĒĘ÷Ćū³Ę£©”£
£Ø2£©ÅäÖĘøĆČÜŅŗӦєÓĆ mLČŻĮæĘ棬Ź¹ÓĆČŻĮæĘæÖ®Ē°±ŲŠė½ųŠŠ ”£
£Ø3£©ÅäÖĘøĆČÜŅŗÓ¦ÓĆĶŠÅĢĢģĘ½³ĘČ” gĪŽĖ®CuSO4”£
£Ø4£©Ź¹ÓĆČŻĮæĘæÅäÖĘČÜŅŗŹ±£¬ÓÉÓŚ²Ł×÷²»µ±£¬»įŅżĘšĪó²ī£¬ĻĀĮŠĒéæö»įŹ¹ĖłÅäČÜŅŗÅضČ
Ę«µĶµÄŹĒ £ØĢī±ąŗÅ£©”£
¢ŁÓĆĢģĘ½£ØŹ¹ÓĆÓĪĀė£©³ĘĮæŹ±£¬±»³ĘĮæĪļÓėķĄĀėµÄĪ»ÖĆ·Åµßµ¹ĮĖ
¢ŚĻņČŻĮæĘæÖŠ×ŖŅĘČÜŅŗŹ±²»É÷½«ŅŗµĪČ÷ŌŚČŻĮæĘæĶāĆę
¢ŪČÜŅŗ×ŖŅʵ½ČŻĮæĘæŗó£¬ÉÕ±¼°²£Į§°ōĪ“ÓĆÕōĮóĖ®Ļ“µÓ
¢Ü×ŖŅĘČÜŅŗĒ°ČŻĮæĘæÄŚÓŠÉŁĮæÕōĮóĖ®
¢Ż¶ØČŻŹ±£¬ŃöŹÓČŻĮæĘæµÄæĢ¶ČĻß
¢Ž¶ØČŻŗóŅ”ŌČ£¬·¢ĻÖŅŗĆę½µµĶ£¬ÓÖ²¹¼ÓÉŁĮæĖ®£¬ÖŲŠĀ“ļµ½æĢ¶ČĻß
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2011-2012ѧğ½ĖÕŹ”ĪŽĪżŹŠĀåÉē֊ѧøßŅ»ÉĻŃ§ĘŚĘŚÖŠæ¼ŹŌ»ÆѧŹŌ¾ķ ĢāŠĶ£ŗŹµŃéĢā
£Ø9·Ö£©ÓūÓĆĪŽĖ®CuSO4·ŪÄ©Åä ÖĘ500mL 0.1mol”¤L”Ŗ1 CuSO4ČÜŅŗ”£øł¾ŻĢāŅāĢīæÕ£ŗ
ÖĘ500mL 0.1mol”¤L”Ŗ1 CuSO4ČÜŅŗ”£øł¾ŻĢāŅāĢīæÕ£ŗ
£Ø1£©ČēĶ¼ĖłŹ¾ŅĒĘ÷£¬ÅäÖĘÉĻŹöČÜŅŗæĻ¶Ø²»ŠčÓƵ½µÄŹĒ £ØĢī×ÖÄø£©ÅäÖĘÉĻŹöČÜŅŗ»¹ŠčŅŖµÄ²£Į§ŅĒĘ÷ÓŠ  £ØĢīŅĒĘ÷Ćū³Ę£©”£
£ØĢīŅĒĘ÷Ćū³Ę£©”£
£Ø2£©ÅäÖĘøĆČÜŅŗӦєÓĆ mLČŻĮæĘ棬Ź¹ÓĆČŻĮæĘæÖ®Ē°±ŲŠė½ųŠŠ ”£
£Ø3£©ÅäÖĘøĆČÜŅŗ Ó¦ÓĆĶŠÅĢĢģ
Ó¦ÓĆĶŠÅĢĢģ Ę½³Ę
Ę½³Ę Č” gĪŽĖ®CuSO4”£
Č” gĪŽĖ®CuSO4”£
£Ø4£©Ź¹ÓĆČŻĮæĘæÅäÖĘČÜŅŗŹ±£¬ÓÉÓŚ²Ł×÷²»µ±£¬»įŅżĘšĪó²ī£¬ĻĀĮŠĒéæö»įŹ¹ĖłÅäČÜŅŗÅضČ
Ę«µĶµÄŹĒ £ØĢī±ąŗÅ£©”£
¢ŁÓĆĢģĘ½£ØŹ¹ÓĆÓĪĀė£©³ĘĮæŹ±£¬±»³ĘĮæĪļÓėķĄĀėµÄĪ»ÖĆ·Åµßµ¹ĮĖ
¢ŚĻņČŻĮæĘæÖŠ×ŖŅĘČÜŅŗŹ±²»É÷½«ŅŗµĪČ÷ŌŚČŻĮæĘæĶāĆę
¢ŪČÜŅŗ×ŖŅʵ½ČŻĮæĘæŗó£¬ÉÕ±¼°²£Į§°ōĪ“ÓĆÕōĮóĖ®Ļ“µÓ
¢Ü×ŖŅĘČÜŅŗĒ°ČŻĮæĘæÄŚÓŠÉŁĮæÕōĮóĖ®
¢Ż¶ØČŻŹ±£¬ŃöŹÓČŻĮæĘæµÄæĢ¶ČĻß
¢Ž¶ØČŻŗóŅ”ŌČ£¬·¢ĻÖŅŗĆę½µµĶ£¬ÓÖ²¹¼ÓÉŁĮæĖ®£¬ÖŲŠĀ“ļµ½æĢ¶ČĻß
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2014½ģ½ĖÕŹ”ĪŽĪżŹŠøßŅ»ÉĻŃ§ĘŚĘŚÖŠæ¼ŹŌ»ÆѧŹŌ¾ķ ĢāŠĶ£ŗŹµŃéĢā
£Ø9·Ö£©ÓūÓĆĪŽĖ®CuSO4·ŪÄ©ÅäÖĘ500mL 0.1mol”¤L”Ŗ1 CuSO4ČÜŅŗ”£øł¾ŻĢāŅāĢīæÕ£ŗ
£Ø1£©ČēĶ¼ĖłŹ¾ŅĒĘ÷£¬ÅäÖĘÉĻŹöČÜŅŗæĻ¶Ø²»ŠčÓƵ½µÄŹĒ £ØĢī×ÖÄø£©ÅäÖĘÉĻŹöČÜŅŗ»¹ŠčŅŖµÄ²£Į§ŅĒĘ÷ÓŠ £ØĢīŅĒĘ÷Ćū³Ę£©”£
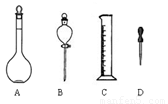
£Ø2£©ÅäÖĘøĆČÜŅŗӦєÓĆ mLČŻĮæĘ棬Ź¹ÓĆČŻĮæĘæÖ®Ē°±ŲŠė½ųŠŠ ”£
£Ø3£©ÅäÖĘøĆČÜŅŗÓ¦ÓĆĶŠÅĢĢģĘ½³ĘČ” gĪŽĖ®CuSO4”£
£Ø4£©Ź¹ÓĆČŻĮæĘæÅäÖĘČÜŅŗŹ±£¬ÓÉÓŚ²Ł×÷²»µ±£¬»įŅżĘšĪó²ī£¬ĻĀĮŠĒéæö»įŹ¹ĖłÅäČÜŅŗÅضČ
Ę«µĶµÄŹĒ £ØĢī±ąŗÅ£©”£
¢ŁÓĆĢģĘ½£ØŹ¹ÓĆÓĪĀė£©³ĘĮæŹ±£¬±»³ĘĮæĪļÓėķĄĀėµÄĪ»ÖĆ·Åµßµ¹ĮĖ
¢ŚĻņČŻĮæĘæÖŠ×ŖŅĘČÜŅŗŹ±²»É÷½«ŅŗµĪČ÷ŌŚČŻĮæĘæĶāĆę
¢ŪČÜŅŗ×ŖŅʵ½ČŻĮæĘæŗó£¬ÉÕ±¼°²£Į§°ōĪ“ÓĆÕōĮóĖ®Ļ“µÓ
¢Ü×ŖŅĘČÜŅŗĒ°ČŻĮæĘæÄŚÓŠÉŁĮæÕōĮóĖ®
¢Ż¶ØČŻŹ±£¬ŃöŹÓČŻĮæĘæµÄæĢ¶ČĻß
¢Ž¶ØČŻŗóŅ”ŌČ£¬·¢ĻÖŅŗĆę½µµĶ£¬ÓÖ²¹¼ÓÉŁĮæĖ®£¬ÖŲŠĀ“ļµ½æĢ¶ČĻß
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com