
 ŹĒŅ»ÖÖŅ½Ņ©ÖŠ¼äĢå,³£ÓĆĄ“Öʱøæ¹ÄżŃŖŅ©,æÉĶعżĻĀĮŠĀ·ĻßŗĻ³É:
ŹĒŅ»ÖÖŅ½Ņ©ÖŠ¼äĢå,³£ÓĆĄ“Öʱøæ¹ÄżŃŖŅ©,æÉĶعżĻĀĮŠĀ·ĻßŗĻ³É:
(1)Š“³öGµÄŗ¬Ńõ¹ŁÄÜĶŵÄĆū³Ę””””””””””””””””””;B”śCµÄ·“Ó¦ĄąŠĶŹĒ”””””””””””””£
(2)Š“³öD EµÄ»Æѧ·½³ĢŹ½: ”””£
EµÄ»Æѧ·½³ĢŹ½: ”””£
(3)Āś×ćĻĀĮŠĢõ¼žµÄDµÄĶ¬·ÖŅģ¹¹ĢåÓŠ””””””””ÖÖ”£
¢ŁÄÜ·¢ÉśŅų¾µ·“Ó¦;¢ŚÄÜŹ¹FeCl3ČÜŅŗĻŌÉ«;¢Ūõ„Ąą”£
(4)ĻĀĮŠ¹ŲÓŚGµÄĖµ·ØÕżČ·µÄŹĒ””””””
a.ÄÜŹ¹äåĖ®ĶŹÉ«
b.ÄÜÓė½šŹōÄĘ·“Ó¦
c.1 mol G×ī¶ąÄÜŗĶ3 molĒāĘų·“Ó¦
d.·Ö×ÓŹ½ŹĒC9H8O3
 æŚĖćĢāæؼÓÓ¦ÓĆĢā¼ÆѵĻµĮŠ“š°ø
æŚĖćĢāæؼÓÓ¦ÓĆĢā¼ÆѵĻµĮŠ“š°ø ×ŪŗĻ×Ō²āĻµĮŠ“š°ø
×ŪŗĻ×Ō²āĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
ÓŠ»śĪļAæÉ×÷ĪŖÖ²ĪļÉś³¤µ÷½Ś¼Į£¬ĪŖ±ćÓŚŹ¹ÓĆ£¬Ķس£½«ĘäÖĘ³É»ÆŗĻĪļD( )£¬DŌŚČõĖįŠŌĢõ¼žĻĀ»į»ŗĀżŹĶ·Å³öA”£ŗĻ³ÉDµÄŅ»ÖÖ·½·Ø¼°ø÷ĪļÖŹ¼äµÄ×Ŗ»Æ¹ŲĻµČēĻĀĶ¼ĖłŹ¾£ŗ
)£¬DŌŚČõĖįŠŌĢõ¼žĻĀ»į»ŗĀżŹĶ·Å³öA”£ŗĻ³ÉDµÄŅ»ÖÖ·½·Ø¼°ø÷ĪļÖŹ¼äµÄ×Ŗ»Æ¹ŲĻµČēĻĀĶ¼ĖłŹ¾£ŗ
Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
(1)AµÄĆū³ĘŹĒ £¬AÓėĀČĘų·“Ó¦æÉÉś³ÉC£¬CµÄĆū³ĘŹĒ £»
(2)¾²ā¶ØEÖŠŗ¬ÓŠĀČŌŖĖŲ£¬ĒŅEæÉŅŌÓėŅŅĖį·¢Éśõ„»Æ·“Ó¦£¬ŌņEµÄ½į¹¹¼ņŹ½ĪŖ £¬ÓÉAÖ±½ÓÉś³ÉEµÄ·“Ó¦ĄąŠĶŹĒ £»
(3)ŌŚČõĖįŠŌĢõ¼žĻĀ£¬DÓėĖ®·“Ӧɜ³ÉAµÄ»Æѧ·½³ĢŹ½ĪŖ £»
(4)Š“³öEµÄĮ½ÖÖĶ¬·ÖŅģ¹¹ĢåµÄ½į¹¹¼ņŹ½£ŗ ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
ijŗ¬ŃõÓŠ»śĪļX£¬ĘäĻą¶Ō·Ö×ÓÖŹĮæĪŖ46£¬ĘäÖŠĢ¼µÄÖŹĮæ·ÖŹżĪŖ52.2%£¬ĒāµÄÖŹĮæ·ÖŹżĪŖ13.0%£¬ĘäÓąĪŖŃõ”£
£Ø1£©XµÄ·Ö×ÓŹ½ŹĒ________”£
£Ø2£©XÄÜÓė½šŹōÄĘ·“Ó¦·Å³öĒāĘų£¬ŌņX½į¹¹ÖŠŗ¬ÓŠµÄ¹ŁÄÜĶŵÄĆū³ĘĪŖ________”£
£Ø3£©XÓėæÕĘųÖŠµÄŃõĘųŌŚ“߻ƼĮ×÷ÓĆĻĀ·“Ӧɜ³ÉĮĖY£¬øĆ·“Ó¦µÄ»Æѧ·½³ĢŹ½ŹĒ______________________£¬·“Ó¦ĄąŠĶĪŖ________”£
£Ø4£©XÓėøßĆĢĖį¼ŲĖįŠŌČÜŅŗ·“Ó¦æÉÉś³ÉZ£¬ŌŚ¼ÓČČŗĶÅØĮņĖį×÷ÓĆĻĀ£¬XÓėZ·“Ó¦æÉÉś³ÉŅ»ÖÖÓŠĻćĪ¶µÄĪļÖŹW£¬Čō184 g XŗĶ120 g Z·“Ӧɜ³É132 g W£¬øĆ·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ____________________________£¬ĘäÖŠXµÄ×Ŗ»ÆĀŹĪŖ________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
ŅŅ“¼ŹĒŅ»ÖÖÖŲŅŖµÄ»Æ¹¤ŌĮĻ”£
¢ń£®ĘĻĢŃĢĒŌŚ¾Ę»ÆĆøµÄ×÷ÓĆĻĀ×Ŗ»ÆĪŖŅŅ“¼µÄ»Æѧ·½³ĢŹ½ŹĒ£ŗ__________________________________”£
¢ņ£®Ļć¶¹ĖŲŹĒŅ»ÖÖÓĆĶ¾¹ć·ŗµÄĻćĮĻ£¬æÉŅŌĄūÓĆŅŅ“¼ŗĶB£Ø·Ö×ÓŹ½ĪŖC7H6O2£©ĶعżŅŌĻĀĶ¾¾¶ŗĻ³É”£
£Ø1£©CÖŠŗ¬Ńõ¹ŁÄÜĶŵÄĆū³ĘĪŖ_______£»DµÄ½į¹¹¼ņŹ½ŹĒ_________£»D”śEÖŠ¢ŁµÄ·“Ó¦ĄąŠĶĪŖ______________”£
£Ø2£©¢ŁŅŅ“¼Éś³ÉAµÄ»Æѧ·½³ĢŹ½ĪŖ£ŗ_________ ¢ŚÓÉEÉś³ÉĻć¶¹ĖŲµÄ»Æѧ·½³ĢŹ½ĪŖ£ŗ______________”£
£Ø3£©BÓŠ¶ąÖÖĶ¬·ÖŅģ¹¹Ģ壬ĘäÖŠ±½»·ÉĻÖ»ÓŠŅ»øö²ąĮ“µÄĶ¬·ÖŅģ¹¹ĢåµÄ½į¹¹¼ņŹ½ŹĒ_______________”£
£Ø4£©ÓŠ¹ŲĻć¶¹ĖŲµÄĖµ·ØÕżČ·µÄŹĒ_____________________Ģī×ÖÄø£©”£
| A£®Ö»ÄÜÓėäåµ„ÖŹ·¢Éś¼Ó³É·“Ó¦£¬²»ÄÜ·¢ÉśČ”“ś·“Ó¦ |
| B£®1 molĻć¶¹ĖŲæÉÓė5 mol H2·¢Éś¼Ó³É·“Ó¦ |
| C£®³¤ĘŚ·ÅÖĆĻć¶¹ĖŲ£¬ČŻŅ×Ńõ»Æ±äÖŹ |
| D£®1 molĻć¶¹ĖŲĶźČ«Č¼ÉÕĻūŗÄ9.5 mol O2 |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
ŅŃÖŖ2RCH2CHO 

Ė®ŃīĖįõ„EĪŖ×ĻĶāĻßĪüŹÕ¼Į£¬æÉÓĆÓŚÅäÖĘ·ĄÉ¹ĖŖ”£EµÄŅ»ÖÖŗĻ³ÉĀ·ĻßČēĻĀ£ŗ
Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
(1)Ņ»ŌŖ“¼AÖŠŃõµÄÖŹĮæ·ÖŹżŌ¼ĪŖ21.6%£¬ŌņAµÄ·Ö×ÓŹ½ĪŖ________£»½į¹¹·ÖĪöĻŌŹ¾AÖ»ÓŠŅ»øö¼×»ł£¬AµÄĆū³ĘĪŖ________”£
(2)BÄÜÓėŠĀÖʵÄCu(OH)2·¢Éś·“Ó¦£¬øĆ·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ____________________________”£
(3)CÓŠ________ÖÖ½į¹¹£»ČōŅ»“ĪȔѳ£¬¼ģŃéCÖŠĖłŗ¬¹ŁÄÜĶÅ£¬°“Ź¹ÓƵÄĻČŗóĖ³ŠņŠ“³öĖłÓĆŹŌ¼Į£ŗ______________________________________________”£
(4)µŚ¢Ū²½µÄ·“Ó¦ĄąŠĶĪŖ________£»DĖłŗ¬¹ŁÄÜĶŵÄĆū³ĘĪŖ________”£
(5)Š“³öĶ¬Ź±·ūŗĻĻĀĮŠĢõ¼žµÄĖ®ŃīĖįĖłÓŠĶ¬·ÖŅģ¹¹ĢåµÄ½į¹¹¼ņŹ½£ŗ_____________________________”£
a£®·Ö×ÓÖŠÓŠ6øöĢ¼Ō×ÓŌŚŅ»ĢõÖ±ĻßÉĻ£»
b£®·Ö×ÓÖŠĖłŗ¬¹ŁÄÜĶÅ°üĄØĖ®ŃīĖį¾ßÓŠµÄ¹ŁÄÜĶÅ”£
(6)µŚ¢Ü²½µÄ·“Ó¦Ģõ¼žĪŖ______________£»Š“³öEµÄ½į¹¹¼ņŹ½£ŗ________________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
ŅŃÖŖ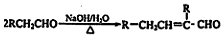 Ė®ŃīĖįõ„EĪŖ×ĻĶāĻßĪüŹÕ¼Į£¬æÉÓĆÓŚÅäÖĘ·ĄÉ¹ĖŖ”£EµÄŅ»ÖÖŗĻ³ÉĀ·ĻßČēĻĀ£ŗ
Ė®ŃīĖįõ„EĪŖ×ĻĶāĻßĪüŹÕ¼Į£¬æÉÓĆÓŚÅäÖĘ·ĄÉ¹ĖŖ”£EµÄŅ»ÖÖŗĻ³ÉĀ·ĻßČēĻĀ£ŗ
Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©Ņ»ŌŖ“¼AÖŠŃõµÄÖŹĮæ·ÖŹżŌ¼ĪŖ21.6%”£ŌņAµÄ·Ö×ÓŹ½ĪŖ ”£½į¹¹·ÖĪöĻŌŹ¾AÖ»ÓŠŅ»øö¼×»ł£®AµÄĆū³ĘĪŖ ”£
£Ø2£©BÄÜ·¢ÉśŅų¾µ·“Ó¦£¬øĆ·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ ”£
£Ø3£©CÓŠ ÖÖ½į¹¹£»ČōŅ»“ĪȔѳ£¬¼ģŃéCÖŠĖłŗ¬¹ŁÄÜĶÅ£¬°“Ź¹ÓƵÄĻČŗóĖ³ŠņŠ“³öĖłÓĆŹŌ¼Į ”£
£Ø4£©µŚ¢Ū²½µÄ·“Ó¦ĄąŠĶĪŖ £»DĖłŗ¬¹ŁÄÜĶŵÄĆū³ĘĪŖ ”£
£Ø5£©Š“³öĶ¬Ź±·ūŗĻĻĀĮŠĢõ¼žµÄĖ®ŃīĖįµÄĶ¬·ÖŅģ¹¹ĢåµÄ½į¹¹¼ņŹ½£ŗ£ØČĪŠ“Ņ»ÖÖ£© ”£
a£®·Ö×ÓÖŠŗ¬ÓŠ6øöĢ¼Ō×ÓŌŚŅ»ĢõĻßÉĻ£»
b£®·Ö×ÓÖŠĖłŗ¬¹ŁÄÜĶÅ°üĄØĖ®ŃīĖį¾ßÓŠµÄ¹ŁÄÜĶÅ
£Ø6£©Š“³öEµÄ½į¹¹¼ņŹ½ ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
ĶŃĖ®»·»ÆŹĒŗĻ³ÉÉśĪļ¼īĄąĢģČ»²śĪļµÄÖŲŅŖ²½Öč£¬Ä³ÉśĪļ¼īŗĻ³ÉĀ·ĻßČēĻĀ£ŗ
£Ø1£©»ÆŗĻĪļ¢óµÄ»ÆѧŹ½ĪŖ ”£
£Ø2£©»ÆŗĻĪļAµÄ½į¹¹¼ņŹ½ĪŖ ”£
£Ø3£©·“Ó¦¢ŁµÄ»Æѧ·½³ĢŹ½ĪŖ ”£
£Ø4£©ĻĀĮŠĖµ·Ø“ķĪóµÄŹĒ ”£
| A£®»ÆŗĻĪļ¢ņÄÜ·¢ÉśŅų¾µ·“Ó¦ | B£®»ÆŗĻĪļ¢ń”«¢õ¾łŹōÓŚ·¼ĻćĢž |
| C£®·“Ó¦¢ŪŹōÓŚõ„»Æ·“Ó¦ | D£®»ÆŗĻĪļ¢ņÄÜÓė4 molH2·¢Éś¼Ó³É·“Ó¦ |

²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
ŅŃÖŖ:
¢ń.AŹĒŹÆÓĶĮŃ½āĘųµÄÖ÷ŅŖ³É·ÖÖ®Ņ»”£
¢ņ.HĪŖ±½µÄĶ¬ĻµĪļ;ŌŚĻąĶ¬µÄĢõ¼žĻĀ,ĘäĘųĢå¶ŌH2µÄĻą¶ŌĆܶČĪŖ46;ĘäŗĖ“Ź²ÕńĒāĘ×ÓŠ4øö·å,·åĆ껿֮±ČĪŖ1”Ć2”Ć2”Ć3”£
¢ó. +CH3Br
+CH3Br
 +HBr
+HBr
¢ō.CĪļÖŹ±½»·ÉĻµÄŅ»Ā±“śĪļÖ»ÓŠĮ½ÖÖ”£
ÓŠ»śĪļA“ęŌŚĻĀĮŠ×Ŗ»Æ¹ŲĻµ:
»Ų“šĻĀĮŠĪŹĢā:
(1)BµÄĆū³ĘŹĒ”” ”£
(2)DµÄ½į¹¹¼ņŹ½ŹĒ”” ”£
(3)·“Ó¦¢ŽµÄ·“Ó¦ĄąŠĶŹĒ”” ”£
(4)Š“³ö·“Ó¦¢ŻµÄ»Æѧ·½³ĢŹ½”” ”£
(5)·Ö×ÓÖŠ¾ßÓŠ±½»·,±½»·ÉĻÓŠĮ½øöČ”“ś»ł,ĒŅÄÜÓėNaHCO3ČÜŅŗ·“Ó¦µÄEµÄĶ¬·ÖŅģ¹¹ĢåÓŠ””””””””ÖÖ(²»°üŗ¬E),Š“³öĘäÖŠŅ»ÖÖĶ¬·ÖŅģ¹¹ĢåµÄ½į¹¹¼ņŹ½”””””””””£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
ŅŃÖŖijĶéĢž·Ö×ÓÖŠµē×ÓŹżĪŖ42”£
(1)øĆĶéĢžµÄ·Ö×ÓŹ½ĪŖ_______________________”£
(2)Š“³öøĆĶéĢž·Ö×ÓµÄĖłÓŠĶ¬·ÖŅģ¹¹ĢåµÄ½į¹¹¼ņŹ½_____________”£
(3)ÉĻŹöĶ¬·ÖŅģ¹¹ĢåÖŠ£¬ŌŚĻąĶ¬Ģõ¼žĻĀ·Šµć×īµĶµÄŹĒ_____________”£(Š“½į¹¹¼ņŹ½)
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com