
| A£®“æ¼ī | B£®ŹģŹÆ»Ņ | C£®ŹÆ»ŅŹÆ | D£®š¤ĶĮ |
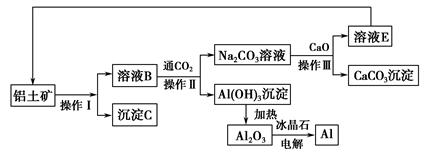
 CaSiO3+CO2ӟ
CaSiO3+CO2”ü Al(OH)3+3H+””Mg(HCO3)2
Al(OH)3+3H+””Mg(HCO3)2 MgCO3”ż+CO2”ü+H2O””MgCO3+H2O
MgCO3”ż+CO2”ü+H2O””MgCO3+H2O Mg(OH)2+CO2”ü
Mg(OH)2+CO2”ü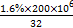 mol,n(H2SO4)=n(S)=105 mol,Ņ»ĢģÉś²śm(H2SO4)=98”Į105 g,98%µÄĮņĖįÖŹĮæ=1”Į107 g="10" t;(2)Ńō¼«NiŹ§Č„µē×ÓŠĪ³ÉÄųĄė×Ó,ÓėŅõ¼«µĆµē×ÓÄųĄė×ÓµÄĪļÖŹµÄĮæĻąĶ¬
mol,n(H2SO4)=n(S)=105 mol,Ņ»ĢģÉś²śm(H2SO4)=98”Į105 g,98%µÄĮņĖįÖŹĮæ=1”Į107 g="10" t;(2)Ńō¼«NiŹ§Č„µē×ÓŠĪ³ÉÄųĄė×Ó,ÓėŅõ¼«µĆµē×ÓÄųĄė×ÓµÄĪļÖŹµÄĮæĻąĶ¬


| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā
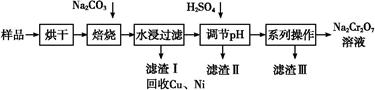
 ””””””””Na2CrO4+””””””””CO2+””””””””””””
””””””””Na2CrO4+””””””””CO2+””””””””””””  Cr2O72”Ŗ+H2O
Cr2O72”Ŗ+H2O| ĪĀ¶Č Čܽā¶Č »ÆѧŹ½ | 20 ”ę | 60 ”ę | 100 ”ę |
| Na2SO4 | 19.5 | 45.3 | 42.5 |
| Na2Cr2O7 | 183 | 269 | 415 |
| Na2CrO4 | 84 | 115 | 126 |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗµ„Ń”Ģā
| A£®½šŹō±»ČĖĄąæŖ·¢ĄūÓƵēóÖĀÄźĻŽÖ®ĖłŅŌÓŠĻČŗó£¬Ö÷ŅŖČ”¾öÓŚ½šŹōŌŚµŲæĒÖŠµÄŗ¬Įæ¶ąÉŁ |
| B£®ŗ£Ė®ÖŠŗ¬ÓŠ·įø»µÄµāŌŖĖŲ£¬Ņņ“Ėµā±»³ĘĪŖ”°ŗ£ŃóŌŖĖŲ”± |
| C£®øßĀÆĮ¶ĢśµÄÖ÷ŅŖ·“Ó¦ŌĄķŹĒĢ¼ŌŚøßĪĀĻĀÖ±½Ó½«Ńõ»ÆĢś»¹Ō³ÉĢś |
| D£®ŌŚŗ£Ė®ÖŠ¼ÓČėŹÆ»ŅČéæɵĆĒāŃõ»ÆĆ¾³Įµķ£¬ÕāŹĒ“Óŗ£Ė®ÖŠø»¼ÆĆ¾µÄ»ł±¾·½·Ø |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā
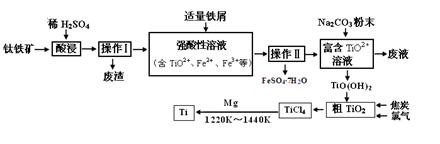

²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗµ„Ń”Ģā
| A£®Na Mg Al | B£®Na K Zn Fe |
| C£®Zn Fe Cu Ag | D£®Mg Al Zn Fe |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗµ„Ń”Ģā
| A£®“óĮæĀĮĶĮæó | B£®µē½āŃõ»ÆĀĮÖĘČ”ĀĮµÄ·½·Ø |
| C£®ÓĆ½šŹōÄĘ»¹ŌŃõ»ÆĀĮµÄ·½·Ø | D£®ĪŽĖ®ĀČ»ÆĀĮÓė¼ŲÖĘČ”ĀĮµÄ·½·Ø |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā
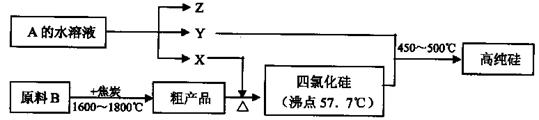
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com