
| A��2HCl(g)=H2(g)��Cl2(g)�ķ�Ӧ�Ȧ�H<0 |
| B��H(g)��Cl(g)=HCl(g)����H����431 kJ��mol��1 |
| C����ͬ�����£�H2(g)��Cl2(g)=2HCl(g)�ڹ��պ͵�ȼ�����µĦ�H��� |
| D��H2(g)��Cl2(g)=2HCl(g)����H����183 kJ |

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��H2O������ֻ���зǼ��Թ��ۼ� |
| B��H2O������ֻ���м��Թ��ۼ� |
| C��ˮ���Ӽ�ֻ������� |
| D����ȴ��Ӧ����ˮ�ڵ����ϳ������ǹ�̬ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

 O2(g)=CO(g)�Ħ�H���������ʵ�顢���ø�˹���ɼ�����÷�Ӧ�Ħ�H������ʱ��Ҫ��õ�ʵ��������________��
O2(g)=CO(g)�Ħ�H���������ʵ�顢���ø�˹���ɼ�����÷�Ӧ�Ħ�H������ʱ��Ҫ��õ�ʵ��������________���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
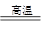 CO(g)��H2(g)��C(s)��CO(g)��H2(g)��ȫȼ�յ��Ȼ�ѧ����ʽΪ��
CO(g)��H2(g)��C(s)��CO(g)��H2(g)��ȫȼ�յ��Ȼ�ѧ����ʽΪ��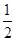 O2(g)=H2O(g)����H����242.0 kJ��mol��1
O2(g)=H2O(g)����H����242.0 kJ��mol��1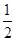 O2(g)=CO2(g)����H����283.0 kJ��mol��1
O2(g)=CO2(g)����H����283.0 kJ��mol��1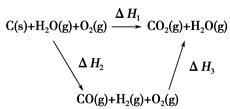
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
A����100 �桢101 kPa�����£�1molҺ̬ˮ����ʱ��Ҫ����40��69 kJ����������H2O(g) H2O(l) �Ħ�H = +40��69 kJ��mol-1 H2O(l) �Ħ�H = +40��69 kJ��mol-1 |
| B����֪CH4 (g)+2O2(g)=CO2(g)+2H2O(l)����H=-802��33kJ/mol,��CH4��ȼ����Ϊ802��33 kJ |
C��H2(g)��Br2(g)=2HBr(g)��H=��72kJ��mol��1��������������±��� �����aΪ230 |
| D����֪S (g)+O2(g)=SO2(s)����H1��S (g)+O2(g)=SO2(g)����H2�����H2����H1 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������



�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A�������� | B����������ľ̿���� |
| C���ƾ�����ˮ | D���Ȼ�����������ˮ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
 2SO3��g�� ��H =" a" kJ·mol��1����Ӧ���̵������仯��ͼ��ʾ����֪1mol SO2��g����ȫת��Ϊ1mol SO3��g������99 kJ����ش�
2SO3��g�� ��H =" a" kJ·mol��1����Ӧ���̵������仯��ͼ��ʾ����֪1mol SO2��g����ȫת��Ϊ1mol SO3��g������99 kJ����ش��鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com