
����P(s)��Cl2(g)������Ӧ������PCl3(g)��PCl5(g)����Ӧ���̺������Ĺ�ϵ��ͼ��ʾ��ͼ�С�H��ʾ����1 mol ��������ݣ���
����ͼ�ش��������⣺
 ��1��P(s)��Cl2(g)��Ӧ����PCl3(g)���Ȼ�ѧ����ʽ��
��1��P(s)��Cl2(g)��Ӧ����PCl3(g)���Ȼ�ѧ����ʽ��
��2��PCl5(g)�ֽ�����PCl3(g)��Cl2(g)���Ȼ�ѧ����ʽ��
____________________��
��3�������ֽⷴӦ��һ�����淴Ӧ���¶�T1ʱ�������Ϊ1L���ܱ������м���0.80mol PCl5����Ӧ��ƽ��ʱPCl5��ʣ0.60mol����ֽ��ʦ�1���� ��ƽ�ⳣ��K1���� ������Ӧ�¶���T1���ߵ�T2��ƽ��ʱPCl5�ķֽ���Ϊ��2��ƽ�ⳣ��ΪK2�����2 ��1��
K2 K1��������ڡ�����С�ڡ����ڡ���
��4��P��Cl2��������Ӧ����1mol PCl5�ġ�H3 =
 ������ҵ��ͬ����ϰ��ϵ�д�
������ҵ��ͬ����ϰ��ϵ�д� С��ſ�ʱ��ҵϵ�д�
С��ſ�ʱ��ҵϵ�д� һ������ϵ�д�
һ������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��a%��ij���ʵ�ˮ��Һ����������m gˮ�����ʲ��ӷ���������������������������������Һ���ΪV L�����ʵ���������Ϊ����ǰ��2���������ʵ���Է�������ΪM����������������Һ�����ʵ���Ũ��Ϊ�� ��mol/L
A ma/2VM B ma/100VM C 20ma/2VM D ma/50VM
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�ڡ�ʯ����Һ��ʯ����ʯ���������ѻ������ı仯�����У����ƻ���������������
A.���»��������»��������ۼ� B.���»��������»��������»���
C.���»��������ۼ������ۼ� D.���ۼ������ۼ������ۼ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ͨ������£���A��BΪ���ӣ�C��EΪ�����ӣ�DΪ�����ӣ����Ƕ�����10�����ӣ�B����A�����õ����ʿɵ����C��D��A��B��E��������Ӧ��ɵ�C��һ�ְ�ɫ��������ش�
(1)�û�ѧ���ű�ʾ����4������
A��________��B��________��C��________��D________��
(2)д��A��B��E��������Ӧ�����ӷ���ʽ�� ___________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
���Խ���ӦZn+Br2 = ZnBr2��Ƴ����أ�����4���缫��Ӧ
��Br2 + 2e�� = 2Br- ����2Br- - 2e�� = Br2 ����Zn �C 2e�� = Zn2+�� ��Zn2+ + 2e�� = Zn
�� ���б�ʾ���ʱ��������Ӧ�ͷŵ�ʱ�ĸ�����Ӧ�ķֱ���
�� A���ںۡ͢��� �� B���ں͢� ����C���ۺ͢١������� D���ܺ͢�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����˵����ȷ����( )
A.H2SO4��H3PO4��Ħ��������� B.�ڱ�״����18gˮ�����ԼΪ22.4L
C.1molH2��ռ�����ԼΪ22.4L D.ͨ��״���£�NA�� CO2����ռ�е����Ϊ22.4L
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ʵ������Ҫ����2mol/L��NaCl��Һ480 ml������ʱӦѡ�õ�����ƿ�Ĺ���Ӧ��ȡ��NaCl�ֱ���
A.480 mL��117g B.500mL��58.5g C.480 mL��58.5 g D.500mL��117g
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��ѧ֪ʶ������������Ҫ���壬����˵������ȷ���� (����)
A����ľ�ң�����K2CO3��ˮ��Һ��������ϴ������
B���Ҵ�������ͼ�ȩ�㷺Ӧ����ʳƷ�ӹ�
C�������ŷŵ�CO2�ܽ��͵��������ЧӦ
D���ں������������п�飬�ɼ�������ĸ�ʴ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��ͼ��ʵ�����Ʊ�����������һϵ�����ʵ���װ�ã��г��豸���ԣ���
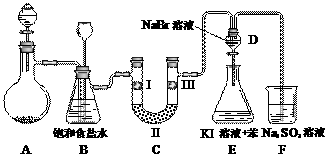
��1���Ʊ�����ѡ�õ�ҩƷΪ��Ư�۾������Ũ���ᣬ��صĻ�ѧ��Ӧ����ʽΪ��
��
��2��װ��B�б���ʳ��ˮ�������� ��ͬʱװ��B���ǰ�ȫƿ�����ʵ�����ʱC���Ƿ�����������д����������ʱB�е����� ��
��
��3��װ��C��ʵ��Ŀ������֤�����Ƿ����Ư���ԣ�Ϊ��C��I��II��III���η��� ��
| a | b | c | d | |
| I | �������ɫ���� | �������ɫ���� | ʪ�����ɫ���� | ʪ�����ɫ���� |
| II | ��ʯ�� | �轺 | Ũ���� | ��ˮ�Ȼ��� |
| III | ʪ�����ɫ���� | ʪ�����ɫ���� | �������ɫ���� | �������ɫ���� |
��4�����װ��D��E��Ŀ���DZȽ��ȡ��塢��ķǽ����ԡ�����D�л���ͨ����������ʱ�����Կ�����ɫ��Һ��Ϊ���غ�ɫ��˵���ȵķǽ����Դ����塣��������װ��D��������Һ����װ��E�У����۲쵽�������� �������� ����ܡ����ܡ���˵����ķǽ�����ǿ�ڵ⣬ԭ���� ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com