
������ҵ�ǹ��ҹ�ҵ�Ļ�������ش����ұ������ʴ�����������е��й����⡣
��1����ҵ���Ȼ�ԭ��������д����CO��ԭ��������Ҫ�ɷ�ΪFe2O3���Ļ�ѧ����ʽ��
_______________________________________ ��
��2�����ڳ�ʪ�Ŀ��������ױ���ʴΪ���⣨Fe2O3•x H2O������Ӧ�Ļ�ѧ����ʽΪ��
____________________________________________ ��
��3�������п��������������⡣�ֽ�һ�������Ƭ���������У������ⱻ������
��Һ�з����Ļ��Ϸ�Ӧ�Ļ�ѧ����ʽ______________________________ ��
��4�������ĸ�װ�ÿɷ�ֹ��������ʴ_____________ ��

��5����ʵ�������У����������ı����ͭ��ֹ������ʴ��װ��ʾ��ͼ���£�
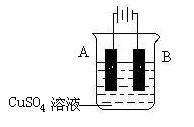
��ش�
�� A�缫��Ӧ�Ľ�����______(дԪ������)��B�缫�ĵ缫��Ӧʽ�� ��
�� �����ǰ����ͭ���缫��������ͬ�������ɺ�����ȡ��ϴ������ɡ���������
��������Ϊ5.12 g������ʱ��·��ͨ���ĵ���Ϊ__________mol��
�� �Ʋ������ͭ���ȶ�п�������ױ���ʴ�����Ҫ˵��ԭ��
��
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012�����ʡ�߶���ѧ����ĩ���Ի�ѧ�Ծ� ���ͣ������
��6�֣�������ҵ�ǹ��ҹ�ҵ�Ļ�������ش����ұ������ʴ�����������е��й����⡣
(1)��ҵ���Ȼ�ԭ��������д����CO��ԭ������(��Ҫ�ɷ�ΪFe2O3)�Ļ�ѧ����ʽ��____ ______________��
(2)���ڳ�ʪ�Ŀ��������ױ���ʴΪ����(Fe2O3��xH2O)����Ӧ�Ļ�ѧ����ʽΪ_____________ ___ _________��
(3)�����п��������������⡣�ֽ�һ�������Ƭ���������У������ⱻ��ȫ��������Һ�м��������Ļ��Ϸ�Ӧ�Ļ�ѧ����ʽΪ____________________________��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com