
ЁОЬтФПЁПЃЈЛЏбЇЃЮяжЪНсЙЙгыаджЪЃЉ
ЃЈ1ЃЉвбжЊAКЭBЮЊЕкШ§жмЦкдЊЫиЃЌЦфдзгЕФЕквЛжСЕкЫФЕчРыФмШчЯТБэЫљЪОЃК
ЕчРыФм/kJЁЄmolЃ1 | I1 | I2 | I3 | I4 |
A | 578 | 1817 | 2745 | 11578 |
B | 738 | 1451 | 7733 | 10540 |
AЭЈГЃЯд_____МлЃЌAЕФЕчИКад________BЕФЕчИКадЃЈЬюЁАЃОЁБЁЂЁАЃМЁБЛђЁАЃНЁБЃЉЁЃ
ЃЈ2ЃЉзЯЭтЙтЕФЙтзгЫљОпгаЕФФмСПдМЮЊ399 kJЁЄmolЃ1ЁЃИљОнЯТБэгаЙиЕААзжЪЗжзгжаживЊЛЏбЇМќЕФаХЯЂЃЌЫЕУїШЫЬхГЄЪБМфееЩфзЯЭтЙтКѓЦЄЗєвзЪмЩЫКІЕФдвђ__________________________________________________________________________ЁЃ
ЙВМлМќ | CЃC | CЃN | CЃS |
МќФм/ kJЁЄmolЃ1 | 347 | 305 | 259 |
зщГЩЕААзжЪЕФзюМђЕЅЕФАБЛљЫсжаЕФЬМдзгдгЛЏРраЭЪЧ________________ЁЃ
ЃЈ3ЃЉЪЕбщжЄУїЃКKClЁЂMgOЁЂCaOЁЂTiNет4жжОЇЬхЕФНсЙЙгыNaClОЇЬхНсЙЙЯрЫЦЃЈШчЭМЫљЪОЃЉЃЌ
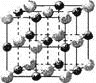
Цфжа3жжРызгОЇЬхЕФОЇИёФмЪ§ОнШчЯТБэЃК
РызгОЇЬх | NaCl | KCl | CaO |
ОЇИёФм/kJЁЄmolЃ1 | 786 | 715 | 3401 |
дђИУ 4жжРызгОЇЬхЃЈВЛАќРЈaClЃЉШлЕуДгИпЕНЕЭЕФЫГађЪЧЃК_________________ЁЃ
ЦфжаMgOОЇЬхжавЛИіMg2+жмЮЇКЭЫќзюСкНќЧвЕШОрРыЕФMg2+га_______ИіЁЃ
ЃЈ4ЃЉН№ЪєбєРызгКЌЮДГЩЖдЕчзгдНЖрЃЌдђДХаддНДѓЃЌДХМЧТМадФмдНКУЁЃРызгаЭбѕЛЏЮяV2O5КЭCrO2жаЃЌЪЪКЯзїТМвєДјДХЗлдСЯЕФЪЧ_____________ЁЃ
ЃЈ5ЃЉФГХфКЯЮяЕФЗжзгНсЙЙШчгвЭМЫљЪОЃЌЦфЗжзгФкВЛКЌга____ЃЈЬюађКХЃЉЁЃ
AЃЎРызгМќ BЃЎМЋадМќ
CЃЎН№ЪєМќ DЃЎХфЮЛМќ
EЃЎЧтМќ FЃЎЗЧМЋадМќ
ЃЈ6ЃЉЮТЪваЇгІЃЌПЦбЇМвЩшМЦЗДгІЃКCO2+4H2ЁњCH4+2H2OвдМѕаЁПеЦјжаCO2ЁЃ
Шєга1mol CH4ЩњГЩЃЌдђга______molІвМќКЭ______molІаМќЖЯСбЁЃ
ЁОД№АИЁП+3 ЃО зЯЭтЙтОпгаЕФФмСПБШЕААзжЪЗжзгжаЕФЛЏбЇМќCЃCЁЂCЃNЁЂCЃSЕФМќФмДѓЃЌзЯЭтЙтЕФФмСПзувдЪЙетаЉМќЖЯСбЃЌДгЖјЦЦЛЕЕААзжЪЗжзг sp2ЁЂsp3 TiNЃОMgOЃОCaOЃОKCl 12 CrO2 AC 6 2
ЁОНтЮіЁП
ЃЈ1ЃЉAКЭBОљЮЊЕкШ§жмЦкдЊЫиЃЌИљОнЦфж№МЖЕчРыФмПЩШЗЖЈЫќУЧЕФЛЏКЯМлЃЌНјЖјШЗЖЈЫќУЧЕФЕчИКадЁЃ
ЃЈ2ЃЉЕААзжЪЗжзгжаЕФЛЏбЇМќЕФФмСПКЭзЯЭтЙтЕФЙтзгЫљОпгаЕФФмСПБШНЯПЩжЊзЯЭтЙтЖдШЫЬхЪЧЩЫКІЁЃзщГЩЕААзжЪЕФзюМђЕЅЕФАБЛљЫсЪЧИЪАБЫсH2NCH2COOHЃЌИљОнЗжзгжаЕФЛЏбЇМќПЩжЊЬМдзгдгЛЏРраЭЁЃ
ЃЈ3ЃЉОЇИёФмКЭРызгАыОЖвдМАРызгЫљДјЕчКЩгаЙиЃЌОЇИёФмдНДѓЃЌШлЗаЕудНИпЁЃЗТееNaClЕФОЇЬхНсЙЙПЩжЊMgOОЇЬхжавЛИіMg2+жмЮЇКЭЫќзюСкНќЧвЕШОрРыЕФMg2+ЕФИіЪ§ЁЃ
ЃЈ4ЃЉИљОндзгКЫЭтЕчзгХХВМПЩжЊVКЭCrжаЕФЮДГЩЖдЕчзгЁЃ
ЃЈ5ЃЉИљОнХфКЯЮяЕФЗжзгНсЙЙЃЌПЩжЊЦфЗжзгФкВЛКЌгаЕФзїгУСІЁЃ
ЃЈ6ЃЉЗДгІжаCO2+4H2ЁњCH4+2H2OЃЌга1mol CH4ЩњГЩЃЌдђЯћКФ1molCO2КЭ4molH2ЁЃ
ЃЈ1ЃЉAЕФЕкЫФЕчРыФмЬиБ№ДѓЃЌЫљвдAЭЈГЃЯдЃЋ3МлЁЃЖјBЕФЕкШ§ЕчРыФмЬиБ№ДѓЃЌЫљвдBЯдЃЋ2МлЃЌЫљвдBЕФдзгађЪ§аЁгкAЕФЃЌЫљвдЕчИКадBЕФаЁгкAЕФЁЃ
ЃЈ2ЃЉИљОнЫљИјЪ§ОнПЩжЊЃЌзЯЭтЙтОпгаЕФФмСПБШЕААзжЪЗжзгжаЕФЛЏбЇМќCЃCЁЂCЃNЁЂCЃSЕФМќФмДѓЃЌзЯЭтЙтЕФФмСПзувдЪЙетаЉМќЖЯСбЃЌДгЖјЦЦЛЕЕААзжЪЗжзгЃЌЪЙШЫЬхЪмЕНЩЫКІЁЃзюМђЕЅЕФАБЛљЫсЪЧИЪАБЫсЃЌНсЙЙМђЪНЮЊH2NCH2COOHЃЌЦфжаКЭАБЛљЯрСЌЕФЬМдзгЕФЛЏбЇМќШЋВПЪЧЕЅМќЃЌЫљвдЪЧsp3дгЛЏЃЌєШЛљжаЕФЬМдзгКЌгаЫЋМќЃЌЪєгкsp2дгЛЏЁЃ
ЃЈ3ЃЉгАЯьРызгОЇЬхШлЕуИпЕЭЕФЪЧРызгМќЕФЧПШѕЃЌЖјгАЯьРызгМќЧПШѕЕФЪЧаЮГЩРызгМќЕФвѕбєРызгЕФАыОЖДѓаЁКЭЕчКЩЪ§ЕФЖрЩйЁЃдкTiNжавѕбєРызгЕФЕчКЩЪ§зюЖрЃЌвѕРызгЕФАыОЖзюаЁЃЌЫљвдРызгМќзюЧПЃЌШлЕузюИпЁЃУОРызгАыОЖаЁгкИЦРызгЕФЃЌЫљвдбѕЛЏУОЕФШлЕуИпгкбѕЛЏИЦЕФЃЌИљОнОЇИёФмДѓаЁПЩжЊЃЌбѕЛЏИЦЕФШлЕугІИУИпгкТШЛЏМиЕФЁЃИљОнТШЛЏФЦЕФОЇАћПЩжЊвЛИіNa+жмЮЇКЭЫќзюСкНќЧвЕШОрРыЕФNa+га12ИіЃЌЫљвдMgOОЇЬхжавЛИіMg2+жмЮЇКЭЫќзюСкНќЧвЕШОрРыЕФMg2+вВга12ИіЁЃ
ЃЈ4ЃЉИљОнЙЙдьдРэПЩжЊЃЌН№ЪєVКЭCrжаКЌгаЕФЮДГЩЖдЕчзгЗжБ№ЮЊ3КЭ6ЃЌЫљвдЪЪКЯзїТМвєДјДХЗлдСЯЕФЪЧCrO2ЁЃ
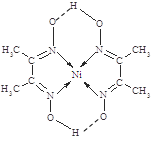
ЃЈ5ЃЉИљОнНсЙЙМђЪНПЩжЊЬМдзгКЭЬМдзгаЮГЩЗЧМЋадМќЃЌЬМКЭЧтаЮГЩМЋадМќЃЌЕЊдзгКЭNiаЮГЩХфЮЛМќЃЌСэЭтбѕКЭЧтЛЙгаЧтМќЃЌВЛДцдкРызгМќКЭН№ЪєМќЁЃ
ЃЈ6ЃЉЧтЦјжаШЋВПЪЧІвМќЁЃCO2жаКЌга2ИіЫЋМќЃЌЫЋМќЪЧгЩ1ИіІвМќКЭ1ИіІаМќЙЙГЩЕФЁЃЫљвдга1mol CH4ЩњГЩЃЌдђга6mlІвМќЖЯСбЃЌ2molІаМќЖЯСбЁЃ


 зДдЊЗЛШЋГЬЭЛЦЦЕМСЗВтЯЕСаД№АИ
зДдЊЗЛШЋГЬЭЛЦЦЕМСЗВтЯЕСаД№АИ жБЭЈЙѓжнУћаЃжмВтдТПМжБЭЈУћаЃЯЕСаД№АИ
жБЭЈЙѓжнУћаЃжмВтдТПМжБЭЈУћаЃЯЕСаД№АИ
| ФъМЖ | ИпжаПЮГЬ | ФъМЖ | ГѕжаПЮГЬ |
| ИпвЛ | ИпвЛУтЗбПЮГЬЭЦМіЃЁ | ГѕвЛ | ГѕвЛУтЗбПЮГЬЭЦМіЃЁ |
| ИпЖў | ИпЖўУтЗбПЮГЬЭЦМіЃЁ | ГѕЖў | ГѕЖўУтЗбПЮГЬЭЦМіЃЁ |
| ИпШ§ | ИпШ§УтЗбПЮГЬЭЦМіЃЁ | ГѕШ§ | ГѕШ§УтЗбПЮГЬЭЦМіЃЁ |
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПЯТСагаЙиЕчНтжЪШмвКЕФа№Ъіе§ШЗЕФЪЧЃЈ ЃЉ
A.ЮЊШЗЖЈ![]() ЪЧЧПЫсЛЙЪЧШѕЫсЃЌПЩВтNaHAШмвКЕФpHЃЌШє
ЪЧЧПЫсЛЙЪЧШѕЫсЃЌПЩВтNaHAШмвКЕФpHЃЌШє![]() ЃЌдђ
ЃЌдђ![]() ЪЧШѕЫсЃЛШє
ЪЧШѕЫсЃЛШє![]() ЃЌдђ
ЃЌдђ![]() ЪЧЧПЫс
ЪЧЧПЫс
B.![]() ЪБЃЌдкАБЫЎЬхЯЕжаВЛЖЯЭЈШы
ЪБЃЌдкАБЫЎЬхЯЕжаВЛЖЯЭЈШы![]() ЃЌЫцзХ
ЃЌЫцзХ![]() ЕФЭЈШыЃЌ
ЕФЭЈШыЃЌ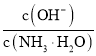 ВЛЖЯМѕЩй
ВЛЖЯМѕЩй
C.ГЃЮТЯТЁЃНЋЕШЬхЛ§ЁЂЕШЮяжЪЕФСПХЈЖШЕФ![]() гыNaC1ШмвКЛьКЯЃЌЮіГіВПЗж
гыNaC1ШмвКЛьКЯЃЌЮіГіВПЗж![]() ОЇЬхЃЌЙ§ТЫЃЌЫљЕУТЫвК
ОЇЬхЃЌЙ§ТЫЃЌЫљЕУТЫвК![]() ЃЌдђТЫвКжаЃК
ЃЌдђТЫвКжаЃК![]()
![]()
D.ЪвЮТЯТЃЌНЋ![]()
![]() ЙЬЬхШмгкЫЎХфГЩ100mLШмвКЃЌЯђШмвКжаМгШы
ЙЬЬхШмгкЫЎХфГЩ100mLШмвКЃЌЯђШмвКжаМгШы![]() ЃЌЫљЕУШмвКжа
ЃЌЫљЕУШмвКжа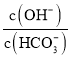 МѕаЁ
МѕаЁ
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПЪЏгЭКЭУКЖМЪЧживЊЕФФмдДКЭЛЏЙЄдСЯЃЌШчЭМ1ЪЧУКЛЏЙЄВњвЕСДЕФвЛВПЗжЁЃ

ЪдгУЫљбЇжЊЪЖЃЌНтОіЯТСаЮЪЬтЃК
ЃЈ1ЃЉВёгЭЪЧгЩЪЏгЭМгЙЄЕУЕНЕФживЊВњЦЗЃЌЫќдкШМЩеЪБЭљЭљУАКкбЬЃЌПЩФмЕФдвђЪЧ____ЁЃ
ЃЈ2ЃЉУКОЙ§ИЩСѓПЩвдЕУЕННЙТЏЦјЁЂУКНЙгЭКЭНЙЬПЕШЁЃУКНЙгЭОЙ§____ЃЈЬюМгЙЄЗНЗЈЃЉПЩЕУЕНЗМЯузхЛЏКЯЮяЁЃ
ЃЈ3ЃЉУКЕФжБНгвКЛЏЪЧУККЭЪЪЕБШмМСЛьКЯдкИпЮТКЭ____ДцдкЯТгы____зїгУЩњГЩвКЬЌШМСЯЕФЙ§ГЬЁЃ
ЃЈ4ЃЉУККЭЪЏгЭЕШЛЏЪЏШМСЯШМЩеХХЗХЕФДѓСПЖўбѕЛЏЬМЛсв§Ц№ШЋЧђЦјКђБфХЏЁЃвЛжжаТЕФДІРэЗНЗЈЪЧНЋЖўбѕЛЏЬМЦјЬхЭЈШыКЌгаГЄЪЏЃЈЕиПЧжазюГЃМћЕФПѓЪЏЃЌКЌСПИпДя60%ЃЉГЩЗжЕФЫЎШмвКРяЃЌЦфжавЛжжЗДгІЕФЛЏбЇЗНГЬЪНЃКKAlSi3O8+CO2+2H2O=KHCO3+XЁ§+3SiO2Ё§ЃЌдђXЕФЛЏбЇЪНЮЊ____ЁЃ
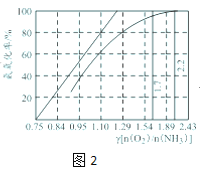
ЃЈ5ЃЉЙЄвЕЩЯжївЊВЩгУАБбѕЛЏЗЈЩњВњЯѕЫсЃЌШчЭМ2ЪЧАБбѕЛЏТЪгыАБ--ПеЦјЛьКЯЦјЬхжабѕАББШЕФЙиЯЕЁЃЦфжажБЯпБэЪОЗДгІЕФРэТлжЕЃЛЧњЯпБэЪОЪЕМЪЩњВњЧщПіЁЃЕБАБбѕЛЏТЪДяЕН100%ЃЌРэТлЩЯІУ[nЃЈO2ЃЉ/nЃЈNH3ЃЉ]=___ЃЌЪЕМЪЩњВњНЋІУЮЌГждк1.7ЁЋ2.2жЎМфЃЌдвђЪЧ____ЁЃ
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПгаЙиДпЛЏМСЕФДпЛЏЛњРэЕШЮЪЬтПЩвдДгЁАввДМДпЛЏбѕЛЏЪЕбщЁБЕУЕНвЛаЉШЯЪЖ,ФГНЬЪІЩшМЦСЫШчЭМЫљЪОзАжУ(МаГжзАжУЕШвбЪЁТд),ЦфЪЕбщВйзїЮЊ:ЯШАДЭМАВзАКУзАжУ,ЙиБеЛюШћaЁЂbЁЂc,дкЭЫПЕФжаМфВПЗжМгШШЦЌПЬ,ШЛКѓДђПЊЛюШћaЁЂbЁЂc,ЭЈЙ§ПижЦЛюШћaКЭb,ЖјгаНкзр(МфаЊад)ЕиЭЈШыЦјЬх,МДПЩдкMДІЙлВьЕНУїЯдЕФЪЕбщЯжЯѓЁЃЪдЛиД№вдЯТЮЪЬт:
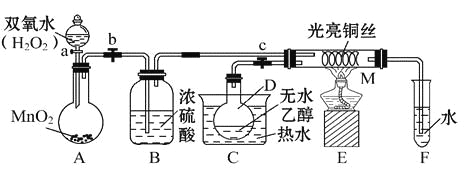
ЃЈ1ЃЉAжаЗЂЩњЗДгІЕФЛЏбЇЗНГЬЪН:__________________,BЕФзїгУ:_____________;CжаШШЫЎЕФзїгУ:_____________________ЁЃ
ЃЈ2ЃЉMДІЗЂЩњЗДгІЕФЛЏбЇЗНГЬЪНЮЊ_________________ЁЃ
ЃЈ3ЃЉДгMЙмжаПЩЙлВьЕНЕФЯжЯѓ:_______________,ДгжаПЩШЯЪЖЕНИУЪЕбщЙ§ГЬжаДпЛЏМС______(ЬюЁАВЮМгЁБЛђЁАВЛВЮМгЁБ)ЛЏбЇЗДгІ,ЛЙПЩвдШЯЪЖЕНДпЛЏМСЦ№ДпЛЏзїгУашвЊвЛЖЈЕФ_____________ЁЃ
ЃЈ4ЃЉЪЕбщНјаавЛЖЮЪБМфКѓ,ШчЙћГЗЕєОЦОЋЕЦ,ЗДгІ__________(ЬюЁАФмЁБЛђЁАВЛФмЁБ)МЬајНјаа,ЦфдвђЪЧ_____________ЁЃ
ЃЈ5ЃЉбщжЄввДМбѕЛЏВњЮяЕФЛЏбЇЗНЗЈЪЧ______________________ЁЃ
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПНсЙЙЕФбаОПЪЧгаЛњЛЏбЇзюживЊЕФбаОПСьгђЃЎФГгаЛњЮяXЃЈC12H13O6BrЃЉЗжзгжаКЌгаЖржжЙйФмЭХЃЌЦфНсЙЙМђЪНШчЯТЃКЃЈЦфжаЂёЁЂЂђЮЊЮДжЊВПЗжЕФНсЙЙЃЉЃЎ

ЮЊЭЦВтXЕФЗжзгНсЙЙЃЌНјааШчЭМзЊЛЏЃК

вбжЊЯђDЕФЫЎШмвКжаЕЮШыFeCl3ШмвКЯдзЯЩЋЃЌЖдDЕФНсЙЙНјааЙтЦзЗжЮіЃЌдкЧтКЫДХЙВеёЦзЩЯЯдЪОжЛгаСНжжаХКХЃЎMЁЂNЛЅЮЊЭЌЗжвьЙЙЬхЃЌMжаКЌгавЛИіСљдзгЛЗЃЌNФмЪЙфхЕФЫФТШЛЏЬМШмвКЭЪЩЋЃЌGФмгыNaHCO3ШмвКЗДгІЃЎЧыЛиД№ЃК
ЃЈ1ЃЉGЗжзгЫљКЌЙйФмЭХЕФУћГЦЪЧ__ЃЛ
ЃЈ2ЃЉDВЛПЩвдЗЂЩњЕФЗДгІгаЃЈбЁЬюађКХЃЉ__ЃЛ
ЂйМгГЩЗДгІ ЂкЯћШЅЗДгІ ЂлбѕЛЏЗДгІ ЂмШЁДњЗДгІ
ЃЈ3ЃЉаДГіЩЯЭМзЊЛЏжаЗДгІЂйКЭЂкЕФЛЏбЇЗНГЬЪН
ЂйB+FЁњM__ЃЛ
ЂкGЁњN__ЃЛ
ЃЈ4ЃЉвбжЊЯђXжаМгШыFeCl3ШмвКЃЌФмЗЂЩњЯдЩЋЗДгІЃЌдђXЕФНсЙЙМђЪНЪЧЃК___________ЃЌ1molИУ XгызуСПЕФNaOHШмвКзїгУЃЌзюЖрПЩЯћКФNaOH__molЃЛ
ЃЈ5ЃЉгавЛжжЛЏЙЄВњЦЗЕФжаМфЬхWгыGЛЅЮЊЭЌЗжвьЙЙЬхЃЌWЕФЗжзгжажЛКЌгаєШЛљЁЂєЧЛљКЭШЉЛљШ§жжЙйФмЭХЃЌЧвЭЌвЛИіЬМдзгЩЯВЛФмЭЌЪБСЌгаСНИієЧЛљЃЎдђWЕФЗжзгНсЙЙга__жжЃЌаДГіШЮвтвЛжжЕФНсЙЙМђЪН___________ЁЃ
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПдк25ЁцЪБ,НЋ1.0L c molЁЄL-1 CH3COOHШмвКгы0.1mol NaOHЙЬЬхЛьКЯЃЌЪЙжЎГфЗжЗДгІЁЃШЛКѓЯђИУЛьКЯШмвКжаЭЈШыHClЦјЬхЛђМгШыNaOHЙЬЬх(КіТдЬхЛ§КЭЮТЖШБфЛЏ)ЃЌШмвКpHЫцЭЈШы(ЛђМгШы)ЮяжЪЕФЮяжЪЕФСПЕФБфЛЏШчЭМЫљЪОЁЃЯТСаа№ЪіДэЮѓЕФЪЧ( )
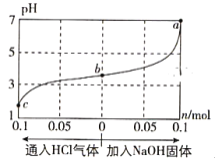
A. ЫЎЕФЕчРыГЬЖШЃКa>b>c
B. cЕуЖдгІЕФЛьКЯШмвКжаЃКc(CH3COOH)>c(Na+)>c(OH-)
C. aЕуЖдгІЕФЛьКЯШмвКжаЃКc(Na+)=c(CH3COO-)
D. ИУЮТЖШЯТЃЌCH3COOHЕФЕчРыЦНКтГЃЪ§![]()
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁП11.2LМзЭщЁЂввЭщЁЂМзШЉзщГЩЕФЛьКЯЦјЬхЃЌЭъШЋШМЩеКѓЩњГЩ15.68L CO2(ЦјЬхЬхЛ§ОљдкБъзМзДПіЯТВтЖЈ)ЃЌЛьКЯЦјЬхжаввЭщЕФЬхЛ§АйЗжКЌСПЮЊЃЈ ЃЉ
A.80ЃЅB.60ЃЅC.40ЃЅD.20ЃЅ
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПГЃЮТЯТЃЌЯрЭЌХЈЖШЕФСНжжвЛдЊЫсHXЁЂHYЗжБ№гУЭЌвЛХЈЖШЕФNaOHБъзМШмвКЕЮЖЈЃЌЕЮЖЈЧњЯпШчЭМЫљЪОЁЃЯТСаЫЕЗЈе§ШЗЕФЪЧ![]()
![]()
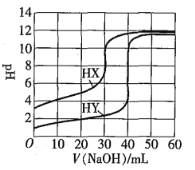
A.HXЁЂHYЦ№ЪМШмвКЬхЛ§ЯрЭЌ
B.ОљПЩгУМзЛљГШзїЕЮЖЈжИЪОМС
C.pHЯрЭЌЕФСНжжЫсШмвКжаЃК![]()
D.ЭЌХЈЖШKXгыHXЕФЛьКЯШмвКжаЃЌСЃзгХЈЖШМфДцдкЙиЯЕЪНЃК![]()
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПЯТСаЭМЯёЗжБ№БэЪОгаЙиЗДгІЕФЗДгІЙ§ГЬгыФмСПБфЛЏЕФЙиЯЕЃЌЦфжаХаЖЯе§ШЗЕФЪЧ( )
A. 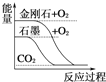 ЪЏФЋзЊБфГЩН№ИеЪЏЪЧЮќШШЗДгІ
ЪЏФЋзЊБфГЩН№ИеЪЏЪЧЮќШШЗДгІ
B. 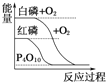 АзСзБШКьСзЮШЖЈ
АзСзБШКьСзЮШЖЈ
C. 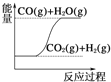 CO(g)+H2O(g)ЃНCO2(g)+H2(g) ІЄHЃО0
CO(g)+H2O(g)ЃНCO2(g)+H2(g) ІЄHЃО0
D. 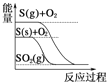 S(g)+O2(g)ЃНSO2(g) ІЄH1 S(s)+O2(g)ЃНSO2(g) ІЄH2,дђІЄH1ЃОІЄH2
S(g)+O2(g)ЃНSO2(g) ІЄH1 S(s)+O2(g)ЃНSO2(g) ІЄH2,дђІЄH1ЃОІЄH2
ВщПДД№АИКЭНтЮі>>
АйЖШжТаХ - СЗЯАВсСаБэ - ЪдЬтСаБэ
КўББЪЁЛЅСЊЭјЮЅЗЈКЭВЛСМаХЯЂОйБЈЦНЬЈ | ЭјЩЯгаКІаХЯЂОйБЈзЈЧј | ЕчаХеЉЦОйБЈзЈЧј | ЩцРњЪЗащЮожївхгаКІаХЯЂОйБЈзЈЧј | ЩцЦѓЧжШЈОйБЈзЈЧј
ЮЅЗЈКЭВЛСМаХЯЂОйБЈЕчЛАЃК027-86699610 ОйБЈгЪЯфЃК58377363@163.com