
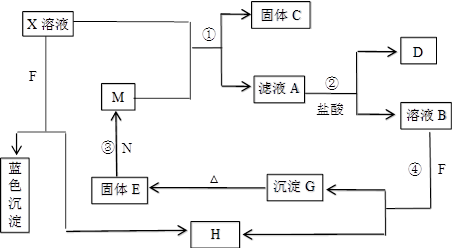
��15�֣���֪X����ѧ��ѧ�г������Σ�����Һ����ɫ�����ɻ�����F������Ԫ�ص�ԭ�Ӿ����γ�����ԭ����ͬ���Ӳ�ṹ�����ӣ�������F����ǿ�����ԡ�M�dz����Ľ������ʣ�����EΪ����ɫ���dz��õ����ϡ�N�����������Ϊ�ͻ���ϡ�D��H��Ϊ���壬��������������ֺ���ɫ�������ʵ�ת����ϵ��ͼ��ʾ(���ַ�Ӧ��������ȥ)��

(1)��M˿����ʢ��X��Һ���Թ��У���Ӧһ��ʱ����������____________________��
(2)д����Ӧ�ڵ����ӷ�Ӧ����ʽ��_________________________________________��
(3)D�Ļ�ѧʽΪ________��X�Ļ�ѧʽΪ______ __��F�ĵ���ʽΪ___ __��
(4)��ʯī���缫���1 L X��Һ�����һ��ʱ�������ȡ����ϴ�ӡ�����������缫����3.2 g���������ĵ缫��ӦʽΪ____________________________________��
 ������Һ��pHΪ________(������ǰ����Һ�������)��
������Һ��pHΪ________(������ǰ����Һ�������)��
(5)��Ӧ�ܵ����ӷ���ʽΪ_____________________________________________��
(6)����A��Һʱ����Ҫ����������________����Ŀ����________________________��
 �¿α�����Ķ�ѵ��ϵ�д�
�¿α�����Ķ�ѵ��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��8�֣���֪XΪ��ѧ��ѧ�е�һ�ֳ������Σ�FΪ����ɫ���壻M��NΪ�����Ľ�����N�����Ԫ�ص����Ӱ뾶�ǵ�������Ԫ�������Ӱ뾶��С�ġ�N��������������ͻ���ϣ����ľ���Ӳ�Ⱥܴ����ֱ�����У�HΪ���嵥�ʣ�DΪ��ɫ���壬�ڿ����л���ֺ���ɫ�������ʵ�ת����ϵ����ͼ�����ַ�Ӧ��������ȥ����
��ش��������⣺
��1��X�Ļ�ѧʽΪ ��F�ĵ���ʽΪ ��
��2����Ӧ�ڵ����ӷ���ʽΪ_______________________________________________��
��Ӧ�۵Ļ�ѧ����ʽΪ_______________________________________________��
��3������100 mL ��X����Һ�м���10 g��������M�ķ�ĩ����ֽ�����ˣ����
��10.16g����C������ҺA�����ʵ����ʵ���Ũ��Ϊ________________________��(�����������)
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ��ӱ�ʡ��ˮ��ѧ��һ��ѧ���������Ի�ѧ�Ծ� ���������� ���ͣ������
��8�֣���֪XΪ��ѧ��ѧ�е�һ�ֳ������Σ�FΪ����ɫ���壻M��NΪ�����Ľ�����N�����Ԫ�ص����Ӱ뾶�ǵ�������Ԫ�������Ӱ뾶��С�ġ�N��������������ͻ���ϣ����ľ���Ӳ�Ⱥܴ����ֱ�����У�HΪ���嵥�ʣ�DΪ��ɫ���壬�ڿ����л���ֺ���ɫ�������ʵ�ת����ϵ����ͼ�����ַ�Ӧ��������ȥ����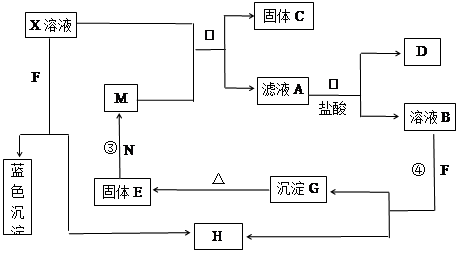
��ش��������⣺
��1��X�Ļ�ѧʽΪ ��F�ĵ���ʽΪ ��
��2����Ӧ�ڵ����ӷ���ʽΪ_______________________________________________��
��Ӧ�۵Ļ�ѧ����ʽΪ_______________________________________________��
��3������100 mL ��X����Һ�м���10 g��������M�ķ�ĩ����ֽ�����ˣ����
��10.16g����C������ҺA�����ʵ����ʵ���Ũ��Ϊ________________________��(�����������)
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2014��ӱ�ʡ��һ��ѧ���������Ի�ѧ�Ծ��������棩 ���ͣ������
��8�֣���֪XΪ��ѧ��ѧ�е�һ�ֳ������Σ�FΪ����ɫ���壻M��NΪ�����Ľ�����N�����Ԫ�ص����Ӱ뾶�ǵ�������Ԫ�������Ӱ뾶��С�ġ�N��������������ͻ���ϣ����ľ���Ӳ�Ⱥܴ����ֱ�����У�HΪ���嵥�ʣ�DΪ��ɫ���壬�ڿ����л���ֺ���ɫ�������ʵ�ת����ϵ����ͼ�����ַ�Ӧ��������ȥ����
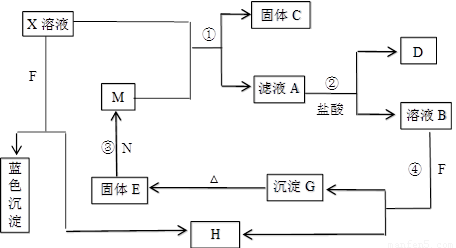
��ش��������⣺
��1��X�Ļ�ѧʽΪ ��F�ĵ���ʽΪ ��
��2����Ӧ�ڵ����ӷ���ʽΪ_______________________________________________��
��Ӧ�۵Ļ�ѧ����ʽΪ_______________________________________________��
��3������100 mL ��X����Һ�м���10 g��������M�ķ�ĩ����ֽ�����ˣ����
��10.16g����C������ҺA�����ʵ����ʵ���Ũ��Ϊ________________________��(�����������)
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2009-2010ѧ��ɽ��ʡ��Ӫ�ж�Ӫ��ʤ��һ�и������ϣ�ģ���⻯ѧ�Ծ�������2���������棩 ���ͣ������
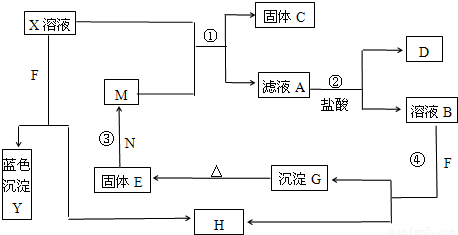
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com