
ʵ����� | �������A�� | ��������ˮ | �����Լ���B�� | ���ˡ�ϴ�ӡ�����ù��壨C�� | ������Һ�õ����壨D�� | ����������� |
�� | BaCl2��NaCl | �ܽ� | ����CO2 | �� | D | A��D |
�� | CaCl2��NaCl | �ܽ� | ��������Na2CO3��Һ | CaCO3 | NaCl | A��C |
�� | Na2SO4��MgSO4 | �ܽ� |
|
|
|
|
�Իش��������⣺
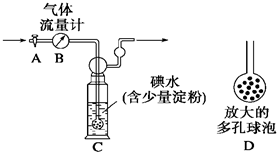
��1��ʵ�����D�ijɷ���____________��������____________����ܡ����ܡ���ͨ������õ�A�и���ֵ������ȡ�
��2��ʵ�����д�����������Դ������____________����ԭ����_____________________��
��3����ʵ��������ƣ�������Ӧ����������ж�Ӧ�Ŀո��ڣ������ܵó�A�и���ֵ�������Ϊ�����ú���ĸ�ı���ʽ��ʾ��_____________________________________________��
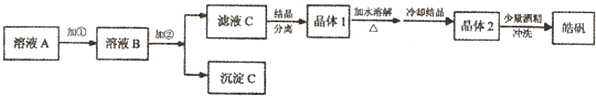
��1��BaCl2��NaCl ����
��2��D D�������NaCl������Na2CO3
��3������NaOH��Һ Mg(OH)2 Na2SO4��NaOH A��C��A![]() ����
����![]() �������������𰸣�
�������������𰸣�
��������3���ʣ�Na2SO4��MgSO4�����������ײ�����������壬���������׳��������ij�����ѡ�ù���NaOH��Һ������
Mg(OH)2һ�ֳ��������������Ӧ�����������Ϊ��������A��Mg(OH)2����C��Na2SO4��MgSO4�����ȵı���ʽΪ��A![]() ����
����![]() ��
��


 ����Ӣ��ϵ�д�
����Ӣ��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
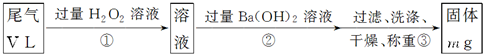
| 22.4m |
| 233V |
| 22.4m |
| 233V |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
 ��I���ζ��������ǻ�ѧ�������е���Ҫ��������֮һ��
��I���ζ��������ǻ�ѧ�������е���Ҫ��������֮һ��| �ζ����� ʵ�����ݣ�mL�� |
1 | 2 | 3 | 4 |
| V����Ʒ�� | 20.00 | 20.00 | 20.00 | 20.00 |
| V��NaOH�������ģ� | 15.95 | 15.00 | 15.05 | 14.95 |

| ʵ�鲽�� | ʵ������ | ʵ����� | |
| ʵ��һ | �������ǵ�ľ������װ�й���������Һ���Թ��� | ľ������ȼ | �����¹���������Һ���ֽ⣨��ֽ���٣� �� �� |
| ʵ��� | ��װ��H2O2��Һ���Թ��м�������Al2O3��Ȼ�����ǵ�ľ�������Թ��� | ľ����ȼ | Al2O3�ܼӿ�H2O2��Һ�ķֽ����� Al2O3�ܼӿ�H2O2��Һ�ķֽ����� |

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| �¶ȣ��棩 | 60 | 240 | 930 | 1000 |
| ��������������g�� | 19.7 | 16.1 | 8.1 | 8.1 |
| ||
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
 ��2013?����һģ��ijУ��ѧ��ȤС�����������ʵ��װ�ã�ͼ�в��ּг���������ȥ������ ��ij��̼�Ͻ�������������������̽������Ũ����ķ�Ӧ����ش��������⣺
��2013?����һģ��ijУ��ѧ��ȤС�����������ʵ��װ�ã�ͼ�в��ּг���������ȥ������ ��ij��̼�Ͻ�������������������̽������Ũ����ķ�Ӧ����ش��������⣺| 3b |
| 11m |
| 3b |
| 11m |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| V��HCl��/mL | 0.00 | 12.00 | 18.00 | 22.00 | 23.00 | 23.96 | 24.00 | 24.04 | 25.00 | 26.00 | 30.00 |
| pH | 13.1 | 12.6 | 12.2 | 11.7 | 11.4 | 9.9 | 7.0 | 4.0 | 2.7 | 2.4 | 1.9 |
| ָʾ�� | ��ɫ��Χ ��pH�� |
����Χ����ɫ | ||
| ǰ | �м� | �� | ||
| ���� | 3.1��4.4 | �� | ��ɫ | �� |
| ʯ�� | 5.0��8.0 | �� | ��ɫ | �� |
| ��̪ | 8.2��10.0 | �� | �ۺ� | �� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com