
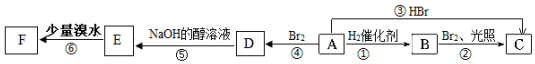
 £Ø½į¹¹¼ņŹ½£©£®
£Ø½į¹¹¼ņŹ½£©£®·ÖĪö A·Ö×ÓŹ½ĪŖC6H12£¬ĒŅAĖłÓŠĢ¼Ō×Ó¶¼ŌŚĶ¬Ņ»Ę½ĆęÉĻ£¬Ó¦ĪŖC£ØCH3£©2=C£ØCH3£©2£¬ĪŖ2£¬3-¶ž¼×»ł-2-¶”Ļ©£¬ÓÉ×Ŗ»Æ¹ŲĻµæÉÖŖBĪŖCH£ØCH3£©2CH£ØCH3£©2£¬CĪŖCH3CH£ØCH3£©CBr£ØCH3£©2£¬DĪŖC£ØCH3£©2BrC£ØCH3£©2Br£¬EĪŖCH2=C£ØCH3£©C£ØCH3£©=CH2£¬ÓėÉŁĮæäåĖ®·“Ӧɜ³ÉµÄFĪŖCH2BrC£ØCH3£©=C£ØCH3£©CH2Br£¬ŅŌ“Ė½ā“šøĆĢā£®
½ā“š ½ā£ŗ£Ø1£©ÓÉŅŌÉĻ·ÖĪöæÉÖŖAĪŖC£ØCH3£©2=C£ØCH3£©2£¬Ćū³ĘĪŖ2£¬3-¶ž¼×»ł-2-¶”Ļ©£¬AµÄĶ¬·ÖŅģ¹¹ĢåÖŠŅ»ĀČ“śĪļÖ»ÓŠŅ»ÖֵďĒ»·¼ŗĶ飬½į¹¹¼ņŹ½ĪŖ £¬
£¬
¹Ź“š°øĪŖ£ŗC£ØCH3£©2=C£ØCH3£©2£»2£¬3-¶ž¼×»ł-2-¶”Ļ©£» £»
£»
£Ø2£©ÓÉ×Ŗ»Æ¹ŲĻµæÉÖŖ¢Ł¢Ū¢Ü¢ŽĪŖ¼Ó³É·“Ó¦£¬¢ŚĪŖČ”“ś·“Ó¦£¬¢ŻĪŖĻūČ„·“Ó¦£¬¹Ź“š°øĪŖ£ŗ¢Ś£»
£Ø3£©ÓÉŅŌÉĻ·ÖĪöæÉÖŖCĪŖCH3CH£ØCH3£©CBr£ØCH3£©2£¬¹Ź“š°øĪŖ£ŗCH3CH£ØCH3£©CBr£ØCH3£©2£»
£Ø4£©D·¢ÉśĻūČ„·“Ӧɜ³ÉE£¬ÓÉD”śEµÄ»Æѧ·½³ĢŹ½ĪŖC£ØCH3£©2BrC£ØCH3£©2Br+2NaOH$”ś_{”÷}^{ŅŅ“¼}$CH2=C£ØCH3£©C£ØCH3£©=CH2+2NaBr+2H2O£¬
¹Ź“š°øĪŖ£ŗC£ØCH3£©2BrC£ØCH3£©2Br+2NaOH$”ś_{”÷}^{ŅŅ“¼}$CH2=C£ØCH3£©C£ØCH3£©=CH2+2NaBr+2H2O£¬
£Ø5£©EĪŖCH2=C£ØCH3£©C£ØCH3£©=CH2£¬ÓėÉŁĮæäåĖ®·“Ӧɜ³ÉµÄFĪŖCH2BrC£ØCH3£©=C£ØCH3£©CH2Br£¬·½³ĢŹ½ĪŖCH2=C£ØCH3£©C£ØCH3£©=CH2+Br2”śCH2BrC£ØCH3£©=C£ØCH3£©CH2Br£¬
¹Ź“š°øĪŖ£ŗCH2=C£ØCH3£©C£ØCH3£©=CH2+Br2”śCH2BrC£ØCH3£©=C£ØCH3£©CH2Br£®
µćĘĄ ±¾Ģāæ¼²éÓŠ»śĪļµÄĶʶĻ£¬ĪŖøßĘµæ¼µć£¬²ąÖŲæ¼²éѧɜµÄ·ÖĪöÄÜĮ¦£¬×¢Ņā°ŃĪÕÓŠ»śĪļµÄ½į¹¹ĢŲµćŗĶŠŌÖŹ£¬°ŃĪÕĪļÖŹµÄ×Ŗ»Æ¹ŲĻµŗĶ·“Ó¦Ģõ¼ž£¬ÄŃ¶Č²»“ó£®



| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŃ”ŌńĢā
| A£® | Ļņ NaAlO2ČÜŅŗÖŠµĪ¼Ó NaHCO3 ČÜŅŗ£¬ÓŠ³ĮµķŗĶĘųĢåÉś³É | |
| B£® | Na”¢Mg”¢Fe µČ½šŹōŌŚŅ»¶ØĢõ¼žĻĀÓėĖ®·“Ó¦¶¼Éś³É H2ŗĶ¶ŌÓ¦µÄ¼ī | |
| C£® | Ķ¬Ö÷×åŌŖĖŲµÄ¼ņµ„ŅõĄė×Ó»¹ŌŠŌŌ½Ē棬Ė®½ā³Ģ¶ČŌ½“ó | |
| D£® | ·ūŗĻĶØŹ½ CnH2n+2µÄ²»Ķ¬Ģž²»Ņ»¶Ø»„ĪŖĶ¬ĻµĪļ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŃ”ŌńĢā
| A£® | 0.50mol•L-1 | B£® | 0.75mol•L-1 | C£® | 1.00mol•L-1 | D£® | 3.18mol•L-1 |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŃ”ŌńĢā
| A£® | AµÄÄÜĮæŅ»¶ØøßÓŚC | |
| B£® | øĆ·“Ó¦ĪŖ·ÅČČ·“Ó¦£¬¹Ź²»±Ų¼ÓČČ¾ĶŅ»¶ØÄÜ·¢Éś | |
| C£® | BµÄÄÜĮæŅ»¶ØøßÓŚD | |
| D£® | AŗĶBµÄ×ÜÄÜĮæŅ»¶ØøßÓŚCŗĶDµÄ×ÜÄÜĮæ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŃ”ŌńĢā
| A£® | Ō×ÓŗĖÄŚÓŠ10 øöÖŠ×ÓµÄŃõŌ×Ó£ŗ${\;}_{8}^{18}$O | |
| B£® | “ĪĀČĖįµÄ½į¹¹Ź½£ŗH-Cl-O | |
| C£® | CO2µÄ±ČĄżÄ£ŠĶ£ŗ | |
| D£® | äå»Æļ§µÄµē×ÓŹ½£ŗ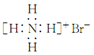 |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŃ”ŌńĢā
| A£® | ×ŌČ»½ēÖŠ²»“ęŌŚÓĪĄėĢ¬µÄĢ¼ | B£® | Ģ¼µÄ»ÆŗĻĪļŹżĮæÖŚ¶ą£¬·Ö²¼¼«¹ć | ||
| C£® | ĻņæÕĘųÖŠÅŷŶžŃõ»ÆĢ¼»įŠĪ³ÉĖįÓź | D£® | Ćŗ”¢ŹÆÓĶ”¢ĢģČ»ĘųŹōÓŚæÉŌŁÉśĢ¼×ŹŌ“ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŃ”ŌńĢā
| A£® | ČōĻņNa2SČÜŅŗÖŠĶØČėSO2Ōņ²śÉśµ»ĘÉ«³Įµķ | |
| B£® | ÕāČżÖÖĖį¶¼ŹĒ¶žŌŖĖį£¬¶¼ÄÜÓėĒāŃõ»ÆÄĘ·“Ӧɜ³ÉĖįŹ½ŃĪŗĶÕżŃĪ | |
| C£® | ÕāČżÖÖĖįµÄĖ®ČÜŅŗ¾ĆÖĆæÕĘųÖŠ¶¼»į±äÖŹ | |
| D£® | ĻņĒāĮņĖį”¢ŃĒĮņĖįČÜŅŗÖŠµĪ¼ÓĀČĖ®¶¼»į·¢ÉśŃõ»Æ»¹Ō·“Ó¦ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ¶ąŃ”Ģā
| A£® | ŅŅČ©ÓėŅų°±ČÜŅŗ·“Ó¦£ŗCH3CHO+2[Ag£ØNH3£©2]OH $\stackrel{”÷}{”ś}$CH3COO-+NH4++2Ag”ż+3NH3+H2O | |
| B£® | “×ĖįČÜŅŗÓėŠĀÖĘĒāŃõ»ÆĶ·“Ó¦£ŗ2CH3COOH+Cu£ØOH£©2”śCu2++2CH3COO-+2H2O | |
| C£® | ±½·ÓÄĘČÜŅŗÖŠĶØČėÉŁĮ涞Ńõ»ÆĢ¼£ŗ2C6H5O-+CO2+H2O”ś2C6H5OH+CO32- | |
| D£® | äåŅŅĶéĖ®½āÉś³ÉŅŅ“¼£ŗCH3CH2Br+OH-$”ś_{”÷}^{H_{2}O}$CH3CH2OH+Br- |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŃ”ŌńĢā
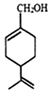
| A£® | ·Ö×ÓŹ½ĪŖC10H16O | |
| B£® | ×ĻĖÕ“¼ĪŖ·¼Ļć×å»ÆŗĻĪļ | |
| C£® | »·ÉĻµÄŅ»ĀČČ”“śĪļÓŠĖÄÖÖ | |
| D£® | Ź¹ĖįŠŌøßĆĢĖį¼ŲČÜŅŗŗĶäåĖ®ĶŹÉ«µÄŌĄķĻąĶ¬ |
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com