��2010?������һģ���ס��ҡ����������ɶ�����Ԫ����ɵ����ʣ�����֮���������ת����ϵ����+�ҡ���+��
��1��ת����ϵ�����漰�ķ�ӦΪ��������ԭ��Ӧ������Ϊˮ��
������Ϊ��״���壬��Ϊ��ȼ�����壬������ڼȺ��м��Լ��ֺ��зǼ��Լ�������ĵ���ʽ��
��
����������N��ClԪ����ɵĻ��������ӽṹģ��
��ͼ1��ʾ��������Ư���ԣ������ClԪ�صĻ��ϼ���
+1
+1
��
��2��ת����ϵ�����漰�ķ�ӦΪ������ԭ��Ӧ������Ϊˮ��
�����ͱ���ͬ����Ԫ����ɵĵ��ʣ�����ɼ�Ԫ��λ�ڵ������ڣ��˷�Ӧ�����ӷ���ʽ��
2Na+2H2O=2Na++2OH-+H2��
2Na+2H2O=2Na++2OH-+H2��
��
�������Ͷ�������һ�������»�ԭCuO���˷�Ӧ�Ļ�ѧ����ʽ��
��
����������N��OԪ����ɵ���̬���ʣ��ʺ���ɫ����3.36g Fe�ӵ�һ��������Һ�У��ռ���1.12L���嶡��������Ϊ��״��������Ӧ�����ӷ���ʽ��
6Fe+20HNO3=3Fe��NO3��2+3Fe��NO3��3+5NO+10H2O
6Fe+20HNO3=3Fe��NO3��2+3Fe��NO3��3+5NO+10H2O
��
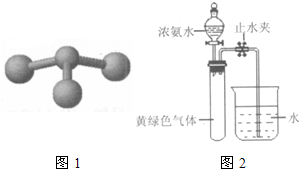
��3��Hofmann��������ת����ϵ�ⶨ�����ӵ���ɣ�������ͼ2��ʾ��װ�ý���ʵ�飬��Һ©���Ļ���������Ũ��ˮ�������ٷ�ӦΪֹ���رշ�Һ©���Ļ��������ָ������£���ֹˮ�У��Թ���Һ��������
����
�ٵ���Ũ��ˮһ��ʱ����Թ��ڷ�����Ӧ�Ļ�ѧ����ʽ��
8NH3+3C12=N2+6NH4Cl
8NH3+3C12=N2+6NH4Cl
��
��֤���Թ��ڡ����ٷ�Ӧ����ʵ�������������
�������Ĵ������̳��������μ�Ũ��ˮ�����ٲ�������
�������Ĵ������̳��������μ�Ũ��ˮ�����ٲ�������
��


 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�



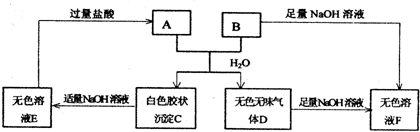
 ��2010?������һģ���ס��ҡ����������ɶ�����Ԫ����ɵ����ʣ�����֮���������ת����ϵ����+�ҡ���+��
��2010?������һģ���ס��ҡ����������ɶ�����Ԫ����ɵ����ʣ�����֮���������ת����ϵ����+�ҡ���+��

 �ס����������ɶ�����Ԫ����ɵ����ʣ�����֮���������ת����ϵ��
�ס����������ɶ�����Ԫ����ɵ����ʣ�����֮���������ת����ϵ��