
[2012����������һģ]��12�֣��������ƣ�Na2FeO4�����к�ǿ�������ԣ���һ�����͵���ɫ��ˮ����������ҵ�Ͽ���ͨ�����������������Ʊ��������ƣ������������£�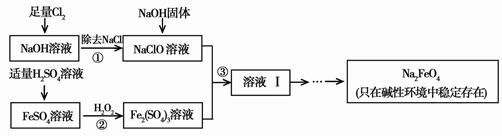
�ش��������⣺
��1����������ٺ���NaOH�����ԭ����____ ____��
��2������ڷ�Ӧ�����ӷ���ʽ��_____ ___��
��3������Һ���з����Na2FeO4���и���ƷNa2SO4��NaCl��������з�Ӧ�����ӷ���ʽΪ___ _____��
��4����һ������Na2FeO4Ͷ�뵽pH��ͬ����ˮ�У���ˮ������ɷ־���ͬ������Һ��Na2FeO4Ũ�ȱ仯����ͼ���ߢ���ʾ�����Ʋ�����I������II��Ӧ����ˮpH_____����ߡ��͡�����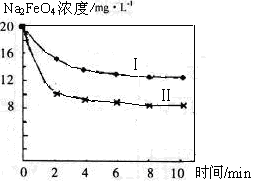
��5��ͨ�������֪Na2FeO4������Ч�ʣ��Ե�λ�����õ��ĵ�������ʾ����������_____����ߡ��͡������ø������ƴ�����������ˮ���������ŵ���____ _
��������㼴�ɣ���
 �����Ļ���������人������ϵ�д�
�����Ļ���������人������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ���¿α���������¿���һ����ѧ�Ծ���B�����������棩 ���ͣ�ʵ����
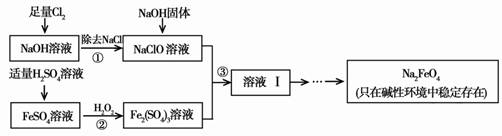
[2012����������һģ]��12�֣��������ƣ�Na2FeO4�����к�ǿ�������ԣ���һ�����͵���ɫ��ˮ����������ҵ�Ͽ���ͨ�����������������Ʊ��������ƣ������������£�
�ش��������⣺
��1����������ٺ���NaOH�����ԭ����____ ____��
��2������ڷ�Ӧ�����ӷ���ʽ��_____ ___��
��3������Һ���з����Na2FeO4���и���ƷNa2SO4 ��NaCl��������з�Ӧ�����ӷ���ʽΪ___ _____��
��4����һ������Na2FeO4Ͷ�뵽pH��ͬ����ˮ�У���ˮ������ɷ־���ͬ������Һ��Na2FeO4Ũ�ȱ仯����ͼ���ߢ���ʾ�����Ʋ�����I������II��Ӧ����ˮpH_____����ߡ��͡�����
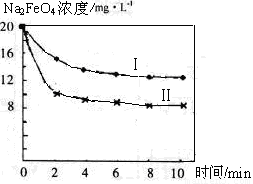
��5��ͨ�������֪Na2FeO4������Ч�ʣ��Ե�λ�����õ��ĵ�������ʾ����������_____����ߡ��͡������ø������ƴ�����������ˮ���������ŵ���____ _
��������㼴�ɣ���
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com