
ijŠ©»Æѧ·“Ó¦æÉÓĆĻĀŹ½±ķŹ¾£ØĪ“ÅäĘ½£©£ŗA+B”śC+D+H2O
Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©ČōA”¢C”¢D¾łŗ¬ÓŠĀČŌŖĖŲ£¬ĒŅAÖŠĀČŌŖĖŲµÄ»ÆŗĻ¼Ū½éÓŚCÓėDÖ®¼ä£¬Š“³öøĆ·“Ó¦µÄĄė×Ó·½³ĢŹ½£ŗ___________________________________________”£
£Ø2£©ČōCĪŖĀČ»ÆÄĘ£¬DŹĒÄÜŹ¹³ĪĒåŹÆ»ŅĖ®±ä»ė×ĒµÄĪŽĪ¶ĘųĢ壬ŌņAÓėBµÄ×éŗĻŹĒ£ØĢīĖ×Ćū£©£ŗA________________»ņ________________ , B______________________”£
£Ø3£©ČōAĪŖ×ĻŗģÉ«½šŹō£¬DĪŖĪŽÉ«“Ģ¼¤ŠŌĘųĢ壬ĒėŠ“³ö·ūŗĻÉĻŹ½µÄ»Æѧ·½³ĢŹ½£ŗ____________________________________________”£
£Ø4£©ČōC”¢D¾łĪŖĘųĢåĒŅ·Ö×Ó¾ßÓŠĻąĶ¬µÄŌ×ÓøöŹż±Č£¬Ōņ·ūŗĻÉĻŹ½»Æѧ·½³ĢŹ½ŹĒ£ŗ____________________________________________”£
£Ø5£©ČōAĪŖ¹żŃõ»ÆÄĘ£¬BĪŖĮņĖį£¬ŌņCŹĒ____________£¬DŹĒ_______________”£
£Ø1£©Cl2+2OH£=Cl£+ ClO£+H2O£Ø2·Ö£©
£Ø2£©A£ŗ“æ¼ī»ņĖÕ“ņ Š”ĖÕ“ņ B£ŗŃĪĖį£ØĆææÕ1·Ö£¬¹²3·Ö£©
£Ø3£©Cu£«2H2SO4£ØÅØ£©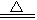 CuSO4£«2H2O£«SO2”ü£Ø2·Ö£©
CuSO4£«2H2O£«SO2”ü£Ø2·Ö£©
£Ø4£©CŹ®2H2SO4£ØÅØ£© CO2”ü£«2SO2”ü£«2H2O£Ø2·Ö£©
CO2”ü£«2SO2”ü£«2H2O£Ø2·Ö£©
£Ø5£©ĮņĖįÄĘŗĶŃõĘų»ņŃõĘųŗĶĮņĖįÄĘ£ØŠ“»ÆѧŹ½Ņ²øų·Ö£©£ØĆææÕ1·Ö£¬¹²2·Ö£©
½āĪöŹŌĢā·ÖĪö£ŗ£Ø1£©AÖŠĀČŌŖĖŲµÄ»ÆŗĻ¼Ū½éÓŚCÓėDÖ®¼ä£¬ŌņAÖŠClŌŖĖŲ»ÆŗĻ¼Ū²æ·ÖÉżøß”¢²æ·Ö½µµĶ£¬·ūŗĻµÄ·“Ó¦ĪŖCl2ÓėĒæ¼īČÜŅŗµÄ·“Ó¦£¬Ąė×Ó·½³ĢŹ½ĪŖ£ŗCl2+2OH£ ="=" Cl£+ ClO£+H2O”£
£Ø2£©AÓėB·“Ӧɜ³ÉNaClŗĶĪŽĪ¶µÄĘųĢåCO2”¢H2O£¬ŌņAæÉÄÜĪŖNa2CO3£¬Ė×ĆūĪŖ“æ¼ī»ņĖÕ“ņ£¬Ņ²æÉÄÜĪŖNaHCO3£¬Ė×ĆūĪŖŠ”ĖÕ“ņ£»BĪŖŃĪĖį”£
£Ø3£©AĪŖ×ĻŗģÉ«½šŹō£¬ŹĒĶµ„ÖŹ£¬DĪŖĪŽÉ«“Ģ¼¤ŠŌĘųĢ壬·ūŗĻµÄ·“Ó¦ĪŖCuÓėÅØĮņĖį·“Ӧɜ³ÉCuSO4”¢SO2ŗĶH2O£¬æɵƻÆѧ·½³ĢŹ½£ŗCu£«2H2SO4£ØÅØ£© CuSO4£«2H2O£«SO2”ü”£
CuSO4£«2H2O£«SO2”ü”£
£Ø4£©C”¢D¾łĪŖĘųĢåĒŅ·Ö×Ó¾ßÓŠĻąĶ¬µÄŌ×ÓøöŹż±Č£¬·ūŗĻµÄ·“Ó¦ĪŖCÓėÅØĮņĖį·“Ӧɜ³ÉCO2”¢SO2ŗĶH2O£¬»Æѧ·½³ĢŹ½ĪŖ£ŗCŹ®2H2SO4£ØÅØ£© CO2”ü£«2SO2”ü£«2H2O”£
CO2”ü£«2SO2”ü£«2H2O”£
£Ø5£©Na2O2ÓėH2SO4·“Ӧɜ³ÉNa2SO4”¢O2ŗĶH2O£¬ĖłŅŌC”¢DĪŖĮņĖįÄĘŗĶŃõĘų»ņŃõĘųŗĶĮņĖįÄĘ”£
æ¼µć£ŗ±¾Ģāæ¼²éĄė×Ó·½³ĢŹ½Óė»Æѧ·½³ĢŹ½µÄŹéŠ“”¢·“Ó¦ĪļŗĶÉś³ÉĪļµÄĶʶĻ”£



| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
ijŠ©»Æѧ·“Ó¦æÉÓĆĻĀŹ½±ķŹ¾ 
Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©ČōA”¢C”¢D¾łŗ¬ÓŠĀČŌŖĖŲ£¬ĒŅAµÄ»ÆŗĻ¼Ū½éÓŚCÓėDÖ®¼ä£¬Š“³öøĆ·“Ó¦µÄĄė×Ó·½³ĢŹ½£ŗ ”£
£Ø2£©ČōC”¢D¾łĪŖĘųĢåĒŅ¶¼ÄÜŹ¹³ĪĒåŹÆ»ŅĖ®±ä»ė×Ē£¬ŌņAÓėBµÄ×éŗĻŹĒ ”£
£Ø3£©ČōA”¢C¾łŗ¬ÓŠĀĮŌŖĖŲ”£¢Łµ±BŹĒŃĪĖįŹ±£¬CŹĒ £»¢Śµ±BŹĒNaOHŹ±£¬CŹĒ ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
ÅēČŖŹĒŅ»ÖÖ³£¼ūµÄŹµŃéĻÖĻó(ČēĻĀĶ¼)£¬Ęä²śÉśŌŅņŹĒ“ęŌŚŃ¹Ēæ²ī”£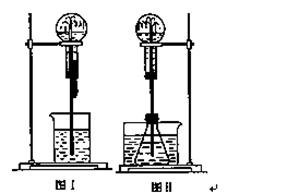
£Ø1£©Ķ¼¢ńĪŖ»Æѧ½Ģѧ֊ĖłÓƵÄÅēČŖŹµŃé×°ÖĆ”£ŌŚÉÕĘæÖŠ³äĀśøÉŌļĘųĢ壬½ŗĶ·µĪ¹Ü¼°ÉÕ±ÖŠ·Ö±šŹ¢ÓŠŅŗĢ唣ĻĀĮŠ×éŗĻÖŠ²»æÉÄÜŠĪ³ÉÅēČŖµÄŹĒ(Ģī±ąŗÅ×ÖÄø) ”£
A.HCl ŗĶH2O B.O2ŗĶH2O
C.SO2ŗĶNaOHČÜŅŗ D.CO2ŗĶNaOHČÜŅŗ
£Ø2£©Ä³Ń§Éś»ż¼«Ė¼æ¼²śÉśÅēČŖµÄĘäĖū°ģ·Ø£¬²¢Éč¼ĘĮĖĶ¼¢ņĖłŹ¾µÄ ×°ÖĆ”£
ŌŚĶ¼¢ņµÄ׶ŠĪĘæÖŠ£¬·Ö±š¼ÓČė×ćĮæµÄĻĀĮŠĪļÖŹ£¬·“Ó¦ŗóæÉÄܲśÉś ÅēČŖµÄŹĒ(Ģī±ąŗÅ×ÖÄø) ”£
A£®CaCO3ŗĶĻ”H2SO4 B£®NaOHÓėĻ”HCl
C£®ZnÓėĻ”HCl D£®NaClÓėĻ”HNO3
£Ø3£©±Č½ĻĶ¼¢ńŗĶĶ¼¢ņĮ½Ģ××°ÖĆ£¬“Ó²śÉśÅēČŖµÄŌĄķ·ÖĪö£¬¶žÕߵIJ»Ķ¬µćŌŚÓŚ ”£
£Ø4£©³ĒŹŠÖŠ³£¼ūµÄČĖŌģÅēČŖ¼°»šÉ½±¬·¢µÄŌĄķÓėÉĻŹö£ØĢī”°Ķ¼¢ń”±»ņ”°Ķ¼¢ņ”±£©×°ÖƵÄŌĄķĻąĖĘ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
¼×”¢ŅŅ”¢±ū”¢¶”ĖÄÖÖĪļÖŹÖŠ£¬¼×”¢ŅŅ”¢±ū¾łŗ¬ÓŠÄ³ÖÖĻąĶ¬µÄŌŖĖŲ£¬ĖüĆĒÖ®¼ä¾ßÓŠČēĻĀ×Ŗ»Æ¹ŲĻµ£Ø·“Ó¦Ģõ¼žŗĶ²æ·Ö²śĪļŅŃĀŌČ„£©£ŗ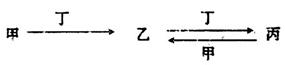
£Ø1£©Čō¼×ĪŖ³£¼ū½šŹōµ„ÖŹ£¬¼×ŗĶ¶”µÄĻ”ČÜŅŗ·“Ӧɜ³ÉĪŽÉ«ĘųĢ壬øĆĘųĢåÓöæÕĘų±äĪŖŗģ×ŲÉ«”£
¢Ł¼ģŃé±ūČÜŅŗÖŠ½šŹōŃōĄė×ӵďŌ¼ĮĪŖ________£ØĢīŹŌ¼ĮĆū³Ę£©”£
¢ŚĻņŗ¬n mol¶”µÄĻ”ČÜŅŗÖŠ¼ÓČėm molµ„ÖŹ¼×£¬¶žÕßĒ”ŗĆĶźČ«·“Ó¦£Ø¼ŁÉč»¹Ō²śĪļÖ»ÓŠÉĻŹöĪŽÉ«ĘųĢ壩£¬Ōņn£ŗmæÉÄÜĪŖ__________£ØĢī“śŗÅ£©”£
a£®3£ŗ2 b£®4£ŗ1 c£®5£ŗ1 d£®3£ŗ1
¢Ū³£ĪĀĻĀ£¬¶”µÄÅØČÜŅŗæÉ“ę·ÅÓŚ¼×ÖĘ×÷µÄČŻĘ÷ÖŠ£¬ĘäŌŅņŹĒ________________”£
£Ø2£©Čō¼×ĪŖĀČ¼ī¹¤ŅµµÄ²śĘ·£¬ŗ¬±ūµÄijÖÖŅ©Ę·æÉÓĆÓŚÖĪĮĘĪøĖį¹ż¶ą”£
¢Ł¼ų±šµČÅØ¶ČµÄŅŅČÜŅŗŗĶ±ūČÜŅŗ£¬æÉŃ”ÓƵďŌ¼ĮĪŖ________£ØĢī“śŗÅ£©
a£®NaOHČÜŅŗ b£®CaC12ČÜŅŗ c£®Ļ”ĮņĖį d£®³ĪĒåŹÆ»ŅĖ®
¢Ś½«¼×”¢ŅŅ”¢±ūČżÖÖČÜŅŗ»ģŗĻ£¬Ļņ»ģŗĻŅŗÖŠÖšµĪ¼ÓČėŅ»¶ØÅØ¶ČµÄŃĪĖį£¬Éś³ÉĘųĢåµÄĢå»ż£Ø±ź×¼×“æö£©Óė¼ÓČėŃĪĖįµÄĢå»ż¹ŲĻµČēĻĀĶ¼ĖłŹ¾£ŗ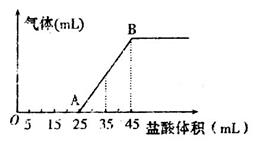
Š“³öOA¶ĪĖł·¢Éś·“Ó¦µÄĄė×Ó·½³ĢŹ½__________________________£¬µ±¼ÓČė35mLŃĪĖįŹ±£¬²śÉśĘųĢåµÄĢå»żĪŖ22£®4mL£Ø±ź×¼×“æö£©£¬ŌņŃĪĖįµÄĪļÖŹµÄĮæÅضČĪŖ_______”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
 Ӣ
Ӣ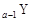 Ӣ
Ӣ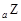 Ӣ
Ӣ Ӣ
”¢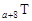 ŹĒĪåÖÖ³£¼ūµÄ¶ĢÖÜĘŚŌŖĖŲ£¬ĘäŌ×Ó°ė¾¶ĖęŌ×ÓŠņŹż±ä»ÆČēĻĀĶ¼ĖłŹ¾”£ZŹĒµŲæĒÖŠŗ¬Įæ×īøßµÄŌŖĖŲ”£WŌ×Ó×īĶā²ćµē×ÓŹżŹĒK²ćµē×ÓŹżµÄ2±¶”£
ŹĒĪåÖÖ³£¼ūµÄ¶ĢÖÜĘŚŌŖĖŲ£¬ĘäŌ×Ó°ė¾¶ĖęŌ×ÓŠņŹż±ä»ÆČēĻĀĶ¼ĖłŹ¾”£ZŹĒµŲæĒÖŠŗ¬Įæ×īøßµÄŌŖĖŲ”£WŌ×Ó×īĶā²ćµē×ÓŹżŹĒK²ćµē×ÓŹżµÄ2±¶”£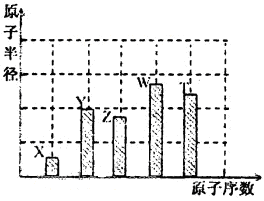
£Øl£©WŌŚÖÜĘŚ±ķÖŠµÄĪ»ÖĆĪŖ_______________________”£
£Ø2£©Y”¢ZµÄ10µē×ÓĒā»ÆĪļ·Ö×ÓÖŠ¾łŗ¬ÓŠµÄ»Æѧ¼üĄąŠĶĪŖ___________£»ĘäĪČ¶ØŠŌ½ĻĒæµÄŹĒ___________
£ØĢī»ÆѧŹ½£©”£
£Ø3£©ÓĆ»Æѧ·½³ĢŹ½±ķŹ¾YZ2ŠĪ³ÉĖįÓźµÄ·“Ó¦£ŗ______________________”£
ÓĆ¾«ĆÜpHŹŌÖ½£Ø¾«Č·µ½0.l£©²ā¶ØĖįÓźpHµÄ²Ł×÷ŹĒ______________________”£
£Ø4£©³£ĪĀĻĀÓĆ1 mol/L100 mL NaOHČÜŅŗĒ”ŗĆĶźČ«ĪüŹÕ0.1molTZ2ĘųĢ壬“Ė·“Ó¦µÄĄė×Ó·½³ĢŹ½ĪŖ
______________________£»“ĖŹ±ČÜŅŗpH<7µÄŌŅņŹĒ___________________________”£
£Ø5£©ŌŚ25”ę”¢101kPaĻĀ£¬¼ŗÖŖWÓėXŠĪ³ÉµÄĘųĢ¬»ÆŗĻĪļÓė×ćĮæZ2µćČ¼ĶźČ«·“Ó¦£ØÉś³ÉWµÄ×īøß¼Ū
»ÆŗĻĪļ£©£¬»Öø“ÖĮŌדĢ¬£¬Ę½¾łĆæ×ŖŅĘl mol e£ß·ÅČČ190 kJ£¬øĆ·“Ó¦µÄČČ»Æѧ·½³ĢŹ½ĪŖ___________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
¶ĢÖÜĘŚŌŖĖŲX”¢Y”¢ZŌ×ÓŠņŹżŅĄ“ĪµŻŌö”£X”¢Y”¢ZµÄ×īĶā²ćµē×ÓŹżÖ®ŗĶĪŖ13£¬YŹĒĖłŌŚÖÜĘŚÖŠ½šŹōŠŌ×īĒæµÄ£¬X”¢ZŌŚĶ¬Ņ»Ö÷×唣
£Ø1£©ZŌŖĖŲŌŚÖÜĘŚ±ķÖŠµÄĪ»ÖĆŹĒ ”£
£Ø2£©Y2ZX3ČÜŅŗĻŌ ŠŌ£¬ÄÜÖ¤Ć÷øĆČÜŅŗÖŠ“ęŌŚĖ®½āĘ½ŗāµÄŹĀŹµŹĒ £ØĢīŠņŗÅ£©”£
a£®µĪČė·ÓĢŖČÜŅŗ±äŗģ£¬ŌŁ¼ÓČėĻ”H2SO4ŗģÉ«ĶĖČ„
b£®µĪČė·ÓĢŖČÜŅŗ±äŗģ£¬ŌŁ¼ÓČėĀČĖ®ŗģÉ«ĶĖČ„
c£®µĪČė·ÓĢŖČÜŅŗ±äŗģ£¬ŌŁ¼ÓČėBaCl2ČÜŅŗ²śÉś³ĮµķĒŅŗģÉ«ĶĖČ„
£Ø3£©»ÆŗĻĪļCu2XŗĶCu2ZæÉ·¢ÉśČēĻĀ×Ŗ»Æ£ØĘäÖŠDŹĒĻĖĪ¬ĖŲĖ®½āµÄ×īÖÕ²śĪļ£©£ŗ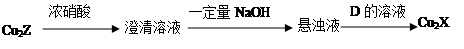
¢Ł DµÄ»ÆѧŹ½ŹĒ £»
¢Ś Cu2XÓėÅØĻõĖį·“Ó¦ÓŠŗģ×ŲÉ«ĘųĢåÉś³É£¬Ōņ»Æѧ·“Ó¦·½³ĢŹ½ŹĒ ”£
£Ø4£©Ä³ĪĀ¶ČĻĀ£¬ŌŚŅ»øö5LµÄĆܱÕČŻĘ÷ÖŠ³äČė0.2 mol ZX2ŗĶ0.1 mol X2£¬20 sŗó“ļµ½Ę½ŗā£¬²āµĆČŻĘ÷ÖŠŗ¬ÓŠ0.18 mol ZX3£¬ŌņÓĆX2±ķŹ¾øĆ·“Ó¦µÄĘ½¾łĖŁĀŹŹĒv(X2) = £¬“ĖĪĀ¶ČĻĀøĆ·“Ó¦µÄ»ÆŃ§Ę½ŗā³£ŹżK= ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŹµŃéĢā
ÓĆČēĶ¼ĖłŹ¾×°ÖĆ½ųŠŠŹµŃé(¼Š³Ö×°ÖĆŅŃĀŌČ„)”£Ēė»Ų“šĻĀĮŠĪŹĢā: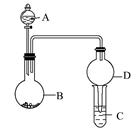
(1)ČōAÖŠĪŖÅØĻõĖį,BÖŠĪŖĶµ„ÖŹ,CÖŠĪŖĒāŃõ»ÆÄĘČÜŅŗ”£½«ÅØĻõĖįµĪČėBÖŠ,ŌņBÖŠ·¢Éś·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ”£
(2)ČōAÖŠĪŖÅØ°±Ė®,BÖŠĪŖÉśŹÆ»Ņ,CÖŠĪŖĆ÷·ÆČÜŅŗ”£
¢Ł½«ÅØ°±Ė®ÖšµĪµĪČėBÖŠ,æɲśÉś“óĮæ°±Ęų,ŌŅņæÉÄÜŹĒ(ĢīŠņŗÅ)”£
a.ÉśŹÆ»ŅŗĶÅØ°±Ė®·“Ó¦²śÉś°±Ęų,°±ĘųŅ×ČÜÓŚĖ®
b.ÉśŹÆ»ŅŗĶĖ®·“Ó¦,ĻūŗÄĖ®,Ź¹°±ĘųµÄČܽāĮæ¼õÉŁ
c.·“Ó¦·Å³ö“óĮæČČ,Ź¹°±ĘųŌŚĖ®ÖŠµÄČܽā¶ČĆ÷ĻŌ½µµĶ
¢ŚCÖŠĶØČė¹żĮæ°±ĘųŹ±£¬ĻąÓ¦µÄĄė×Ó·½³ĢŹ½ĪŖ”£
(3)ČōAÖŠĪŖĖ®,BÖŠĪŖ¹żŃõ»ÆÄĘ,CÖŠĪŖĖįŠŌµķ·Ūµā»Æ¼ŲČÜŅŗ,½«Ė®µĪČėBÖŠŗó,BÖŠµÄŹµŃéĻÖĻóĪŖ;ČōCÖŠČÜŅŗ±äĪŖĄ¶É«,ŌņCÖŠ·¢Éś·“Ó¦µÄĄė×Ó·½³ĢŹ½ĪŖ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗµ„Ń”Ģā
 ĻĀ±ķĪŖŌŖĖŲÖÜĘŚ±ķĒ°ĖÄÖÜĘŚµÄŅ»²æ·Ö£¬ĻĀĮŠÓŠ¹ŲR”¢W”¢X”¢Y”¢ZĪåÖÖŌŖĖŲµÄŠšŹöÖŠÕżČ·µÄŹĒ
ĻĀ±ķĪŖŌŖĖŲÖÜĘŚ±ķĒ°ĖÄÖÜĘŚµÄŅ»²æ·Ö£¬ĻĀĮŠÓŠ¹ŲR”¢W”¢X”¢Y”¢ZĪåÖÖŌŖĖŲµÄŠšŹöÖŠÕżČ·µÄŹĒ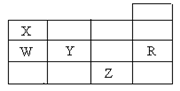
| A£®³£Ń¹ĻĀĪåÖÖŌŖĖŲµÄµ„ÖŹÖŠ£¬Zµ„ÖŹµÄ·Šµć×īøß |
| B£®Y”¢ZµÄŅõĄė×Óµē×Ó²ć½į¹¹¶¼ÓėRŌ×ÓµÄĻąĶ¬ |
| C£®WµÄĒā»ÆĪļ±ČXµÄĒā»ÆĪļĪČ¶Ø |
| D£®YŌŖĖŲ×īøß¼ŪŃõ»ÆĪļ¶ŌÓ¦Ė®»ÆĪļ±ČWŌŖĖŲµÄ×īøß¼ŪŃõ»ÆĪļ¶ŌÓ¦Ė®»ÆĪļµÄĖįŠŌĒæ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗµ„Ń”Ģā
A”¢B·Ö±šĪŖĶ¬Ņ»Ö÷×åµŚČżÖÜĘŚ”¢µŚĖÄÖÜĘŚµÄ²»Ķ¬ŌŖĖŲµÄŌ×Ó£¬ĖüĆĒŌ×ÓŗĖÄŚÖŹ×ÓŹż¾łµČÓŚÖŠ×ÓŹż”£ČōAĪŖ¢ņ£Į ×壬ĘäÖŹĮæŹżĪŖx£¬ŌņBµÄÖŹ×ÓŹżĪŖZ£»ČōAĪŖ¢ō£Į×壬ĘäÖŹ×ÓŹżĪŖy£¬ŌņBµÄÖŹĮæŹżĪŖN”£ZŗĶNĪŖĻĀĮŠÄÄŅ»×éÖµ
A£®Z£ŗ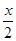 +18£¬N£ŗ2y+18 B£®Z£ŗ
+18£¬N£ŗ2y+18 B£®Z£ŗ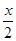 +8, N:2y+18
+8, N:2y+18
C£®Z£ŗ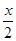 +8, N£ŗ2y+36 D£®Z£ŗ
+8, N£ŗ2y+36 D£®Z£ŗ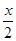 +18, N:2y+36
+18, N:2y+36
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com