
������A��B����ѧ���������ʣ�������������ֻ�ܴ��±���ѡ��
| ������ | K����Na����Fe2����Ba2����NH |
| ������ | OH����NO |
(1)��A��B��ˮ��Һ��Ϊ��ɫ����A��ˮ��Һ��ǿ���ԣ�B��ˮ��Һ��ǿ���ԡ���Ϻ����������ϡ����İ�ɫ��������ʹʪ��ĺ�ɫʯ����ֽ����ɫ�����塣
��B�Ļ�ѧʽΪ__________________________________________________________��
��A��B��Һ��ϼ��ȷ�Ӧ�����ӷ���ʽ_____________________________________��
(2)��A��ˮ��Һ��dz��ɫ��B��ˮ��Һ��ɫ������ɫ��ӦΪ��ɫ����A��ˮ��Һ�м���ϡ���������������ټ���B����Һ��ƣ���A��B��ˮ��Һ����������Ա仯����
��A�Ļ�ѧʽΪ__________________________________________________________��
�ھ�����������������Һ��Ƶ�ԭ�����������(�����ӷ���ʽ��ʾ)
��________________________________________________________________________��
��________________________________________________________________________��
������һ������֤��������Һ��Ƶ�ԭ��___________________________________��
�𰸡�(1)��Ba(OH)2����H����SO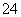 ��NH
��NH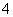 ��Ba2����2OH��
��Ba2����2OH��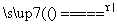 BaSO4����NH3����2H2O
BaSO4����NH3����2H2O
(2)��FeI2
��6I����2NO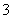 ��8H��===3I2��2NO����4H2O
��8H��===3I2��2NO����4H2O
2I����Fe2����NO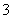 ��4H��===I2��NO����2H2O��Fe3����3Fe2����NO
��4H��===I2��NO����2H2O��Fe3����3Fe2����NO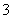 ��4H��===NO����2H2O��3Fe3��(ֻҪ��I����Fe2�����뷴Ӧ����ƽ����)
��4H��===NO����2H2O��3Fe3��(ֻҪ��I����Fe2�����뷴Ӧ����ƽ����)
��ȡ���������Һ���Թ��У��μӼ���KSCN��Һ������Ѫ��������(������������)
������(1)A��Һ��ǿ���ԣ�Ӧ����HSO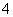 ��B��Һ��ǿ���ԣ�����A��B��ϲ������������ж�AΪNH4HSO4��BΪBa(OH)2��(2)A��Һ��dz��ɫ��Ӧ����Fe2����B��Һ����ɫ��Ӧ�ʻ�ɫ��Ӧ����Na��������Fe2������Һ�м���ϡ��������������˵��������NO
��B��Һ��ǿ���ԣ�����A��B��ϲ������������ж�AΪNH4HSO4��BΪBa(OH)2��(2)A��Һ��dz��ɫ��Ӧ����Fe2����B��Һ����ɫ��Ӧ�ʻ�ɫ��Ӧ����Na��������Fe2������Һ�м���ϡ��������������˵��������NO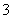 ������B����Һ��ƣ�˵��B�����������¾���ǿ�����ԣ�BӦΪNaNO3��AӦΪFeI2�����ԭ�����Ϊ��6I����2NO
������B����Һ��ƣ�˵��B�����������¾���ǿ�����ԣ�BӦΪNaNO3��AӦΪFeI2�����ԭ�����Ϊ��6I����2NO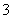 ��8H��===3I2��2NO����4H2O����2I����
��8H��===3I2��2NO����4H2O����2I����
Fe2����NO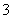 ��4H��===I2��NO����2H2O��Fe3����3Fe2����NO
��4H��===I2��NO����2H2O��Fe3����3Fe2����NO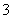 ��4H��===3Fe3����NO����2H2O��ȡ������ɫ��Һ���Թ��У��μӼ���KSCN��Һ������Ѫ����������
��4H��===3Fe3����NO����2H2O��ȡ������ɫ��Һ���Թ��У��μӼ���KSCN��Һ������Ѫ����������



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��ҵ����ƽ�VOSO4�е�K2SO4��SiO2���ʳ�ȥ�����յõ�V2O5���������£���ش��������⣺
�� ��������÷����ijɷ��� ��д��ѧʽ��������I������ ��
�� ����ڡ��۵ı仯���̿ɼ�Ϊ����ʽR��ʾVO2+��HA��ʾ�л���ȡ������
R2(SO4)n (ˮ��)+ 2nHA���л��㣩 2RAn���л��㣩 + nH2SO4 (ˮ��)
2RAn���л��㣩 + nH2SO4 (ˮ��)
������ȡʱ��������������ԭ���� ��
����X�Լ�Ϊ ��
�� �ݵ����ӷ���ʽΪ ��
�� 25��ʱ��ȡ����������������õ��������ʺ���ҺpH֮���ϵ���±���
| pH | 1.3 | 1.4 | 1.5 | 1.6 | 1.7 | 1.8 | 1.9 | 2.0 | 2.1 |
| ��������% | 88.1 | 94.8 | 96.5 | 98.0 | 98.8 | 98.8 | 96.4 | 93.1 | 89.3 |
����ϱ�����ʵ�������У����м��백ˮ��������Һ�����pHΪ ��
����������Ϊ93.1%ʱ������Fe(OH)3����������Һ��c(Fe3+)< ��
(��֪��25��ʱ��Ksp[Fe(OH)3]=2.6��10-39)
�� �ù��������У�����ѭ�����õ������� �� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��������������Һ���ܴ���������� (����)
A��Fe3����SCN����I����K��
B��K����Al3����SO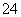 ��MnO
��MnO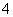
C��H����NO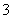 ��Fe2����Na��
��Fe2����Na��
D��Cu2����NH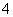 ��Br����OH��
��Br����OH��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
ij��Һ���Т��е�����(���±�)�����â��е��Լ���������Һ���Ƿ��Т��е����ӣ��ܹ��ɹ��������(˵������ʵ������в��������м��ȡ����ˡ���ȡ�Ȳ���)(����)
| ѡ�� | �� | �� | �� |
| A | ϡ��ˮ | Al3�� | Mg2�� |
| B | ϡ���ᡢAgNO3��Һ | Cl�� | I�� |
| C | KSCN��Һ | Fe2�� | Fe3�� |
|
| ϡ���ᡢBaCl2��Һ | SO | SO |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����ʵ�鷽���У����ܴﵽʵ��Ŀ�ĵ��ǣ�
| ѡ�� | ʵ��Ŀ�� | ʵ�鷽�� |
| A | ����CH3CH2Br��NaOH��Һ���Ƿ���ˮ�� | ��CH3CH2Br��NaOH��Һ���ȡ���ȴ��ȡ���ϲ�ˮ��Һ����ϡHNO3�ữ������AgNO3��Һ���۲��Ƿ��������ɫ���� |
| B | ����Fe(NO3)2�����Ƿ����������� | ��Fe(NO3)2��Ʒ����ϡ����μ�KSCN��Һ���۲���Һ�Ƿ��� |
| C | ��֤Br2��������ǿ��I2 | ��������ˮ����KI��Һ�У��ټ���CCl4�������ã��ɹ۲쵽�²�Һ�����ɫ |
| D | ��֤Fe(OH)3���ܽ��С��Mg(OH)2 | ��FeCl3��Һ����Mg(OH)2����Һ�У����ɹ۲쵽�����ɰ�ɫ��Ϊ���ɫ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��Ⱦ�����Чȥ������Դ�ij�������ǻ�ѧ�츣�������Ҫ�о����⡣ij�о�С���������̿���Ҫ�ɷ�ΪMnO2������������������ͭ�����Ƚ�����������������ͨ�����¼����̣����ѳ�ȼúβ���е�SO2�����Ƶõ�ز���MnO2����Ӧ������ʡ�ԣ���

��ش��������⣺
��1��������������ʵ���� ��ѡ��������ĸ��ţ���
A����������ۺ����� B����ɫ��Ⱦ�ļ��� C������ļ���
��2����MnCO3�ܳ�ȥ��Һ��Al3����Fe3������ԭ���� ��
��3����֪��25�桢101kPaʱ��Mn(s)+O2(g)=MnO2(s) DH=-520kJ/mol
S(s)+O2(g)=SO2(g) DH=-297kJ/mol
Mn(s)+S(s)+2O2(g)=MnSO4(s) DH=-1065kJ/mol
SO2��MnO2��Ӧ������ˮMnSO4���Ȼ�ѧ����ʽ��
��4��MnO2�����������ݲ��ϡ��ö��Ե缫���MnSO4��Һ���Ƶ�MnO2���������ĵ缫��Ӧʽ��
��5��MnO2�Ǽ���п�̵�ص��������ϡ�����п�̵�طŵ�ʱ�������ĵ缫��Ӧʽ
��
��6�������ѳ���SO2ֻ�����̿���MnO2��Ӧ������ͼʾ���̣���a m3(��״��)��SO2���������Ϊb%��β��ͨ�����SO2���ѳ���Ϊ89.6%�����յõ�MnO2������ckg�����ȥ��������ͭ����������ʱ�����������Ԫ���൱��MnO2 kg��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��ŵ�����һ���������ʪ�Թؽ���ҩ���ͨ�����·����ϳɣ�
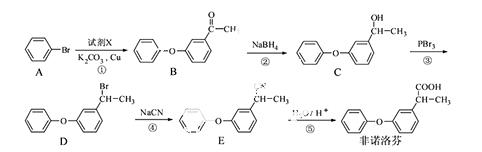
��ش��������⣺
��1����ŵ����еĺ���������Ϊ �� �������ƣ���
��2����Ӧ���м�����Լ�X�ķ���ʽΪC8H8O2��X�Ľṹ��ʽΪ ��
��3���������岽��Ӧ�У�����ȡ����Ӧ���� ������ţ���
��4��B��һ��ͬ���칹����������������
�����ܷ���������Ӧ����ˮ�����֮һ����FeCl3��Һ������ɫ��Ӧ��
������������6�ֲ�ͬ��ѧ�������⣬�ҷ����к�������������
д����ͬ���칹��Ľṹ��ʽ�� ��
��5����������֪ʶ����������Ϣ��д���� Ϊԭ���Ʊ�
Ϊԭ���Ʊ�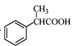 �ĺϳ�·������ͼ�����Լ����ã����ϳ�·������ͼʾ�����£�
�ĺϳ�·������ͼ�����Լ����ã����ϳ�·������ͼʾ�����£�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Զ�����ȡ��һ�ַ��㻯��������ʽΪC8H8O3����FeCl3��Һ�����������ɫ���ܷ���������Ӧ���÷�Ӧ���ܵĽṹ��ʽ��

�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com