
| A��Ũ�����ӷ����ӳ����г����Ũ����Ҫ�ܷⱣ�� |
| B�������Ũ�����к�ǿ�������ԣ�����ֱ�ӱ��������������� |
| C���ڴ���ʱ�����ʢװŨ����IJ����ڽ�ˮ�����ܻᷢ����ը |
| D�����Ṥҵβ����Ȼ���������Ĺ����ŷŻ��������ЧӦ |
 ��ְٷְټ�����Ԫ��ĩ���Ծ�ϵ�д�
��ְٷְټ�����Ԫ��ĩ���Ծ�ϵ�д� Сѧ��ĩ���Ծ�ϵ�д�
Сѧ��ĩ���Ծ�ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
| ʵ����� | ����ͽ��� |
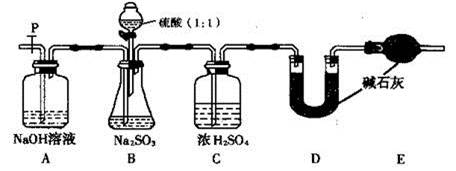
| ����1��ȡ������Ʒ���Թ��У�������������ˮ����ܽ⣬�ٵμ�H2SO4�ữ��KMnO4��Һ�� | ������KMnO4��Һ���Ϻ�ɫ��Ϊ��ɫ �ٽ��ۣ���Ʒ���� ���ӣ�����1�������� ����ɫ�����ӷ���ʽΪ�� �� |
| ����2����ȡ������Ʒ���Թ��У�������������ˮ����ܽ⣬�ٵμ�ϡHCl��ʹ��Һ�����ԣ��ٵμ�����BaCl2��Һ�� | �������� �� ���ۣ�����2������ |
| ���� | ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����

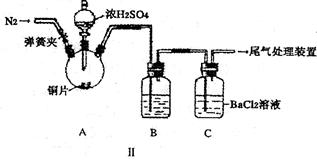
| �������� | �����뾧�������� | ���Ⱥ���������������� | |
| ��һ�γ��� | �ڶ��γ��� | ||
| 11.710g | 22.700g | 18.621g | a |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
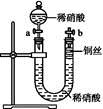
| | �� | ʵ���� |
| a | Ũ���� | ���Ⱥ�����Ӧ,������ɫ�̼������� |
| b | ϡ���� | ����Ҳ��������Ӧ |
| c | Ũ���� | �����ȼ�������Ӧ,��������ɫ���� |
| d | ϡ���� | �ȷ�����Ӧ,������ɫ���� |

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
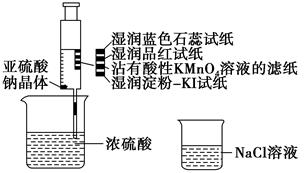
| A����ɫʯ����ֽ�ȱ�����ɫ |
| B��Ʒ����ֽ��մ������KMnO4��Һ����ֽ����ɫ��֤��SO2����Ư���� |
| C��ʪ�����KI��ֽδ����˵��SO2������������I2 |
| D��NaCl��Һ�����ڳ�ȥʵ���ж����SO2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
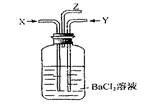
A��ϴ��ƿ�в����ij����� |
B����Z���ܳ������������� |
C��ϴ��ƿ�в����ij����� |
| D����Z���ܿ��к���ɫ������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����1 mol H2SO4��Ũ��������n(H��)��2n(SO42-)��2 mol |
| B����1 mol H2SO4��Ũ�����������п��ȫ��Ӧ��ת�Ƶĵ���Ϊ2 mol |
| C������2.0 mol��L��1�����ᣬ������ʱ���ӣ���������ҺŨ��ƫ��(������������ȷ) |
| D����ͭ�缫���2.0 mol��L��1�����ᣬ��������������������֮��Ϊ2��1 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��SO2��ʹƷ����Һ��ɫ����ΪSO2���������ԣ�������Ҳ��ʹ���ָʾ����ɫ |
| B��������������ͼ�̬��2�ۣ����ֻ���л�ԭ�� |
| C��Ũ����ʹľ��������ڣ�������Ũ�������ˮ�� |
| D��BaSO4���������͡�����X����� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
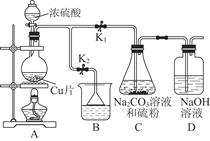
| ��� | 1 | 2 | 3 | 4 |
| ����Na2S2O3�� | | | | |
| ��Һ�����/mL | 18.02 | 17.98 | 18.00 | 20.03 |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com