
��11�֣������Ͱ����ڳ����»�ϼ��ɷ�����Ӧ��ij��ȤС��ͬѧΪ̽������������������백���ķ�Ӧ�����������װ�ã�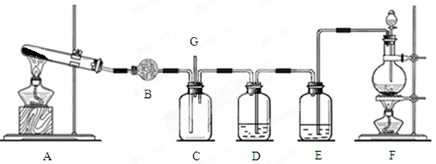
��ش�
��1��Aװ���е��Լ��� ��Bװ�õ����������� ��
��2��д��װ��F�з�����Ӧ�����ӷ���ʽ�� ������Ӧ���ܲ���560mL�������壨��״���£�����������Ҫ12mol/L��Ũ���� mL��
��3��װ��E��ʢ�ŵ��Լ��� ���������� ��
��4��װ��C���а��̲�������д����Ӧ�Ļ�ѧ����ʽ ��
��5������װ��C��G���ݳ���β���к�������Cl2��Ϊ��ֹ����Ⱦ�������ɽ�β��ͨ��ʢ�� ��ϴ��ƿ��
��1���Ȼ�李��������ƣ����Ȼ�李���ʯ�ң����Ȼ�李������ƣ��� �����
��2��MnO2 + 4 H+ + 2 Cl�� Mn2+ + Cl2 ��+ 2 H2O �� 8.3 ����8��8.33��25/3��
Mn2+ + Cl2 ��+ 2 H2O �� 8.3 ����8��8.33��25/3��
��3������ʳ��ˮ ��ȥ�����е�HCl����
��4��8NH3+3Cl2 ="==" N2+6NH4Cl ����2NH3+3Cl2 ="==" N2+6HCl NH3+HCl="=" NH4Cl��
��5������������Һ
���������������1��Aװ������ʵ������ȡ��������ʵ������һ���������������ȡ����������Aװ���е��Լ����Ȼ�李��������ơ�Bװ�õ��������Ƹ���ܡ���2����װ��F����ȡ������������Ӧ�����ӷ���ʽ��MnO2 + 4 H+ + 2 Cl�� Mn2+ + Cl2��+ 2 H2O����n(Cl2)=0.560L��22.4L/mol=0.025mol������ֻ��Ũ����˷�����Ӧ������������Ҫ12mol/L��Ũ���ᣨ0.025mol��4����12mol/L=0.0083L=8.3ml����3������Ũ�����лӷ��ԣ������������ں�������HCl����װ��E��ʢ�ŵ��Լ��dz�ȥ�����е�HCl�ġ�Ӧ��Ϊ����ʳ��ˮ����4����װ��C�з�����Ӧ8NH3+3Cl2=N2+6NH4Cl��������ɫ����NH4Cl.��˻ῴ���а��̲�������5������װ��C��G���ݳ���β���к�������Cl2������Cl2����������ʷ�Ӧ������Ϊ��ֹ����Ⱦ�������ɽ�β��ͨ��ʢ������������Һ���Լ�ƿ�С�
Mn2+ + Cl2��+ 2 H2O����n(Cl2)=0.560L��22.4L/mol=0.025mol������ֻ��Ũ����˷�����Ӧ������������Ҫ12mol/L��Ũ���ᣨ0.025mol��4����12mol/L=0.0083L=8.3ml����3������Ũ�����лӷ��ԣ������������ں�������HCl����װ��E��ʢ�ŵ��Լ��dz�ȥ�����е�HCl�ġ�Ӧ��Ϊ����ʳ��ˮ����4����װ��C�з�����Ӧ8NH3+3Cl2=N2+6NH4Cl��������ɫ����NH4Cl.��˻ῴ���а��̲�������5������װ��C��G���ݳ���β���к�������Cl2������Cl2����������ʷ�Ӧ������Ϊ��ֹ����Ⱦ�������ɽ�β��ͨ��ʢ������������Һ���Լ�ƿ�С�
���㣺���鰱����������ʵ������ȡ�������Լ���ѡ��Ӧԭ����β���Ĵ�����֪ʶ��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��ʴ���
��14�֣�
�о�С������ͼװ����ȡCl2��֤�������������к���HCl��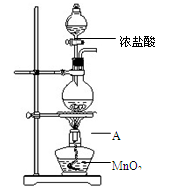
��1������A������Ϊ ��A�з�Ӧ�����ӷ���ʽΪ ��
��2����ͬѧ��A�в���������ͨ��������Һ��
| ʵ����� | �Լ� | ���� |
| a | ��ɫʯ����Һ | |
| b | AgNO3��Һ | ���ְ�ɫ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��16�֣�ij�о���ѧϰС��Ϊ�о�������Ũ���ᷴӦ����������ijɷݲ��ⶨ������ĺ���������������ʵ�飺��ʵ���Ʋ⡿������������Ũ���ᷴӦ������SO2��H2�������塣
��1����С���������Ʋ�������ǣ� ��
��ʵ������a����ѡ���ҩƷ�����ۡ�Ũ���ᡢ����ͭ��ĩ��0.2 mol/L��H2C2O4����Һ��0.1 mol/L������KMnO4����Һ�����ָʾ����
b��ʵ��װ����Ƽ���װ(���ȼ��г�װ�þ�����ȥ)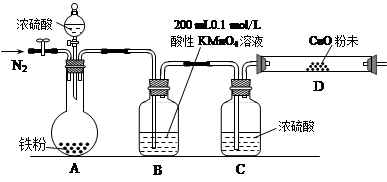
��ʵ����̼����ݴ�����
��2��B�з�����Ӧ�����ӷ���ʽΪ�� ��
��3��ʵ��ǰ��ͨ��һ��ʱ���N2����Ŀ���� ��
��4��B��C��D����ʢ�Լ�����������֤ʵ���ɵ�������ͬʱ����SO2��H2��������
��
��5��A�з�Ӧ��������ͨN2ʹA�����ɵ�����ȫ���ϳ�����B��D�з�Ӧ��ȫ���Ⱥ�����ȡ��B�з�Ӧ�����Һ����ƿ�У�ÿ��ȡ��25 mL����H2C2O4����Һ���вⶨ��
��H2C2O4����Һ������KMnO4��Һ��Ӧ�����ӷ���ʽ���£��뽫�÷���ʽ��ɲ���ƽ��
( )H2C2O4+( )MnO4- +( )H+ ��( )Mn2+ +( ) H2O+( ) ( )
�ڷ�Ӧ��ȫ�������� ��
���ظ��ⶨ���Σ�ƽ��ÿ�κ���H2C2O4����Һ15.63 mL��������Ũ���ᷴӦ������SO2����
�����ʵ���Ϊ ����������ʵ��ǰ��װ��D����������0.8 g���������������SO2���������Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��16�֣��������볱ʪ����ʯ�ҷ�Ӧ��ȡ����Ư�ۣ�Ҳ����Ũ��Һ��������ʯ��ʯ��Ӧ���ɽ�Ũ��HClO��Һ��
��.���о���ѧϰС����������������Ư�ۣ���ͼ��ʾ����
��1��A������������ ����ʢ�Լ��� ��
��2����ʵ��������Ca(ClO)2����̫�͡����������������Ϸ�����Ҫԭ������U���д�����������Ӧ��
���¶Ƚϸ�ʱ��������ʯ�ҷ�Ӧ������Ca(ClO3)2��Ϊ����˸���Ӧ�ķ������ɲ�ȡ�Ĵ�ʩ�� ���˸���Ӧ�Ļ�ѧ����ʽΪ�� ��
��д����һ������Ӧ�Ļ�ѧ����ʽ ��Ϊ����˸���Ӧ����������BC֮��Ӷ�һ��װ�ã����ڴ�����ķ����л�����װ�ã����������õ��Լ���
��. ���о���ѧϰС���о�������ˮ��ʯ��ʯ�ķ�Ӧ��
�����Թ��м�������Ŀ�״̼��ƣ��ټ���Լ20mL������ˮ����ͼ��ʾ������ַ�Ӧ�����������ݲ�������Һdz����ɫ��ȥ��
�ڹ��ˣ�����Һ������ɫ�����ϣ����������ˮ��Ư���Ը�ǿ��
��Ϊ��ȷ����Ӧ�������Һ��Ϊ���ݣ��ֱ��������ʵ�飺
��һ����ʯ��ˮ��ϣ���������������ɫ������
�ڶ�����ϡ�����ϣ���������������ɫ���壻
�������ݼ��ȣ�������Һ��������д�����ɫ��������� ����⣬����ʵ���в�������ɫ�����ΪCO2����ش�
��3����Ӧ�����õ���ҺƯ������ǿ��ԭ���� ��
��4����������ʵ�����֪���ڵ���Һ�е����ʳ�CaCl2��HClO�⣬������ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ij��ȤС����Ƴ���ͼ��ʾװ�����Ľ��̲��С�ͭ�����ᷴӦ��ʵ�飬��̽����ѧʵ�����ɫ����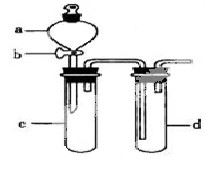
��1��ʵ��ǰ���رջ���b���Թ�d�м�ˮ����û�����ܿڣ������Թ�c��d�Ľ���������c����Ŀ����__________��
��2����d�м�����NaOH��Һ��c�з�һС��ͭƬ���ɷ�Һ©��a��c�м���2mLŨ���ᣬc�з�Ӧ�Ļ�ѧ����ʽ��______________________��
����a��c�м�2mL����ˮ��c�е�ʵ��������_____________��
��3���±�����ȡ����ͭ�����ַ�������������ɫ��ѧ�������ѷ�����_____��������_______��
| ���� | ��Ӧ�� |
| �� | Cu��Ũ���� |
| �� | Cu��ϡ���� |
| �� | Cu��O2��ϡ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ǹ�ҵ���Ʊ�Na2S2O3�ķ���֮һ����Ӧԭ��Ϊ��2Na2S+Na2CO3+4SO2=3Na2S2O3+CO2���÷�Ӧ��H��0����ij�о�С����ʵ��������Ʊ�Na2S2O3��5H2O�������¡�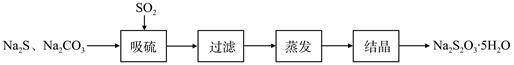
��1������װ����ͼ��ʾ��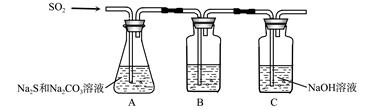
��װ��B�������Ǽ���װ��A��SO2������Ч�ʣ�B���Լ��� ������SO2����Ч�ʵ͵�ʵ��������B����Һ ��
��Ϊ��ʹSO2������������ȫ���ڲ��ı�A����ҺŨ�ȡ�����������£����˼�ʱ���跴Ӧ���⣬���ɲ�ȡ�ĺ�����ʩ�� �� ����д��������
��2�����豾ʵ�����õ�Na2CO3������NaCl��NaOH�����ʵ�鷽�����м��顣������ʱCaCO3������Һ��pH��10��2��
��ѡ�Լ���������ϡ���ᡢAgNO3��Һ��CaCl2��Һ��Ca(NO3)2��Һ����̪��Һ������ˮ��pH�ơ��ձ����Թܡ��ι�
| ��� | ʵ����� | Ԥ������ | ���� |
| �� | ȡ������Ʒ���Թ��У�������������ˮ��������ܽ⣬___________________�� | _______________ | ��Ʒ��NaCl |
| �� | ��ȡ������Ʒ���ձ��У�������������ˮ����ֽ����ܽ⣬___________________�� | _______________ | ��Ʒ��NaOH |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
����ʡ��2013��12��1����ʱ�𣬳�����������Ϊ�������������Զ���������ŷ����˴��ĸ��ơ���֪SO2������Fe( NO3)3��Һ���գ� 0��1mol/L��Fe(NO3)3��Һ��pH��2��ijѧϰС��ݴ�չ���������̽����
��̽��I��ͭ��Ũ����ķ�Ӧ̽����
(l)ȡ12��8gͭƬ��������ƿ�У�ͨN2һ��ʱ����ټ���20 mL 18 mol?L-1��Ũ���ᣬ���ȡ�װ��A���а������������������ɣ�װ��B�в�����ɫ��������ַ�Ӧ��,��ƿ������ͭƬʣ�ࡣ
�ٸ�С��ͬѧ��Ϊ��ƿ�г���ͭƬʣ���Ӧ�н϶������ʣ�࣬��ԭ���ǣ� ___________________ ��
�ڸ�С��ͬѧ��ͨ���ⶨ�����������������������ʵ���������˶���ʵ�鷽�������з��������е���______ ��
A�������������建��ͨ��Ԥ�ȳ�����ʢ�м�ʯ�ҵĸ����,������Ӧ���ٴγ���
B�������������建��ͨ�����������ữ�ĸ��������Һ���ټ���������BaCl2��Һ���������ó���������
C�����ű���NaHSO3��Һ�ķ����ⶨ�������������(����ɱ�״����
��̽��II��װ��B�в���������ԭ��̽����
��2������Ũ����֮ǰ��ͨN2һ��ʱ�䣬��Ŀ����____ ��
��3���������ۣ���С���װ��B�в���������ԭ��������в���(�����Ǹ����صĵ��ӣ���
����1: װ��A�еİ�������B���뷴Ӧ
����2��SO2��Fe3+����ΪSO42-
����3�� ��
��4����ͬѧ��ΪֻҪ��װ��A��B������ϴ��ƿC���Ϳ����ų�װ��A�а���Ӱ�죬��C��ʢ�ŵ��Լ��� ��
��5����ͬѧȡ������װ��B����Һ�����뼸�����Ը�����أ������Ϻ�ɫ��ȥ���ݴ���Ϊ����2���������Ƿ�ͬ������ۣ���˵�����ɣ� ��
��˼���뽻����
��6��ʵ���������ʹ��ƿ��ͭƬ�����ܽ⣬���з���(��Ҫʱ�ɼ��ȣ����е��� ��
A�����ɼУ�ͨ��O2 B���ɷ�Һ©������H2O2��Һ
C���ɷ�Һ©������NaNO3��Һ D���ɷ�Һ©������Na2SO4��Һ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
������ˮ�к��ж������ӣ�ijУ��ѧ�о���ѧϰС���ͬѧΪ̽�������ʣ���������ʵ�飬���������ɣ�
(1)HClO���ȶ��������ֽ�����HCl��O2�������ʵ��֤����O2���� ��
(2)������ʹʪ��ĺ�ɫ������ɫ������ʹʪ��ĺ�ɫ������ɫ�����ʣ�ͬѧ�ǵĿ�����һ�£���Ϊ��ˮ�д��ڵļ������Ӷ��п��ܡ��������ʵ�飬�ó���ȷ���ۡ�
| ������� | �ռ����� | ������� | ��֤���� | �ý��۳� |
| ��ˮ�к���������ʹʪ��ĺ�ɫ������ɫ�� | ��������ǿ������ ����������ˮ��Ӧ��������ʹ����� �۴�������ǿ������ | �� �� �� ��H2Oʹ������ɫ | �٣�ʵ��٣��Ѻ�ɫ�ɲ���������������ļ���ƿ�У���������ɫ ʵʵ��ڣ� �� ʵ��ۣ��Ѻ�ɫ��������ˮ���������ɫ | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
���ü�������������ȡ����Ӧ��ȡ����Ʒ����������ڹ�ҵ���ѳ�Ϊ��ʵ��ij��ѧ��ȤС����ʵ������ģ���������̣�����Ƶ�ģ��װ�����£�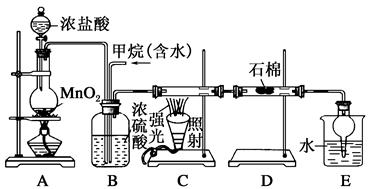
�������Ҫ��ش�
��1��Bװ�������ֹ��ܣ��ٿ��������ٶȣ��ھ��Ȼ�����壻��______________��
��2����V��Cl2��/V��CH4����x��������������������Ȼ��⣬��xֵӦ________��
��3��Dװ�õ�ʯ���о��Ȼ���KI��ĩ����������____________��
��4��Eװ�õ�������________������ţ���
| A���ռ����� | B���������� |
| C����ֹ���� | D�������Ȼ��� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com