
��13�֣�
����ij������Һ�ۺ��ϵ�Ũ�ȵ�Na2Cr2O7��Fe2(SO4)3���Ʊ�K2Cr2O7��
�������£�
����NaOH��Һ��pH��3.6���������ɫ���������ˣ�
������Һ�м���Na2SO3��һ������������Na2SO4��
�������Na2SO4�����Һ��pHԼΪ5���õ�Cr(OH)3������
������KOH���������£���Cr(OH)3�м�������H2O2��Һ���õ���ɫ��Һ��
�������ɫ��Һ�м�������A����Һ��Ϊ�Ⱥ�ɫ��һ��������õ�K2Cr2O7���壻
�����ⶨK2Cr2O7����Ĵ��ȡ�
��֪��Cr2O72�����Ⱥ�ɫ����H2O 2CrO42������ɫ����2H+
2CrO42������ɫ����2H+
��1��������к��ɫ�����Ļ�ѧʽ�� ��
��2��������м���Na2SO3��Ŀ���� ��
��3��������з�Ӧ�����ӷ���ʽ�� ��
��4��������м��������A������ ��������ţ�
a��KOH b��K2CO3 c��H2SO4 d��SO2
��5��������IJ����ǣ�ȡ0.45 g K2Cr2O7��Ʒ�����Һ���ữ�����18.00 mL
0.50 mol/L��FeSO4��Һ��ǡ��ʹCr2O72����ȫת��ΪCr3+����Ʒ��K2Cr2O7�Ĵ����� ����ע��K2Cr2O7��Ħ������Ϊ294 g/mol��
��6����Ⱥ�ɫ��K2Cr2O7��Һ�У��μ�Ba(NO3)2��Һ��������ɫ��������ҺpH��С�����Ʋ��ɫ������ ����ҺpH��С��ԭ���� ��
��13�֣�
��1��Fe(OH)3 ��1�֣�
��2����+6�۵�Cr��ԭΪ+3�� ��2�֣�
��3��2Cr(OH)3��3H2O2��4OH-��2CrO42-��8H2O ��2�֣�
��4��c ��2�֣���
��5��98% ��2�֣�
��6��BaCrO4 ��2�֣���
K2Cr2O7��Һ�д���ƽ�⣺Cr2O72�����Ⱥ�ɫ��+H2O 2CrO42������ɫ��+2H+������Ba(NO3)2��Һ����BaCrO4������c (CrO42��)���ͣ�ƽ�������ƶ���c (H+)����2�֣�
2CrO42������ɫ��+2H+������Ba(NO3)2��Һ����BaCrO4������c (CrO42��)���ͣ�ƽ�������ƶ���c (H+)����2�֣�
���������������1��������к��ɫ������������������ѧʽ��Fe(OH)3��
��2�����������Cr(OH)3�������ɣ�˵��֮ǰ����Һ�д���Cr3+�����Լ���Na2SO3��Ŀ���ǽ�+6�۵�Cr��ԭΪ+3�ۣ�
��3��������֪�û�ɫ��2CrO42������ɫ�����Բ�����з�����������ԭ��Ӧ����Ӧ�����ӷ���ʽ��2Cr(OH)3��3H2O2��4OH-��2CrO42-��8H2O��
��4������Cr2O72�����Ⱥ�ɫ����H2O 2CrO42������ɫ����2H+��֪�������A���ʿ�ʹƽ�������ƶ�������Ӧ���������ʣ��ų�ab������������л�ԭ�ԣ�����CrO42����Ӧ������Cr3+���������CrO42����Ӧ������ʹƽ�������ƶ�����ѡc��
2CrO42������ɫ����2H+��֪�������A���ʿ�ʹƽ�������ƶ�������Ӧ���������ʣ��ų�ab������������л�ԭ�ԣ�����CrO42����Ӧ������Cr3+���������CrO42����Ӧ������ʹƽ�������ƶ�����ѡc��
��5��FeSO4�����ʵ�����0.018L��0.5mol/L=0.009mol����������Fe3+ʧ���ӵ����ʵ�����0.009mol����
K2Cr2O7�����ʵ�����x����ԭ��Cr3+���õ��ӵ����ʵ�����2x��3�����ݵ�ʧ�����غ㣬��
2x��3=0.009mol������x=0.0015mol�����Ʒ��K2Cr2O7�Ĵ�����0.0015mol��294g/mol��0.45g��100%=98%;
��6�������ӵ���ɫ�жϸû�ɫ������BaCrO4 ����Һ�д���ƽ�⣺Cr2O72�����Ⱥ�ɫ��+H2O 2CrO42������ɫ��+2H+������Ba(NO3)2��Һ����BaCrO4������ʹc (CrO42��)���ͣ�ƽ�������ƶ���c (H+)����pH���͡�
2CrO42������ɫ��+2H+������Ba(NO3)2��Һ����BaCrO4������ʹc (CrO42��)���ͣ�ƽ�������ƶ���c (H+)����pH���͡�
���㣺����������жϼ���ѧʽ�����ӷ���ʽ����д��������Ϣ��������������ԭ��Ӧ�ļ��㣬��ѧƽ����ƶ�



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
������ʵ����ʵ�еõ��Ľ��ۣ���ȷ����
| | ʵ����ʵ | ���� |
| A | ����ͭƬ����һ�ֽ���M�õ������ӣ�ͬʱ����ϡ�����У�ͭƬ����������ð�� | ������ԣ�M > Cu |
| B | ��SO2ͨ������KMnO4��Һ�У���ɫ��ȥ | SO2��Ư���� |
| C | CO2ͨ�뱽������Һ�У����ֻ��� | ���ԣ����� < ̼�� |
| D | Cl2��ˮ��Һ���Ե��� | Cl2�ǵ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��16�֣�������ȫ������������ȫ����Ҫ���ϡ�������������ײ��˲�䣬��ȫװ��ͨ����ʹ���еķ�ĩ�ֽ��ͷų������ĵ����γ����ң��Ӷ�����˾�����˿������˺���Ϊ�о���ȫ���ҹ����Ļ�ѧԭ����ȡ��ȫװ���еķ�ĩ����ʵ�顣����ɷ�����ȷ���÷�ĩ��Na��Fe��N��O����Ԫ�ء�ˮ������������������ĩ�����ܽ⡣����⣬������Ϊ������ף�������Ϊ����ɫ���壬���������ᡣ
ȡ13.0g������ף�����ʹ����ȫ�ֽ⣬���ɵ����͵����ң����ɵĵ����ۺϳɱ�״���µ����Ϊ6.72L���������ڸ��¸����������������벻�������ɫ��ĩ��Ӧ���ɵ���ɫ�����������һ�ֵ��ʡ��������������Ӵ���ת��Ϊ�������Ρ�
��ش��������⣺
��1���Ļ�ѧʽΪ �����ĵ���ʽΪ ��
��2�������������ɫ��ĩ������Ӧ�Ļ�ѧ����ʽΪ ��
��3�������ڿ�����ת��Ϊ̼�����Σ���Ӧ�Ļ�ѧ����ʽΪ ��
��4�����������У��п�����Ϊ��ȫ�����к���ɫ��ĩ���Ʒ���� ��
A�� KCl B�� KNO3 C�� Na2S D�� CuO
��5��ijͬѧ��������ڿ�����Ҳ����ת��Ϊ��һ���Ρ������ڿ����з���һ��ʱ���Ϊ̽����ת������ijɷ֣���������·�����
A��ȡa�˻����������ϡ�����ַ�Ӧ���ݳ������ü�ʯ�����գ�����b��
B��ȡa�˻����������ϡ�����ַ�Ӧ�����ȡ����ɡ����գ���b�˹���
C��ȡa�˻�����ּ��ȣ���b�˹���
D��ȡa�˻����������Ca(OH)2��Һ��ַ�Ӧ�����ˡ�ϴ�ӡ���ɣ���b�˹��塣
���з���C���ȵõ���ʦ�϶����ݸ÷���ʵ�����b��a���� ��ϵ����֤�������������εĻ�������Ϊ��������C�⣬�����ķ������� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
������ͼ��ʾװ�ã�β�����������ԣ�����ȡһ����̼�������Բⶨijͭ����Ʒ������CuO��ĩ���н���ͭ�ĺ���
��1���Ʊ�һ����̼�Ļ�ѧ����ʽ���� ����
��2�������У��۲쵽��Ӧ���з������������� ����β������Ҫ�ɷ����� ����
��3����Ӧ��ɺ���ȷ�IJ���˳��Ϊ�� ��������ĸ��
a���ر�©������ b��Ϩ��ƾ�1 c��Ϩ��ƾ���2
��4���������г�ȡͭ����Ʒ5.0g����ַ�Ӧ��Ӧ����ʣ����������Ϊ4.8g����ԭ��Ʒ�е���ͭ����������Ϊ�� ����
��5����Ũ���ᡢŨ���ᡢ����ˮ��˫��ˮ��ѡ�ú��ʵ��Լ������һ���ⶨ��Ʒ�н���ͭ���������ķ�����
����Ʒ�������Ҫ�����ǣ����������������̵�ϸ�ڣ��� ����
��д���йط�Ӧ�Ļ�ѧ����ʽ�� ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
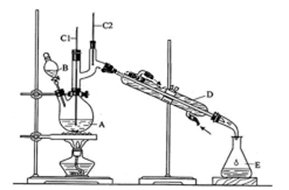
ijʵ��С�����÷�Ӧ2CuO��2Cl2 2CuCl2��O2�ⶨͭ�Ľ������ԭ���������ɹ�ѡ���װ����ͼ��ʾ��
2CuCl2��O2�ⶨͭ�Ľ������ԭ���������ɹ�ѡ���װ����ͼ��ʾ��

����һ��ͨ���ⶨ��Ӧ��CuO������m(CuO)�Ͳ���O2�����V(O2)���ⶨͭ�Ľ������ԭ��������
(1)����������������ý���(ͼ��δ����)��ѡ���������ϳ�һ��ʵ��װ�ã�����˳��Ϊa��(����)(����)��(����)(����)��(����)(����)��(����)(����)��b��
(2)װ��B���ɸ���ܺͼ�ʽ�ζ��ܸ�����ɵIJ������������װ�ã�ʵ��ǰ�ζ���Һ�������ΪV1 L��ʵ���ָ������£�����װ������Һ����ƽ��õ�ĩ����ΪV2 L��������ʱ����Ħ�����ΪVm L��mol��1����Eװ����CuO������Ϊm1 g����ַ�Ӧ������CuCl2������Ϊm2 g����ͭ�Ľ������ԭ�������ı���ʽΪ
[�ú�m1��V1��V2�Ĵ���ʽ��ʾ]��
(3)������ͭ�л���ͭ����ⶨ��� (�ƫ����ƫС������Ӱ�족)��
(4)װ��E��ʵ������е���Ҫ������ ��
������������A��D��E��F����װ��(β����������װ�ô���)��ɲⶨ����
(5)����Ϊ�ⶨ���������� (д��һ��)������ⶨ����������д��ͭ�Ľ������ԭ�������ı���ʽ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ijƷ������Һ��װ˵���IJ�������ժ¼���£���Ҫ��Ч�ɷ�Ϊ�������ƣ���Ч�Ⱥ���8 000 mg/L��10 000 mg/L�������ڸ���Ҿ���Ʒ���;ߡ���֯����ȵ��������Բ�ɫ֯���������ɫ���á���������˿��ë��������Ƥ�������棬����������ͭ��̼����Ʒ����Ʒ���ܷ⣬�������������档���������ʵ��̽�����̣�
(һ)�Ķ����ϣ���ȡ��Ϣ
(1)�Ʋ������Һ������ԣ�________��ԭ����(�����ӷ���ʽ��ʾ)________��
(2)������Һ�����еĻ�ѧ������_____�Ժ�_____�ԡ�
(3)�Ӹ�����Һ�ı���Ҫ�������������ʧЧ�����������Ҫ��_____��
(��)ȷ��Ҫ�о�������
������Һ��̼����Ʒ�Ƿ��и�ʴ���ã�
(��)��Ʒ�����ʵʩ̽��
���ձ�ȡ������Ʒ����һ�����������������ձ�������һ��ʱ�䡣�Կ��ܲ��������������¼��裺����٣�������������ڣ�����������
(4)���ڼ���٣���������������ʴ��Ԥ�ڵ�ʵ��������__________��
(5)���ڼ���ڣ�������______________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
���ü�������������ȡ����Ӧ��ȡ����Ʒ����������ڹ�ҵ���ѳ�Ϊ��ʵ��ij��ѧ��ȤС����ʵ������ģ���������̡�����Ƶ�ģ��װ�����£�
����Ҫ����գ�
(1)Bװ�������ֹ��ܣ��پ��Ȼ�����壻�� ���� ��
(2)��V(Cl2)/V(CH4)��x��������������������Ȼ��⣬��xֵӦ ��
(3)Dװ�õ�ʯ���о��Ȼ���KI��ĩ���������� ��
(4)Eװ�õ������� (����)��
A���ռ����� B.��������
C����ֹ���� D�������Ȼ���
(5)Eװ�ó����������⣬�������л����E�з�����������ѷ���Ϊ (����뷽������)��
(6)��װ�û���ȱ�ݣ�ԭ����û�н���β����������β����Ҫ�ɷ�Ϊ (����)��
A.CH4��B��CH3Cl��C��CH2C12��D��CHCl3��E��CCl4
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ʯ����Ҫ�ɷ�ΪCaC2������ˮ��Ӧ����C2H2����C2H2�׳Ƶ�ʯ����ijͬѧ������·����ⶨ��ʯ��CaC2�Ĵ���(���ʲ��μӷ�Ӧ)��
ʵ��ԭ����CaC2 +2H2O==Ca(OH)2+C2H2 ��
ͨ���ⶨ���ɵ���������(��״��������ȷ����Ʒ��CaC2�ĺ�����
ʵ�鲽�裺
�ٳ�ȡ��Ʒ1.2g��
�ڰ�1.2g��Ʒ�������������õ����巢��װ�ã���ͼ��ʾ��
������Ʒ�е���ˮ�������ٲ������ݣ�����Ͳ��ˮ��������Ͳ��Һ����360mL��ǡ��ˮ����Һ����ƽ��
����Ϊ��Ӧ������ƿ����24mLҺ�塣
��ش��������⣺
��1������ʵ�����õIJ��������е��ܡ�ˮ�ۡ� �� �� ��
��2����ʵ��ǰ��1000mL��500 mL��250 mL����Ͳ���ã���Ӧѡ�� mL����Ͳ��
��3��������������� mL��Ϊ��֤�������������ȷ�ԣ���ȡ��Ͳ�̶�ʱӦע��������� ��
��4������C2H2ͨ��KMnO4��Һ�У�KMnO4��Һ���Ϻ�ɫ���dz����Ӧ�Ļ�ѧ����ʽΪ��KMnO4+��C2H2����H2SO4������K2SO4����MnSO4����CO2���� ������ʵ�����ɵ�C2H2��ȫ��KMnO4��Һ������������0.1 mol��L-1��KMnO4��Һ mL��
��5��ͨ���������ݼ��㣬�ɵ���Ʒ��CaC2�Ĵ���Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
(12��)����ȩ��һ�ֻ���ԭ�ϡ�ijʵ��С����������װ�úϳ�����ȩ�������ķ�Ӧ���£� ����Ӧ��Ͳ������������б����£�
����Ӧ��Ͳ������������б����£�
| | �е�/�� | �ܶ�/g��cm-3 | ˮ���ܽ��� |
| ������ | 117.2 | 0.8109 | �� |
| ����ȩ | 75.7 | 0.8017 | �� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com