
ͨ������£���������CO2�������������0.050%ʱ�����������Ե�����ЧӦ��Ϊ��С������CO2�Ի�����Ӱ�죬������������CO2��������ͬʱҲ��ǿ��CO2�������õ��о���
(1)Ŀǰ���ƹ��ó��ٽ�CO2��������̬��Һ̬֮�䣩��������������������һ�����Ի����Ļ��������� ��
(2)��ѧ��Ϊ��ȡ�����е�CO2���ѿ�������̼�����Һ��Ȼ���ٰ�CO2����Һ����ȡ����������ѧ��Ӧʹ֮��Ϊ������ȼ�ϼ״���������ͼ��
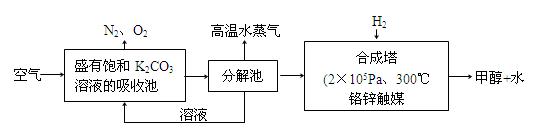
�ٷֽ���з�Ӧ�Ļ�ѧ����ʽΪ�� ��
�ںϳ����У�����4.4g CO2������H2ǡ�÷�Ӧ������̬����ų�4.947kJ��������д���÷�Ӧ���Ȼ�ѧ����ʽ�� ��
(3)ijͬѧ���ó������ⶨ������CO2����������������CaCO3��BaCO3���ܶȻ�Ksp�ֱ�Ϊ4.96��10-9��2.58��10-9������ý�����ͨ�������� ��Һ��ʵ��ʱ���ⶨ�¶ȡ�ѹǿ�Ϳ���������⣬����ⶨ ��
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
![]() �ۻ�ѧ��ѡ��ѧ�뼼����
�ۻ�ѧ��ѡ��ѧ�뼼����
ͨ������£���������CO2�������������0��050��ʱ�����������Ե�����ЧӦ��Ϊ��С������CO2�Ի�����Ӱ�죬������������CO2��������ͬʱҲ��ǿ��CO2�������õ��о���
��1��Ŀǰ���ƹ��ó��ٽ�CO2(������̬��Һ̬֮��)��������������������һ�����Ի����Ļ���������___________________��
��2����ѧ��Ϊ��ȡ�����е�CO2���ѿ�������̼�����Һ��Ȼ���ٰ�CO2����Һ����ȡ����������ѧ��Ӧʹ֮��Ϊ������ȼ�ϼ״����������£�

�ٷֽ���з�Ӧ�Ļ�ѧ����ʽΪ��_____________________________��
�ںϳ����У�����4.4g CO2������H2ǡ�÷�Ӧ������̬����ų�4.947kJ��������д���÷�Ӧ���Ȼ�ѧ����ʽ��________________________��
��3��ijͬѧ���ó������ⶨ������CO2����������������CaCO3��BaCO3���ܶȻ�K(sp)�ֱ�Ϊ4��96��10-9��2��58��10-9������ý�����ͨ��������__________��Һ��ʵ��ʱ���ⶨ�¶ȡ�ѹǿ�Ϳ���������⣬����Ҫ�ⶨ_____________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�ۻ�ѧ��ѡ��ѧ�뼼���ݣ�15�֣�
ͨ������£���������CO2�������������0��050��ʱ�����������Ե�����ЧӦ��Ϊ��С������CO2�Ի�����Ӱ�죬������������CO2��������ͬʱҲ��ǿ��CO2�������õ��о���
��1��Ŀǰ���ƹ��ó��ٽ�CO2(������̬��Һ̬֮��)��������������������һ�����Ի����Ļ���������___________________��
��2����ѧ��Ϊ��ȡ�����е�CO2���ѿ�������̼�����Һ��Ȼ���ٰ�CO2����Һ����ȡ����������ѧ��Ӧʹ֮��Ϊ������ȼ�ϼ״����������£�
�ٷֽ���з�Ӧ�Ļ�ѧ����ʽΪ��_____________________________��
�ںϳ����У�����4.4g CO2������H2ǡ�÷�Ӧ������̬����ų�4.947kJ��������д���÷�Ӧ���Ȼ�ѧ����ʽ��________________________��
��3��ijͬѧ���ó������ⶨ������CO2����������������CaCO3��BaCO3���ܶȻ�K(sp)�ֱ�Ϊ4��96��10-9��2��58��10-9������ý�����ͨ��������__________��Һ��ʵ��ʱ���ⶨ�¶ȡ�ѹǿ�Ϳ���������⣬����Ҫ�ⶨ_____________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������2010������ڶ���ģ�⿼�ԣ����ۣ���ѧ���� ���ͣ������
�ۻ�ѧ��ѡ��ѧ�뼼���ݣ�15�֣�
ͨ������£���������CO2�������������0��050��ʱ�����������Ե�����ЧӦ��Ϊ��С������CO2�Ի�����Ӱ�죬������������CO2��������ͬʱҲ��ǿ��CO2�������õ��о���
��1��Ŀǰ���ƹ��ó��ٽ�CO2(������̬��Һ̬֮��)��������������������һ�����Ի����Ļ���������___________________��
��2����ѧ��Ϊ��ȡ�����е�CO2���ѿ�������̼�����Һ��Ȼ���ٰ�CO2����Һ����ȡ����������ѧ��Ӧʹ֮��Ϊ������ȼ�ϼ״����������£�
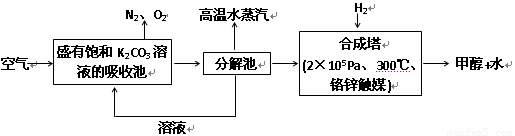
�ٷֽ���з�Ӧ�Ļ�ѧ����ʽΪ��_____________________________��
�ںϳ����У�����4.4g CO2������H2ǡ�÷�Ӧ������̬����ų�4.947kJ��������д���÷�Ӧ���Ȼ�ѧ����ʽ��________________________��
��3��ijͬѧ���ó������ⶨ������CO2����������������CaCO3��BaCO3���ܶȻ�K(sp)�ֱ�Ϊ4��96��10-9��2��58��10-9������ý�����ͨ��������__________��Һ��ʵ��ʱ���ⶨ�¶ȡ�ѹǿ�Ϳ���������⣬����Ҫ�ⶨ_____________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010-2011ѧ�꽭��ʡ��У�������������ۣ���ѧ���� ���ͣ������
ͨ������£���������CO2�������������0.050%ʱ�����������Ե�����ЧӦ��Ϊ��С������CO2�Ի�����Ӱ�죬������������CO2��������ͬʱҲ��ǿ��CO2�������õ��о���
(1)Ŀǰ���ƹ��ó��ٽ�CO2��������̬��Һ̬֮�䣩��������������������һ�����Ի����Ļ��������� ��
(2)��ѧ��Ϊ��ȡ�����е�CO2���ѿ�������̼�����Һ��Ȼ���ٰ�CO2����Һ����ȡ����������ѧ��Ӧʹ֮��Ϊ������ȼ�ϼ״���������ͼ��
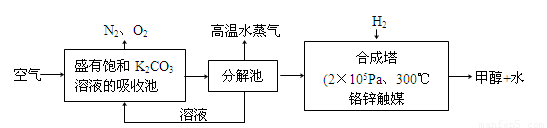
�ٷֽ���з�Ӧ�Ļ�ѧ����ʽΪ�� ��
�ںϳ����У�����4.4g CO2������H2ǡ�÷�Ӧ������̬����ų�4.947kJ��������д���÷�Ӧ���Ȼ�ѧ����ʽ�� ��
(3)ijͬѧ���ó������ⶨ������CO2����������������CaCO3��BaCO3���ܶȻ�Ksp�ֱ�Ϊ4.96��10-9��2.58��10-9������ý�����ͨ�������� ��Һ��ʵ��ʱ���ⶨ�¶ȡ�ѹǿ�Ϳ���������⣬����ⶨ ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com