
”¾ĢāÄæ”æ°±ŹĒ»Æ·Ź¹¤ŅµŗĶ»ł“”ÓŠ»ś»Æ¹¤µÄÖ÷ŅŖŌĮĻ”£
£Ø1£©ŗĻ³É°±·“Ó¦¹ż³ĢÖŠÓŠ¹ŲĪļÖŹµÄ»Æѧ¼ü¼üÄÜŹż¾ŻČēĻĀ±ķĖłŹ¾£ŗ
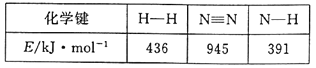
ĒėŠ“³öøĆŗĻ³É°±·“Ó¦µÄČČ»Æѧ·½³ĢŹ½___”£
£Ø2£©Ņ»¶ØĪĀ¶ČĻĀ£¬ŗĻ³É°±·“Ó¦ŌŚa”¢bĮ½ÖÖĢõ¼žĻĀ·Ö±š“ļµ½Ę½ŗā£¬H2µÄÅضČĖꏱ¼äµÄ±ä»ÆČēĶ¼ĖłŹ¾”£
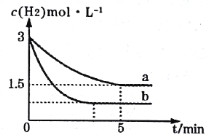
¢ŁaĢõ¼žĻĀ£¬0”«5minÄŚµÄĘ½¾ł·“Ó¦ĖŁĀŹv(N2)=___mol”¤L-1”¤min-1”£
¢ŚĻą¶Ōa¶ųŃŌ£¬bæÉÄÜøıäµÄĢõ¼žŹĒ___”£
£Ø3£©Ä³»ÆѧŠĖȤŠ”×éĻņŅ»ŗćĪĀĆܱÕČŻĘ÷ÖŠ³äČė4molN2ŗĶ12molH2Ä£ÄāŗĻ³É°±·“Ó¦£¬Ę½ŗā»ģŗĻĪļÖŠ°±ĘųµÄĢå»ż·ÖŹżÓė×ÜŃ¹Ēæ(p)µÄ¹ŲĻµČēĶ¼ĖłŹ¾”£
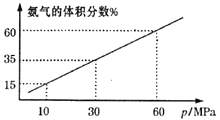
ČōĢåĻµŌŚ60MPaĻĀ“ļµ½Ę½ŗā”£H2µÄĘ½ŗā·ÖŃ¹ĪŖ___MPa”£(·ÖŃ¹=×ÜŃ¹”ĮĪļÖŹµÄĮæ·ÖŹż)£»ĮŠŹ½¼ĘĖć“ĖŹ±µÄĘ½ŗā³£ŹżKp=___”£(ÓĆĘ½ŗā·ÖŃ¹“śĢęĘ½ŗāÅØ¶Č¼ĘĖć£¬½į¹ū±£Įō2Ī»ÓŠŠ§Źż×Ö)
£Ø4£©ĻĀĮŠĶ¼Ź¾ÖŠÄܱķŹ¾ŗĻ³É°±·“Ó¦ŌŚŗćĪĀ”¢ŗćČŻĆܱÕČŻĘ÷ÖŠŌŚt1Ź±æĢŅŃ¾“ļµ½Ę½ŗāדĢ¬µÄŹĒ___”£

”¾“š°ø”æN2(g)+3H2(g)![]() 2NH3(g)”÷H=-93kJ”¤mol-1 0.1mol”¤L-1”¤min-1 Ōö“óN2ÅØ¶Č 18 0.037 b
2NH3(g)”÷H=-93kJ”¤mol-1 0.1mol”¤L-1”¤min-1 Ōö“óN2ÅØ¶Č 18 0.037 b
”¾½āĪö”æ
(1)¾É¼ü¶ĻĮŃĪüČČ£ŗ945 kJ +3”Į436 kJ=2253kJ£»ŠĀ½ØŗĻ³É·ÅČČ£ŗ6”Į391 kJ=2346kJ£»·ÅČȶą£¬»Æѧ·“Ó¦±ķĻÖĪŖ·ÅČČ£¬·Å³öČČĮæ93kJ£¬ČČ»Æѧ·½³ĢŹ½ĪŖN2(g)+3H2(g)![]() 2NH3(g)”÷H=-93kJ”¤mol-1”£
2NH3(g)”÷H=-93kJ”¤mol-1”£
£Ø2£©aĢõ¼žĻĀ£¬0”«5minÄŚµÄĘ½¾ł·“Ó¦ĖŁĀŹv(H2)=![]() £¬øł¾ŻĻµŹż±ČæÉÖŖv(N2)=0.1mol”¤L-1”¤min-1”£
£¬øł¾ŻĻµŹż±ČæÉÖŖv(N2)=0.1mol”¤L-1”¤min-1”£
a”¢bĒāĘųĘšŹ¼ÅضČĻąĶ¬£¬bµ½“ļĘ½ŗāµÄŹ±¼äĖõ¶Ģ£¬ĖµĆ÷·“Ó¦ĖŁĀŹŌö“ó£¬Ę½ŗāŹ±ĒāĘųµÄÅØ¶Č¼õŠ”£¬ĖµĆ÷Ę½ŗāÕżĻņŅĘ¶Æ£¬ĖłŅŌøıäµÄĢõ¼žŹĒŌö“óc£ØN2£©”£
£Ø3£©¢ŁČōĢåĻµŌŚ60MPaĻĀ“ļµ½Ę½ŗā£¬ĻąĶ¬ĪĀ¶ČĻĀ£¬ĘųĢåµÄĢå»ż·ÖŹżµČÓŚĘäĪļÖŹµÄĮæµÄ·ŻŹż£¬Ōņ

Ę½ŗāŹ±°±ĘųĢå»ż·ÖŹż=![]()
½āµĆx=3
H2µÄĘ½ŗā·ÖŃ¹=![]()
øł¾ŻŅŌÉĻĘ½ŗāŹż¾Ż¼ĘĖćKp![]()
£Ø4£©a.»ģŗĻĘųĢåŹ¼ÖÕ×ńŹŲÖŹĮæŹŲŗć£¬ĖłŅŌÖŹĮæ²»±ä²»ŹĒĘ½ŗāµÄ±źÖ¾£»
b.N2Ģå»ż·ÖŹż²»±äĖµĆ÷ŅŃ¾Ę½ŗā£»
c.»ģŗĻĘųĢåŹ¼ÖÕ×ńŹŲÖŹĮæŹŲŗć£¬Ģå»ż²»±äĖłŅŌĆÜ¶Č²»±ä²»ŹĒĘ½ŗāµÄ±źÖ¾£»
d.»ÆŃ§Ę½ŗā³£ŹżÖ»ÓėĪĀ¶ČÓŠ¹Ų£¬²»ŹĒĘ½ŗāµÄ±źÖ¾”£
“š°øŃ”b”£


 ĢģĢģĻņÉĻŅ»±¾ŗĆ¾ķĻµĮŠ“š°ø
ĢģĢģĻņÉĻŅ»±¾ŗĆ¾ķĻµĮŠ“š°ø Š”ѧɜ10·ÖÖÓÓ¦ÓĆĢāĻµĮŠ“š°ø
Š”ѧɜ10·ÖÖÓÓ¦ÓĆĢāĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æAŹĒŅ»ÖÖŗ¬Ģ¼”¢Ēā”¢ŃõČżÖÖŌŖĖŲµÄÓŠ»ś»ÆŗĻĪļ”£ŅŃÖŖ£ŗAÖŠĢ¼µÄÖŹĮæ·ÖŹżĪŖ44.1%£¬ĒāµÄÖŹĮæ·ÖŹżĪŖ8.82%£»AÖ»ŗ¬Ņ»ÖÖ¹ŁÄÜĶÅ£¬ĒŅĆæøöĢ¼Ō×ÓÉĻ×ī¶ąÖ»Į¬Ņ»øö¹ŁÄÜĶÅ£»AÄÜÓėŅŅĖį·¢Éśõ„»Æ·“Ó¦£¬µ«²»ÄÜŌŚĮ½øöĻąĮŚĢ¼Ō×ÓÉĻ·¢ÉśĻūČ„·“Ó¦”£Ķعż¼ĘĖćĒó£ŗ
£Ø1£©AµÄ·Ö×ÓŹ½ŹĒ_____________£ØŠ“³ö¼ĘĖć¹ż³Ģ£©£¬Ęä½į¹¹¼ņŹ½ŹĒ____________________”£
£Ø2£©Š“³öAÓėŅŅĖį·“Ó¦µÄ»Æѧ·½³ĢŹ½£ŗ________________________________________”£
£Ø3£©27.2gAŌŚæÕĘųÖŠ³ä·ÖČ¼ÉÕŹ±ÖĮÉŁŠčŅŖæÕĘų£Ø±źæöĻĀ£©¶ąÉŁL£»Éś³ÉµÄCO2ÓĆ170ml 10mol/LµÄNaOHČÜŅŗĪüŹÕ£¬·“Ó¦ŗóČÜŅŗÖŠČÜÖŹµÄ³É·ÖŹĒ____£¬ĘäĪļÖŹµÄĮæø÷ŹĒ______£»
£Ø4£©Š“³öĖłÓŠĀś×ćĻĀĮŠ3øöĢõ¼žµÄAµÄĶ¬·ÖŅģ¹¹ĢåµÄ½į¹¹¼ņŹ½”£¢ŁŹōÖ±Į“»ÆŗĻĪļ£»¢ŚÓėA¾ßÓŠĻąĶ¬µÄ¹ŁÄÜĶÅ£»¢ŪĆæøöĢ¼Ō×ÓÉĻ×ī¶ąÖ»Į¬Ņ»øö¹ŁÄÜĶÅ”£Š“³öÕāŠ©Ķ¬·ÖŅģ¹¹ĢåµÄ½į¹¹¼ņŹ½_______________________
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æŅŅĖįŅģĪģõ„ŹĒ×é³ÉĆŪ·äŠÅĻ¢ĖŲÖŹµÄ³É·ÖÖ®Ņ»£¬¾ßÓŠĻć½¶µÄĻćĪ¶£¬ŹµŃéŹŅÖʱøŅŅĖįŅģĪģõ„µÄ·“ӦװÖĆŹ¾ŅāĶ¼ŗĶÓŠ¹ŲŹż¾ŻČēĻĀ£ŗ
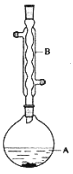
![]() +H2O
+H2O
Ļą¶ŌŌ×ÓÖŹĮæ | ĆܶČ/£Øg£®cm-3£© | ·Šµć/”ę | Ė®ÖŠČܽāŠŌ | |
ŅģĪģ“¼ | 88 | 0.8123 | 131 | Ī¢ČÜ |
ŅŅĖį | 60 | 1.0492 | 118 | ČÜ |
ŅŅĖįŅģĪģõ„ | 130 | 0.8670 | 142 | ÄŃČÜ |
ŹµŃé²½Öč£ŗ
ŌŚAÖŠ¼ÓČė4.4gµÄŅģĪģ“¼£¬6.0gµÄŅŅĖį”¢ŹżµĪÅØĮņĖįŗĶ2”«3ʬĖé“Éʬ£¬æŖŹ¼»ŗĀż¼ÓČČA£¬»ŲĮ÷50·ÖÖÓ£¬·“Ó¦ŅŗĄäÖĮŹŅĪĀŗ󣬵¹Čė·ÖŅŗĀ©¶·ÖŠ£¬·Ö±šÓĆÉŁĮæĖ®£¬±„ŗĶĢ¼ĖįĒāÄĘČÜŅŗŗĶĖ®Ļ“µÓ£¬·Ö³öµÄ²śĪļ¼ÓČėÉŁĮæĪŽĖ®ĮņĖįĆ¾¹ĢĢ壬¾²ÖĆʬæĢ£¬¹żĀĖ³żČ„ĮņĖįĆ¾¹ĢĢ壬£¬½ųŠŠÕōĮó“æ»Æ£¬ŹÕ¼Æ140”«143”ęĮó·Ö£¬µĆŅŅĖįŅģĪģõ„3.9g”£»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©×°ÖĆBµÄĆū³ĘŹĒ£ŗ___”£
£Ø2£©øĆ·“Ó¦ÖŠÅØĮņĖįµÄ×÷ÓĆ___£¬Čō·“Ó¦²śĪļĖ®·Ö×ÓÖŠÓŠĶ¬Ī»ĖŲ18O£¬Š“³öÄܱķŹ¾·“Ó¦Ē°ŗó18OĪ»ÖƵĻÆѧ·½³ĢŹ½___”£
£Ø3£©Čē¹ū¼ÓČČŅ»¶ĪŹ±¼äŗó·¢ĻÖĶü¼Ē¼ÓĖé“Éʬ£¬Ó¦øĆ²ÉČ”µÄÕżČ·²Ł×÷___”£
£Ø4£©ŌŚĻ“µÓ²Ł×÷ÖŠ£¬µŚŅ»“ĪĖ®Ļ“µÄÖ÷ŅŖÄæµÄŹĒ£ŗ___£»µŚ¶ž“ĪĖ®Ļ“µÄÖ÷ŅŖÄæµÄŹĒ£ŗ___”£
£Ø5£©±¾ŹµŃéÖŠ¼ÓČė¹żĮæŅŅĖįµÄÄæµÄŹĒ£ŗ___”£
£Ø6£©ŌŚÕōĮó²Ł×÷ÖŠ£¬ŅĒĘ÷Ń”Ōń¼°°²×°¶¼ÕżČ·µÄŹĒ£ŗ___(Ģī±źŗÅ)
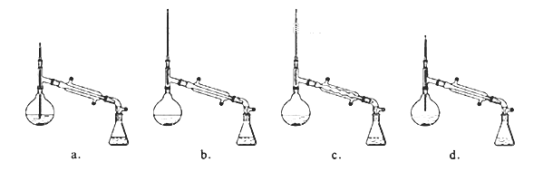
£Ø7£©±¾ŹµŃéµÄ²śĀŹŹĒ___”£
A.30ØG B.40ØG C.50ØG D.60ØG
£Ø8£©ŌŚ½ųŠŠÕōĮó²Ł×÷Ź±£¬Čō“Ó130”ęæŖŹ¼ŹÕ¼ÆĮó·Ö£¬²śĀŹĘ«___(Ģīøß»ņÕßµĶ)ŌŅņŹĒ___”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æijĪĀ¶ČŹ±£¬Ag2SO4ŌŚĖ®ČÜŅŗÖŠµÄ³ĮµķČܽāĘ½ŗāĒśĻßČēĶ¼ĖłŹ¾”£ĻĀĮŠĖµ·ØÕżČ·µÄŹĒ£Ø £©
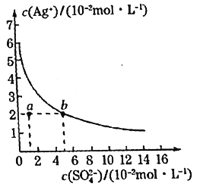
A.ŗ¬ÓŠ“óĮæSO42-µÄČÜŅŗÖŠæĻ¶Ø²»“ęŌŚAg+
B.øĆĪĀ¶ČĻĀ£¬Ag2SO4µÄČܶȻż³£Źż(Ksp)µÄŹżĮ漶ŹĒ3
C.æÉŅŌĶعżøıäĪĀ¶Č»ņ¼ÓČėĮņĖįŅų¹ĢĢåŹ¹aµćŅĘ¶Æµ½bµć
D.øĆĪĀ¶ČĻĀ£¬0.02mol”¤L-1µÄAgNO3ČÜŅŗÓė0.2mol”¤L-1µÄNa2SO4ČÜŅŗµČĢå»ż»ģŗĻ£¬²»»įÉś³É³Įµķ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æ2019ÄźÅµ±“¶ū½±ŹŚÓčĮĖŌŚæŖ·¢ļ®Ąė×Óµē³Ų·½Ćę×ö³ö×æŌ½¹±Ļ×µÄČżĪ»»Æѧ¼Ņ”£ļ®Ąė×Óµē³ŲµÄ¹ć·ŗÓ¦ÓĆŅŖĒ󓦥ķļ®µē³Ų·ĻĮĻŅŌ½ŚŌ¼×ŹŌ“”¢±£»¤»·¾³”£ļ®Ąė×Ó¶ž“Īµē³ŲÕż¼«ĀĮīÜĤÖ÷ŅŖŗ¬ÓŠLiCoO2”¢AlµČ£¬“¦ĄķøĆ·ĻĮĻµÄŅ»ÖÖ¹¤ŅÕČēĶ¼ĖłŹ¾£ŗ

£Ø1£©LiµÄŌ×Ó½į¹¹Ź¾ŅāĶ¼ĪŖ_____________£¬LiCoO2ÖŠCoµÄ»ÆŗĻ¼ŪĪŖ_______”£
£Ø2£©”°¼ī½ž”±Ź±AlČܽāµÄĄė×Ó·½³ĢŹ½ĪŖ__________________________________”£
£Ø3£©”°ĖįČÜ”±Ź±¼ÓČėH2O2µÄÄæµÄŹĒ______________________________________”£
£Ø4£©”°³ĮīÜ”±µÄĄė×Ó·½³ĢŹ½ĪŖ___________________________________________”£
£Ø5£©ÅäÖĘ100 mL 1.0 mol/L (NH4)2C2O4ČÜŅŗ£¬ŠčŅŖµÄ²£Į§ŅĒĘ÷³ö²£Į§°ō”¢ÉÕ±Ķā£¬»¹ÓŠ_____”£
£Ø6£©Č”CoC2O4¹ĢĢå4.41gŌŚæÕĘųÖŠ¼ÓČČÖĮ300 ”ę£¬µĆµ½µÄīܵÄŃõ»ÆĪļ2.41g£¬ŌņøĆ·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ__________________________________________________________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æ25”ꏱ£¬ŌŚ20 mL 0.1 mol”¤L£1Ņ»ŌŖČõĖįHAČÜŅŗÖŠµĪ¼Ó0. 1 mol”¤ L£1 NaOHČÜŅŗ£¬ČÜŅŗÖŠ1g[c(A-)/c(HA)]ÓėpH¹ŲĻµČēĶ¼ĖłŹ¾”£ĻĀĮŠĖµ·ØÕżČ·µÄŹĒ

A. Aµć¶ŌÓ¦ČÜŅŗÖŠ£ŗc(Na+)£¾c(A-)£¾c(H+)£¾c(OH-)
B. 25”ꏱ£¬HAĖįµÄµēĄė³£ŹżĪŖ1. 0”Į 10£5.3
C. Bµć¶ŌÓ¦µÄNaOHČÜŅŗĢå»żĪŖ10 mL
D. ¶ŌCµćČÜŅŗ¼ÓČČ(²»æ¼ĀĒ»Ó·¢)£¬Ōņc(A-)/[c(HA)c(OH-)]Ņ»¶ØŌö“ó
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æĄūÓĆČēĶ¼×°ÖĆ£¬æÉŅŌŌŚŹŅĪĀŗĶŅ»øö±ź×¼“óĘųŃ¹Ģõ¼žĻĀ²ā¶ØĆ¾µÄĻą¶ŌŌ×ÓÖŹĮ棬YŠĶ¹ÜÖŠŅ»²ą·ÅĆ¾£¬Ņ»²ą·ÅĻ”ĮņĖį£¬Ē抱YŠĶ¹Ü£¬½«Ļ”ĮņĖįČ«²æµ¹ČėĮķŅ»²ą£¬·“Ó¦æŖŹ¼”£
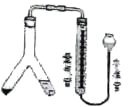
(1)ŌŚYŠĶ¹ÜÖŠĢķ¼ÓŹŌ¼ĮŹ±£¬ŅŖČ·±£________¹żĮ攣
(2)·“Ó¦Ē°ŗó¶¼ŅŖµ÷ÕūĮæĘų¹ÜŗĶĖ®×¼¹ÜŅŗĆęĻąĘ½£¬“Ó¶ų±£Ö¤_____ĻąµČ”£Čō¶ĮŹżŹ±£¬ĮæĘų¹ÜÖŠµÄŅŗĆęµĶÓŚĖ®×¼¹ÜµÄŅŗĆę£¬ŹµŃé²āµĆH2µÄĢå»ż½«______”£(Ģī”°Ę«“ó”±”°Ę«Š””±»ņ”°²»±ä”±)
(3)ČōĆ¾µÄÖŹĮæĪŖm g£¬c mol/LĮņĖįČÜŅŗV mL£¬·“Ó¦ŗóŹÕ¼Æµ½µÄĒāĘųĢå»żĪŖa mL(ŅŃÕŪĖćĪŖ±ź×¼×“æöĻĀ)£¬ŌņĆ¾µÄĻą¶ŌŌ×ÓÖŹĮæĪŖ______”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æµā»ÆĒā³£ÓĆÓŚÖʱøµāµÄ»ÆŗĻĪļŗĶÓĆ×÷ÓŠ»ś·“Ó¦µÄ»¹Ō¼Į”£µā»ÆĒā²»ĪČ¶Ø£¬Ņ×·Ö½ā£¬µā»ÆĒā¼«Ņ×ČÜÓŚĖ®£¬ĘäĖ®ČÜŅŗ³ĘĪŖĒāµāĖį£¬ĒāµāĖįŹĒĒæĖį£¬ÓŠ½ĻĒæµÄ»¹ŌŠŌ”£
(1)ĒāµāĖįČōŌŚæÕĘųÖŠ³¤ĘŚ·ÅÖĆ£¬ČÜŅŗ»į±ä³É»ĘÉ«£¬ĘäŌŅņŹĒ___£ØÓĆ»Æѧ·½³ĢŹ½Ą“±ķŹ¾£©”£
(2)ŹµŃéŹŅÖŠ³£ÓĆøÉŌļµÄŗģĮ×ŗĶµāĻą»„½Ó“„£¬¼ÓÉŁĮæĖ®Ī¢ČČ£¬¼“æÉÉś³Éµā»ÆĒāŗĶŃĒĮ×Ėį£ØH3PO3£©£¬øĆ·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ___”£
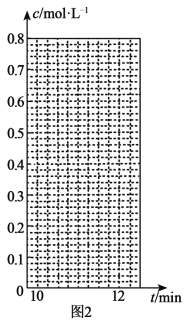
(3)ĒāĘųŗĶµāÕōĘųÄÜÖ±½Ó·“Ӧɜ³Éµā»ÆĒā£¬H2(g)£«I2(g)![]() 2HI(g) ¦¤H£¼0”£T”ꏱ£¬Ļņ1LŗćČŻĆܱÕČŻĘ÷ÖŠ³äČė0.2molH2ŗĶ0.2molI2(g)£¬5minŹ±·“Ó¦“ļµ½Ę½ŗā£¬H2”¢I2(g)ŗĶHIµÄĪļÖŹµÄĮæÅضČ(c)Ėꏱ¼ä(t)±ä»ÆµÄĒśĻßČēĶ¼lĖłŹ¾£ŗ
2HI(g) ¦¤H£¼0”£T”ꏱ£¬Ļņ1LŗćČŻĆܱÕČŻĘ÷ÖŠ³äČė0.2molH2ŗĶ0.2molI2(g)£¬5minŹ±·“Ó¦“ļµ½Ę½ŗā£¬H2”¢I2(g)ŗĶHIµÄĪļÖŹµÄĮæÅضČ(c)Ėꏱ¼ä(t)±ä»ÆµÄĒśĻßČēĶ¼lĖłŹ¾£ŗ
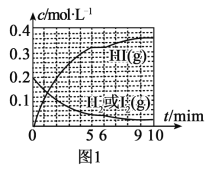
¢Ł0”«5minÄŚ£¬ŅŌHI±ķŹ¾µÄøĆ·“Ó¦ĖŁĀŹv(HI)=___”£
¢ŚT”ꏱ£¬øĆ·“Ó¦µÄĘ½ŗā³£ŹżK=___”£
¢Ū6minŹ±£¬øıäµÄĶā½ēĢõ¼žĪŖ___”£
¢Ü10minŹ±£¬±£³ÖĘäĖūĢõ¼ž²»±ä£¬ŌŁĻņČŻĘ÷ÖŠ³äČė0.1molH2”¢0.1molI2(g)”¢0.2molHI(g)£¬12minŹ±“ļµ½ŠĀĘ½ŗā”£ŌŚĶ¼2ÖŠ»³ö10”«12min£¬H2ŗĶHIµÄÅØ¶Č±ä»ÆĒśĻß___£ØĒśĻßÉĻ±źĆ÷H2ŗĶHI£©£»0”«5minŗĶ0”«2minŹ±¼ä¶Ī£¬H2µÄ×Ŗ»ÆĀŹ·Ö±šÓƦĮ1”¢¦Į2±ķŹ¾£¬Ōņ¦Įl___¦Į2£ØĢī”°£¾”±”¢”°£¼”±»ņ”°=”±£©”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
”¾ĢāÄæ”æijĀĮĶĮæóµÄÖ÷ŅŖ³É·ÖĪŖAl2O3”¤xH2O£¬»¹ŗ¬ÓŠFe2O3ŗĶSiO2ŌÓÖŹ”£³ĘČ”17.5gĀĮĶĮæóѳʷ£¬¼ÓČė200mL1.65mol/LĻ”H2SO4£¬Ē”ŗĆĶźČ«·“Ó¦£¬¹żĀĖµĆµ½ĀĖŌü0.3g£¬Č»ŗóŌŚĀĖŅŗÖŠ¼ÓČė×ćĮæNaOHČÜŅŗ£¬µĆµ½³Įµķ2.14g”££ØĢįŹ¾£ŗSiO2ŌÓ²»ÓėĻ”ĮņĖį·“Ó¦£©
£Ø1£©Š“³ö±¾ĢāÉę¼°µ½µÄČĪŅāĮ½øö·“Ó¦µÄ»Æѧ·½³ĢŹ½»ņĄė×Ó·½³ĢŹ½___”£
£Ø2£©øĆѳʷ֊£¬Fe2O3µÄĪļÖŹµÄĮæ___”£
£Ø3£©ĀĮĶĮæóѳʷ֊Al2O3µÄÖŹĮæ___”£
£Ø4£©ŹŌ¼ĘĖćѳʷ֊Al2O3”¤xH2OµÄxÖµ___”££ØŅŖĒó¼ĘĖć¹ż³Ģ£©
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com