
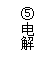
|
|
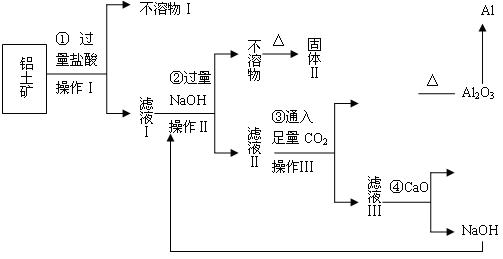
| A�����ֽⷴӦ | B��������ԭ��Ӧ | C���û���Ӧ | D���ֽⷴӦ |

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����

 ���������� ��
���������� ��| ʵ�鷽�� | ���ܵ��������Ӧ�Ľ��� |
| ȡ�����ձ��е��ϲ���Һ��װ��A��B��֧�Թ��� | |
| | |
| | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����



| ѡ�õ�����������ĸ�� | ������Լ� | ���� |
| | | ��Ӧ�����������壩 |
| C | | |
| | ����ͭ | ʹ����������ͭ��Ӧ |
| | | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
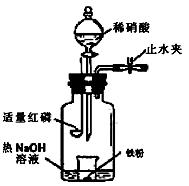
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
| ѡ�� | ���缫 | ����� | ������Ӧ | ������Ӧ |
| A | ���� | NaOH | Al-3e-��Al3+ | 2H2O+2e-��2OH-+H2�� |
| B | ���� | ϡ���� | 2Al-6e-=2Al3+ | 6H++6e-��3H2�� |
| C | ���� | Ũ���� | Cu-2e--��Cu2+ | 2NO3-+4H+-4e-��2NO2��+2H2O |
| D | ���� | ϡ���� | Cu-2e-��Cu2+ | 2NO3-+8H+��2NO��+4H2O+6e- |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
| A��ʵ�������õ��ĵζ��ܡ�����ƿ����ʹ��ǰ����Ҫ��©�� |
| B�����ʵ��������60mL ��ϡ�������Һ������ʱӦѡ��100mL����ƿ�� |
| C������ƿ�к�����������ˮ���ᵼ���������Һ��Ũ��ƫС�� |
| D����ʽ�ζ���������ˮϴ�Ӻ�װ���Ũ�ȵ�ϡ���ᣬ���õ�NaOH��Һ��Ũ�Ƚ�ƫ�� |
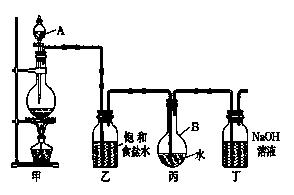
 �����ű�ʾ ����
�����ű�ʾ ���� _______________________________________________________________________
_______________________________________________________________________�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
 ��һ�ִ�����Ⱦ�ij��ȤС����̽��
��һ�ִ�����Ⱦ�ij��ȤС����̽�� �����ʼ���ɫʵ��ķ�����������·�����
�����ʼ���ɫʵ��ķ�����������·�����


 I����֤
I����֤ �������ԣ�Cװ���п�ѡ�Լ� (�����)��
�������ԣ�Cװ���п�ѡ�Լ� (�����)��| A��Ba(HCO3)2��Һ | B�������� | C����ˮ | D��Ʒ����Һ |
 �Ļ�ԭ�ԣ�Cװ���п����Լ� (������)��
�Ļ�ԭ�ԣ�Cװ���п����Լ� (������)�� �Ļ�ѧ��Ӧ����ʽΪ��___________________________________________________��
�Ļ�ѧ��Ӧ����ʽΪ��___________________________________________________���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
 �� �� ��
�� �� ��| A������ˮ�Ҵ��ӵ�ˮ����ȡ���ʵ� |
B��ϡ��Ũ ����ʱ��Ӧ��Ũ���Ỻ��ע��ʢ��ˮ����Ͳ�� ����ʱ��Ӧ��Ũ���Ỻ��ע��ʢ��ˮ����Ͳ�� |
| C�����ƣ�FeCl3��Һʱ������Һ�м�������Fe��ϡ���� |
| D���ᴿ��������������Ҵ������ȼ���ʯ�������� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com