
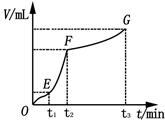
��8�֣���200mL 6mol��L-1�����м���һ�����Ĵ���CaCO3����������������ʱ��ı仯������ͼ��ʾ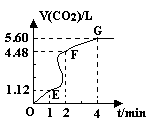 ������������ڱ�״���²ⶨ������ش��������⣺
������������ڱ�״���²ⶨ������ش��������⣺
��1����OE�εķ�Ӧ����Ϊv1��EF�εķ�Ӧ����Ϊv2��
FG�εķ�Ӧ����Ϊv3���� Ӱ��v1��v2��v3��Ӧ���ʵ������� ��
��2��Ϊ�˼���������Ӧ�����ʣ��������Һ�м����������ʣ�����Ϊ���е��� ������ĸ��
| A������ˮ | B���Ȼ��ع��� | C���Ȼ�����Һ | D��Ũ���� |
 ��3������CaCO3������Ϊ ��
��3������CaCO3������Ϊ ����1����Ӧ���¶ȣ���Ӧ���Ũ�ȣ�2��A C ��3��25g ��4��1��5mol��L-1��min-1
���������������1����OE��Ӱ�컯ѧ��Ӧ���ʵ������Ƿ�Ӧ���¶ȣ����ڸտ�ʼ��Ӧ���¶Ƚϵͣ��������ʽ��������ŷ�Ӧ�Ľ��У���Ӧ����ʹ��Һ���¶Ȳ������ߣ�������EF�εķ�Ӧ���ʴ��ӿ죬��Ӧ���Ͻ��У����ڷ�Ӧ�ﲻ�����ģ����������Ũ�Ȳ��Ͻ��͡���FG��Ӱ�컯ѧ��Ӧ���ʵ���Ҫ���ؾ��Ƿ�Ӧ���Ũ�ȡ���ʱŨ�Ƚϵ͡�������������С����Ӱ��v1��v2��v3��Ӧ���ʵ������Ƿ�Ӧ���¶Ⱥͷ�Ӧ���Ũ�ȡ���2��A�� ������ˮ��ʹ�����Ũ�Ƚ��ͣ���Ӧ���ʼ�������ȷ��B�����Ȼ��ع��壬����û�иı䷴Ӧ���Ũ�ȣ����ԶԻ�ѧ��Ӧ������Ӱ�죬����C�����Ȼ�����Һ����Һ�е�ˮ��������ϡ�����ã������Ũ�Ƚ��ͣ���Ӧ���ʼ�������ȷ��D����Ũ���ᣬ���ڷ�Ӧ���Ũ���������Ի�ѧ��Ӧ���ʴ��ӿ졣����3��n(HCl)=0��2L��6mol/L=1��2mol��n(CO2)=5��6L��22��4L/mol=0��25mol�����ݷ�Ӧ����ʽ��2HCl+ CaCO3=CaCl2+H2O+ CO2����֪��HCl��ȫ��Ӧ�ų���CO2������ڱ�״���µ����0��6mol��22��4L/mol=13��44L>5��6L��˵��HCl�������ų�����������Ӧ�ð���CaCO3�����㡣n(CaCO3) = n(CO2)= 0��25mol��m(CaCO3)= 25g��(4)��EF��n(CO2)=(4��48L��1��12L)�� 22��4L/mol= 0��15mol, ��n(HCl)= 2n(CO2) =0��3mol�� V��HCl��=(0��3mol��0��2L)��1min=1��5mol/( L��min)��
���㣺����Ӱ�컯ѧ��Ӧ���ʵ����ء���ѧ��Ӧ���ʵļ��㼰��Ӧ����һ���ǹ���ʱ�ļ����֪ʶ��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
Ǧ���仯���﹤ҵ�������ճ�������зdz��㷺����;��
��1����Ŧ�Ʒ�����Ǧ������ط�Ӧ���Ȼ�ѧ����ʽ����:
2PbS(s)+3O2(g)=2PbO(s)+2SO2(g) ��H=" a" kJ/mol
PbS(s)+2PbO(s)=3Pb(s)+SO2(g) ��H=" b" kJ��mol-1
PbS(s)+PbSO4(s)=2Pb(s)+2SO2(g) ��H=" c" kJ��mol-1
��Ӧ3PbS(s) + 6O2(g) = 3PbSO4(s) ��H="kJ" ��mol-1(�ú�a,b ,c�Ĵ���ʽ��ʾ)��
��2����ԭ����Ǧ��������ӦPbO(s)+CO(g)  Pb(s) + CO2(g) ��H���÷�Ӧ��ƽ�ⳣ���Ķ���ֵ���¶ȵĹ�ϵ���±�
Pb(s) + CO2(g) ��H���÷�Ӧ��ƽ�ⳣ���Ķ���ֵ���¶ȵĹ�ϵ���±�
| �¶� | 300 | 727 | 1227 |
| lgK | 6.17 | 2.87 | 1.24 |



�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��ʴ���
��14�֣��о�CO2��CH4�ķ�Ӧʹ֮ת��ΪCO��H2���Լ���ȼ��Σ������������ЧӦ������Ҫ�����塣
��1����֪����2CO(g)+O2��g����2CO2(g)����H����566 kJ��mol-1
��2H2(g)+O2��g����2H2O(g)����H����484kJ��mol-1
��CH4(g)+2O2(g)��CO2(g)+2H2O(g)����H����802kJ��mol-1
��CH4(g)+CO2(g)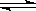 2CO(g)+2H2(g)����H������kJ��mol-1
2CO(g)+2H2(g)����H������kJ��mol-1
���������ܱ�������ͨ�����ʵ���Ũ�Ⱦ�Ϊ0.1mol����-1��CH4��CO2����һ�������·�����ӦCH4(g)+CO2(g)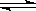 2CO(g)+2H2(g)�����CH4��ƽ��ת�������¶ȡ�ѹǿ�Ĺ�ϵ����ͼ��ʾ��
2CO(g)+2H2(g)�����CH4��ƽ��ת�������¶ȡ�ѹǿ�Ĺ�ϵ����ͼ��ʾ��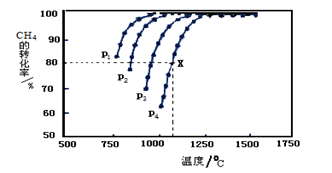
?��ͼ��֪��P1��P2�� P3��P4�ɴ�С��˳��������
?��ѹǿΪP4��1100��������£��÷�Ӧ5minʱ�ﵽƽ���X������CO��ʾ�÷�Ӧ������Ϊ �����¶��£���Ӧ��ƽ�ⳣ��Ϊ ����
��3��CO��H2�ڹ�ҵ�ϻ�����ͨ����ӦC(s)+H2O(g) 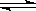 CO(g)+H2 (g)����ȡ��
CO(g)+H2 (g)����ȡ��
���ں��º����£�����ӷ�Ӧ���������ƽ�⣬���϶�ƽ���Ѵﵽ����
| A����ϵѹǿ���ٱ仯 | B��H2��CO�����ʵ���֮��Ϊ1 ��1 |
| C�����������ܶȱ��ֲ��� | D������ƽ����Է�������Ϊ15���ұ��ֲ��� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��ʴ���
��16�֣������еIJ��ֵ�Դ��O3�Ժ�ˮ��I������������O3����ͨ��NaI��Һ�н���ģ���о���
��1��O3��I��������I2�Ĺ�����3����Ӧ��ɣ�
��I��(aq)+ O3(g)==IO��(aq)+O2(g) ��H1
��IO��(aq)+H+(aq)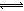 HOI(aq) ��H2
HOI(aq) ��H2
��HOI(aq)+ I��(aq)+ H+(aq)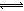 I2(aq)+H2O(l) ��H3
I2(aq)+H2O(l) ��H3
�ܷ�Ӧ�Ļ�ѧ����ʽΪ______���䷴Ӧ��H=______��
��2������Һ�д��ڻ�ѧƽ�⣺I2(aq)+I��(aq)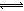 I3��(aq)����ƽ�ⳣ������ʽΪ_______��
I3��(aq)����ƽ�ⳣ������ʽΪ_______��
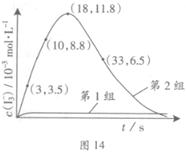
��3��Ϊ̽��Fe2+������I����Ӧ��Ӱ�죨��Ӧ��ϵ��ͼ13����ij�о�С��ⶨ����ʵ����I3��Ũ�Ⱥ���ϵpH�������ͼ14���±���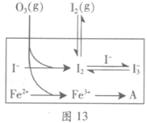
| ��� | ��Ӧ�� | ��ӦǰpH | ��Ӧ��pH |
| ��1�� | O3+ I�� | 5.2 | 11.0 |
| ��2�� | O3+ I��+ Fe2+ | 5.2 | 4.1 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��ʴ���
���ô�������Ӧ��SO2ת��ΪSO3�ǹ�ҵ����������Ĺؼ����裬��֪
SO2��g����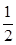 O2��g��
O2��g�� SO3��g����H����98 kJ��mol��1��
SO3��g����H����98 kJ��mol��1��
��1��ij�¶��¸÷�Ӧ��ƽ�ⳣ��K��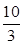 �����ڴ��¶��£���100 L�ĺ����ܱ������У�����3.0 mol
�����ڴ��¶��£���100 L�ĺ����ܱ������У�����3.0 mol
SO2(g)��16.0 mol O2(g)��3.0 mol SO3(g)����Ӧ��ʼʱv�� v�������������������������
��2��һ���¶��£���һ�����������Ϊ2 L���ܱ������г���2.0 mol SO2��1.0 molO2���ﵽƽ��������Ϊ1.6 L����SO2��ƽ��ת����Ϊ ��
��3���ڣ�2���еķ�Ӧ�ﵽƽ��ı�������������ʹSO2(g)ƽ��Ũ�ȱ�ԭ����С���� ������ĸ����
A�����¶Ⱥ�����������䣬����1.0 mol O2
B�����¶Ⱥ�������ѹǿ���䣬����1.0 mol SO3
C�����¶�
D�ƶ�����ѹ������
��4��������ͼ��ʾװ�ã��õ绯ѧԭ���������ᣬд��ͨ��O2�缫�ĵ缫��ӦʽΪ ��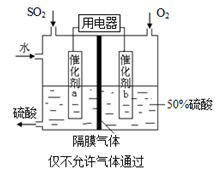
��5��Ϊ�ȶ�����������������Һ��Ũ��Ӧά�ֲ��䣬��ͨ��SO2��ˮ��������Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
��һ�������£����ݻ�Ϊ2L���ܱ������У���2mol����M��3mol����N��ϣ��������·�Ӧ��2M(g)+ 3N(g)  x Q(g)+3R(g)����2min��ƽ�⣬����2.4molR�������Q��Ũ��Ϊ0.4mol/L����
x Q(g)+3R(g)����2min��ƽ�⣬����2.4molR�������Q��Ũ��Ϊ0.4mol/L����
��1��������M����ʾ�÷�Ӧ�Ļ�ѧ��Ӧ�����Ƕ��٣�
��2����Ӧǰ���ѹǿ���Ƕ��٣�
��3��N��ת�����Ƕ��٣�
��4��ƽ��ʱ����Q��ռ���������Ϊ���٣�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
һ�������£����淴Ӧ��A2+B2 2C�ﵽƽ�⡣���ⶨ��ƽ��ʱc(A2)=0.5mol/L��c(B2)=0.1mol/L��c(C)=1.6mol/L����A2��B2��C����ʼŨ�ȷֱ�Ϊamol/L��bmol/L��cmol/L��
2C�ﵽƽ�⡣���ⶨ��ƽ��ʱc(A2)=0.5mol/L��c(B2)=0.1mol/L��c(C)=1.6mol/L����A2��B2��C����ʼŨ�ȷֱ�Ϊamol/L��bmol/L��cmol/L��
��ش�
��1��a��bӦ����Ĺ�ϵ�� ��2��a��ȡֵ��Χ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��7�֣�Ϊ���о���������Թ�������ֽ����ʵ�Ӱ�죬ijͬѧ��������ʵ�飺
| ��� | ���� | ʵ������ |
| �� | �ֱ����Թ�A��B�м���5 mL 5% H2O2��Һ��������2��1 mol��L��1 FeCl3��Һ�����Թ��о����������ݳ���ʱ�����Թ�A����ʢ��5��������ˮ���ձ��н��ݣ����Թ�B����ʢ��40��������ˮ���ձ��н��� | �Թ�A�����������ݲ����� �Թ�B�в��������������� |
| �� | ��ȡ��֧�Թֱܷ����5 mL 5% H2O2��Һ��5 mL 10% H2O2��Һ | �Թ�A��B�о�δ���Լ��������ݲ��� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com