
���� ��1�������ƫ�����µ����ʵ������ٸ������ʵ���֮�ȵ��������������Ӧ�ȣ�Ȼ��д���Ȼ�ѧ����ʽ��
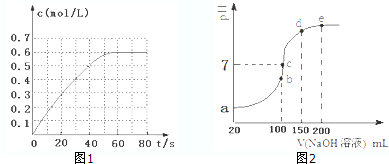
��2������ͼ��֪N2O4��ƽ��Ũ��Ϊ0.6mol/L���ﵽƽ��ʱ��N2O4��Ũ��ΪNO2��2������NO2��ƽ��Ũ��Ϊ0.3mol/L������ƽ�ⳣ������ʽ���㣻
��A��������������䣬�ݻ��㶨�����������ܶ�Ϊһ��ֵ��
B������������ɫ���䣬˵��NO2��Ũ�ȱ��ֺ㶨��
C����Ӧ2NO2��g��?N2O4��g����ѹ�㶨ʱ�������淴Ӧ������ȣ�
�۷�ӦΪ���ȷ�Ӧ�������¶ȣ�Kֵ��С�������֪��ʱ��Ũ����Q=K��298K����K��398K������Ӧ���淴Ӧ�����ƶ������V��������V���棩��
��3����a��b��c��d��e����㣬���ݷ�Ӧ���Ĺ�ϵ��b��ǡ��������H+����Һ��ֻ�У�NH4��2SO4��Na2SO4��
��c����Һ�����ԣ�����Һ���У�NH4��2SO4��Na2SO4��NH3•H2O���ֳɷ֣�
�۸��ݼ�����Һ�У�����������Ũ��Խ��ˮ�ĵ���̶�ԽС���з�����
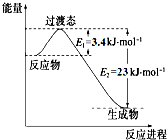
��� �⣺��1��1gȼ����ȫȼ���ͷų�������Ϊ42.5kJ����1molƫ��������ȫȼ���ͷų�������Ϊ42.5kJ��60=2550kJ�����Ȼ�ѧ����ʽΪ��C2H8N2��l��+2N2O4��l���T2CO2��g��+3N2��g��+4H2O��l����H=-2550 kJ/mol��
�ʴ�Ϊ��C2H8N2��l��+2N2O4��l��=2CO2��g��+3N2��g��+4H2O��l����H=-2550 kJ•mol-1��
��2������ͼ��֪N2O4��ƽ��Ũ��Ϊ0.6mol/L���ﵽƽ��ʱ��N2O4��Ũ��ΪNO2��2������NO2��ƽ��Ũ��Ϊ0.3mol/L����K=$\frac{c��{N}_{2}{O}_{4}��}{{c}^{2}��N{O}_{2}��}$=$\frac{0.6}{��{0.3��}^{2}}$=6.67��
�ʴ�Ϊ��6.67��
��A����Ӧ���������ȫ����̬���ʣ�������������䣬�ݻ�Ϊ2L���ֺ㶨���ɦ�=$\frac{m}{V}$��֪������ܶ�Ϊһ��ֵ�����������ܶȱ��ֲ��䲻һ������ƽ��״̬����A��ȷ��
B������������ɫ����˵��NO2��Ũ�Ȳ��䣬˵����Ӧ���ڻ�ѧƽ��״̬����B����
C����Ӧ2NO2��g��?N2O4��g����һ�����������С�ķ�Ӧ����ѹ�㶨ʱ�������淴Ӧ������ȣ�˵����Ӧ���ڻ�ѧƽ��״̬����C����
�ʴ�Ϊ��A��
�۷�ӦΪ���ȷ�Ӧ�������¶ȣ�Kֵ��С���ܱ����������Ϊ2L����˵�N2O4��Ũ��Ϊ0.6mol/L��NO2��Ũ��Ϊ0.3mol/L��Ũ����Q=$\frac{c��{N}_{2}{O}_{4}��}{{c}^{2}��N{O}_{2}��}$=$\frac{0.6}{��{0.3��}^{2}}$=6.67=K��298K����K��398K������Ӧ���淴Ӧ�����ƶ����ʣ�V��������V���棩���ʴ�Ϊ������
��3����a��b��c��d��e����㣬���ݷ�Ӧ���Ĺ�ϵ��b��ǡ��������H+����Һ��ֻ�У�NH4��2SO4��Na2SO4����Һ�з���ˮ�ⷴӦ��������NH4+���ʴ�Ϊ��NH4+��
��c����Һ�����ԣ�����Һ���У�NH4��2SO4��Na2SO4��NH3•H2O���ֳɷ֣�b��ʱc��Na+��=c��SO42-����c��ʱc��Na+����c��SO42-��������NԪ����SԪ�صĹ�ϵ�����Եó�c��SO42-����c��NH4+������Һ������Ũ�ȴ�СΪ��c��Na+����c��SO42-����c��NH4+����c��OH-��=c��H+����
�ʴ�Ϊ��c��Na+����c��SO42-����c��NH4+����c��OH-��=c��H+����
��d��e��Һ��Ϊ������Һ����Һ�е�����������������ˮ�ĵ��룬������������Һ���Խ��ˮ�ĵ������ԽС����d��ˮ�ĵ���̶ȴ���e��ˮ�ĵ��룬
�ʴ�Ϊ������
���� ���⿼���Ȼ�ѧ����ʽ����д��ƽ�ⳣ���ļ��㡢ƽ����ƶ�������Ũ�ȴ�С�Ƚϡ�����ˮ���֪ʶ�㣬��Ŀ�Ѷ��еȣ�����֪ʶ��϶࣬�ۺ��Խ�ǿ����ֿ�����ѧ���ķ�������������������Ũ�ȴ�С�Ƚϳ�������ˮ�⡢������ʵĵ������Ͽ��飬ȷ������Ũ�ȴ�СʱҪ��ϵ���غ㡢�����غ����������


 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | CCl4 | B�� | CH3COOH | C�� | CH3CH2OH | D�� | CaCO3 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
 �о������ḻ��CO2��ȫ������Ϊ��̼Դ�������ǰӦ����㷺��̼Դ��ʯ�ͺ���Ȼ��������������Ҷ���ݽߵ�Σ����ͬʱ�ֿɻ�����CO2�ۻ�������������ЧӦ��ʵ��CO2������ѭ����
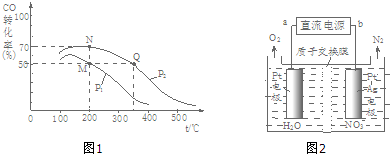
�о������ḻ��CO2��ȫ������Ϊ��̼Դ�������ǰӦ����㷺��̼Դ��ʯ�ͺ���Ȼ��������������Ҷ���ݽߵ�Σ����ͬʱ�ֿɻ�����CO2�ۻ�������������ЧӦ��ʵ��CO2������ѭ����| ʵ���� | �¶� �� | ��ʼ��/mol | ƽ����/mol | �ﵽƽ������ʱ��/min[ | ||
| CO | H2O | H2 | CO | |||
| 1 | 650 | 4 | 2 | 1.6 | 2.4 | 6 |
| 2 | 900 | 2 | 1 | 0.4 | 1.6 | 3 |
| 3 | 900 | a | b | c | d | t |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | �Ȼ�����Һ����ˮ | B�� | NaOH��Һ����ˮ | ||
| C�� | ���������Һ����ˮ | D�� | NaHCO3��Һ����ˮ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | CO2��SiO2 | B�� | Na2O2��H2O2 | C�� | NaCl��HCl | D�� | CCl4��CS2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�� ���� | ��A | ��A | ��A | IVA | VA | VIA | VIIA | O�� |
| 1 | ||||||||
| 2 | G | H | D | |||||
| 3 | B | C | E | |||||
| 4 | F | A | ||||||
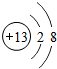 ��GԪ�ص���������ﻯѧʽCO2��H���⻯��Ļ�ѧʽNH3
��GԪ�ص���������ﻯѧʽCO2��H���⻯��Ļ�ѧʽNH3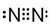 ��
��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ��ϩ�����ʽC2H4 | B�� | ��ͪ�ķ���ʽC3H6O | ||
| C�� | ���Ȼ�̼�ĵ���ʽΪ��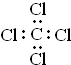 | D�� | �Ҵ��Ľṹ��ʽC2H6O |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com