
ÒŃÖȘ25ĄæʱŁș
ąÙHF(aq)Ł«OHŁ(aq)=FŁ(aq)Ł«H2O(l)ĄĄŠ€H1ŁœŁ67.7 kJĄ€molŁ1
ąÚHŁ«(aq)Ł«OHŁ(aq)=H2O(l)ĄĄŠ€H2ŁœŁ57.3 kJĄ€molŁ1
ąÛBa2Ł«(aq)Ł«SO42ĄȘ(aq)=BaSO4(s)ĄĄŠ€H3ŁŒ0
ÏÂÁĐË”·šŐęÈ·”ÄÊÇ(ĄĄĄĄ)
| AŁźHF”Ä”çÀë·œłÌÊœŒ°ÈÈЧӊŁșHF(aq)=HŁ«(aq)Ł«FŁ(aq)ĄĄŠ€HŁŸ0 |
| BŁźÔÚÇâŃő»ŻÄÆÈÜÒșÓëŃÎËá”Ä·ŽÓŠÖĐŁŹŃÎËáÁżÒ»¶šŁŹÇâŃő»ŻÄÆÈÜÒșÁżÔœ¶àŁŹÖĐșÍÈÈÔœŽó |
| CŁźŠ€H2ŁœŁ57.3 kJĄ€molŁ1ÊÇÇżËáșÍÇżŒîÔÚÏĄÈÜÒșÖĐ·ŽÓŠÉúłÉżÉÈÜŃΔÄÖĐșÍÈÈ |
| DŁźÏĄÁòËáÓëÏĄÇâŃő»Ż±”ÈÜÒș·ŽÓŠ”ÄÈÈ»ŻŃ§·œłÌÊœÎȘH2SO4(aq)Ł«Ba(OH)2(aq)=BaSO4(s)Ł«2H2O(l)Š€HŁœŁ114.6 kJĄ€molŁ1 |
 ÔĶÁżìł”Ï”ÁĐŽđ°ž
ÔĶÁżìł”Ï”ÁĐŽđ°ž
| Äꌶ | žßÖĐżÎłÌ | Äꌶ | łőÖĐżÎłÌ |
| žßÒ» | žßÒ»Ăâ·ŃżÎłÌÍÆŒöŁĄ | łőÒ» | łőÒ»Ăâ·ŃżÎłÌÍÆŒöŁĄ |
| žß¶ț | žß¶țĂâ·ŃżÎłÌÍÆŒöŁĄ | łő¶ț | łő¶țĂâ·ŃżÎłÌÍÆŒöŁĄ |
| žßÈę | žßÈęĂâ·ŃżÎłÌÍÆŒöŁĄ | łőÈę | łőÈęĂâ·ŃżÎłÌÍÆŒöŁĄ |
żÆÄżŁșžßÖĐ»ŻŃ§ ÀŽÔŽŁș ÌâĐÍŁș”„ŃĄÌâ
ÏÂÁĐčęłÌÓĐÈÈÁż·Ćłö”ÄÊÇ
| AŁź¶ÏżȘÇâÆűÖĐ”ÄHŁHŒü |
| BŁźCÓëH2O(g)·ŽÓŠ |
| CŁźBa(OH)2Ą€8H2OŸ§ÌćÓëNH4ClŸ§Ìć·ŽÓŠ |
| DŁźNaÓëH2O·ŽÓŠ |
Č鿎Žđ°žșÍœâÎö>>
żÆÄżŁșžßÖĐ»ŻŃ§ ÀŽÔŽŁș ÌâĐÍŁș”„ŃĄÌâ
ÏÂÁĐ͌ʟÓë¶ÔÓŠ”ÄĐđÊöÏà·û”ÄÊÇ
| AŁźÍŒ1±íÊŸÏòÄłÁòËáșÍÁòËáÂÁ»ìșÏÈÜÒșÖĐŒÓÈëNaOHÈÜÒșŁŹłÁ”íÖÊÁżÓëŒÓÈëNaOHÈÜÒșÌć»ę”ÄčŰÏ” |
| BŁźÍŒ2±íÊŸ25ĄæʱŁŹÓĂ0.1mol?L-1ŃÎËá”ζš20mL0.1mol?L-1NaOHÈÜÒș”ÄpHËæŒÓÈëŃÎËáÌć»ę”ı仯 |
| CŁźÍŒ3±íÊŸtĄæÊ±ÏĄÊͱùŽŚËáčęłÌÖĐÈÜÒș”Œ”çĐԔı仯 |
| DŁźžùŸĘÍŒ4żÉĆжÏÄłżÉÄæ·ŽÓŠ”ÄŐę·ŽÓŠÊÇÎüÈÈ·ŽÓŠ |
Č鿎Žđ°žșÍœâÎö>>
żÆÄżŁșžßÖĐ»ŻŃ§ ÀŽÔŽŁș ÌâĐÍŁș”„ŃĄÌâ
žÇË趚ÂÉÈÏÎȘŁșČ»čÜ»ŻŃ§čęłÌÊÇÒ»Čœ»ò·ÖÎȘÊęČœÍêłÉŁŹŐâžöčęłÌ”ÄÈÈЧӊÊÇÏàÍŹ”ÄĄŁ
ÒŃÖȘŁșH2OŁšgŁ©Łœ H2OŁšlŁ© ĄśH1 ŁœŁQ1 kJĄ€molŁ1ŁšQ1ŁŸ0Ł©
C2H5OHŁšgŁ©Łœ C2H5OHŁšlŁ© ĄśH2 ŁœŁQ2 kJĄ€molŁ1ŁšQ2ŁŸ0Ł©
C2H5OHŁšgŁ©Ł«3O2ŁšgŁ©Łœ2CO2ŁšgŁ©Ł«3H2OŁšgŁ© ĄśH3 Łœ ŁQ3 kJĄ€molŁ1ŁšQ3ŁŸ0Ł©ÈôÊč23gÒșÌŹÒÒŽŒÍêÈ«ÈŒÉŐŁŹŚîșó»ÖžŽ”œÊÒÎÂŁŹÔò·Ćłö”ÄÈÈÁżÎȘŁškJŁ©
| AŁźQ1Ł« Q2Ł«Q3 | BŁź0.5ŁšQ1Ł«Q2Ł«Q3 Ł© |
| CŁź0.5 Q1Ł1.5 Q2Ł«0.5Q3 | DŁź1.5 Q1Ł0.5 Q2Ł«0.5Q3 |
Č鿎Žđ°žșÍœâÎö>>
żÆÄżŁșžßÖĐ»ŻŃ§ ÀŽÔŽŁș ÌâĐÍŁș”„ŃĄÌâ
ÒŃÖȘ¶ÏżȘ»òĐÎłÉ1 mol»ŻŃ§ŒüÎüÊŐ»ò·Ćłö”ÄÄÜÁżłÆÎȘ»ŻŃ§Œü”ÄŒüÄÜŁŹÈçHĄȘHŒü”ÄŒüÄÜÎȘ436 kJĄ€molŁ1ŁŹNĄÔNŒü”ÄŒüÄÜÎȘ945 kJĄ€molŁ1ŁŹNĄȘHŒü”ÄŒüÄÜÎȘ391 kJĄ€molŁ1ĄŁÔòÏÂÁĐÓĐčŰč€Ò”șÏłÉ°±·ŽÓŠ”ÄÈÈ»ŻŃ§·œłÌÊœŐęÈ·”ÄÊÇ (ĄĄĄĄ)
AŁźN2(g)Ł«3H2(g)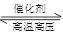 2NH3(g)ĄĄŠ€HŁœŁ93 kJĄ€molŁ1 2NH3(g)ĄĄŠ€HŁœŁ93 kJĄ€molŁ1 |
BŁźN2(g)Ł«3H2(g)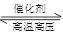 2NH3(g)ĄĄŠ€HŁœŁ«1471 kJĄ€molŁ1 2NH3(g)ĄĄŠ€HŁœŁ«1471 kJĄ€molŁ1 |
CŁźN2(g)Ł«3H2(g)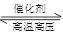 2NH3(g)ĄĄŠ€HŁœŁ«93 kJĄ€molŁ1 2NH3(g)ĄĄŠ€HŁœŁ«93 kJĄ€molŁ1 |
DŁźN2(g)Ł«3H2(g)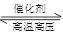 2NH3(g)ĄĄŠ€HŁœŁ1471 kJĄ€molŁ1 2NH3(g)ĄĄŠ€HŁœŁ1471 kJĄ€molŁ1 |
Č鿎Žđ°žșÍœâÎö>>
żÆÄżŁșžßÖĐ»ŻŃ§ ÀŽÔŽŁș ÌâĐÍŁș”„ŃĄÌâ
»ŻŃ§·ŽÓŠA2Ł«B2=2AB”ÄÄÜÁż±ä»ŻÈçÍŒËùÊŸŁŹÔòÏÂÁĐË”·šŐęÈ·”ÄÊÇŁš Ł©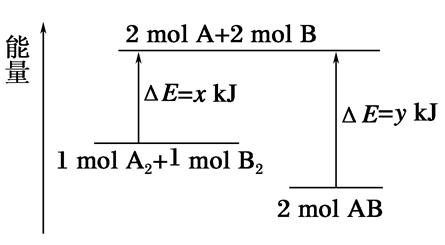
| AŁźžĂ·ŽÓŠÊÇÎüÈÈ·ŽÓŠ |
| BŁź¶ÏÁŃ1mol AĄȘAŒüșÍ1mol BĄȘBŒüÄÜ·ĆłöxkJ”ÄÄÜÁż |
| CŁź¶ÏÁŃ2mol AĄȘBŒüĐèÒȘÎüÊŐykJ”ÄÄÜÁż |
| DŁź2mol AB”ÄŚÜÄÜÁżžßÓÚ1 mol A2șÍ1mol B2”ÄŚÜÄÜÁż |
Č鿎Žđ°žșÍœâÎö>>
żÆÄżŁșžßÖĐ»ŻŃ§ ÀŽÔŽŁș ÌâĐÍŁș”„ŃĄÌâ
žùŸĘ”âÓëÇâÆű·ŽÓŠ”ÄÈÈ»ŻŃ§·œłÌÊœ
(ąĄ)I2(g)Ł«H2(g)  2HI(g)ĄĄŠ€HŁœŁ9Łź48 kJĄ€molŁ1
2HI(g)ĄĄŠ€HŁœŁ9Łź48 kJĄ€molŁ1
(ąą)I2(s)Ł«H2(g)  2HI(g)ĄĄŠ€HŁœŁ«26Łź48 kJĄ€molŁ1
2HI(g)ĄĄŠ€HŁœŁ«26Łź48 kJĄ€molŁ1
ÏÂÁĐĆжÏŐęÈ·”ÄÊÇŁš Ł©
| AŁź1 mol I2(s)ÖĐÍšÈë2 g H2(g)ŁŹ·ŽÓŠÎüÈÈ26Łź48 kJ |
| BŁź1 molčÌÌŹ”âÓë1 molÆűÌŹ”âËùșŹ”ÄÄÜÁżÏàČî17Łź00 kJ |
| CŁź·ŽÓŠ(ąą)”Ä·ŽÓŠÎïŚÜÄÜÁż±È·ŽÓŠ(ąĄ)”Ä·ŽÓŠÎïŚÜÄÜÁż”Í |
| DŁź·ŽÓŠ(ąĄ)·Ćłö”ÄÈÈÁż¶àŁŹËùÒÔČúÎï”ÄÄÜÁż”ÍŁŹ±È·ŽÓŠ(ąą)”ÄČúÎïžüÎȶš |
Č鿎Žđ°žșÍœâÎö>>
żÆÄżŁșžßÖĐ»ŻŃ§ ÀŽÔŽŁș ÌâĐÍŁș”„ŃĄÌâ
ÒŃÖȘŒŚÍéÈŒÉŐÉúłÉ¶țŃő»ŻÌŒșÍÒșÌŹËź·Ćłö”ÄÈÈÁżÎȘ55.625 kJĄ€g-1ĄŁÏÂÁĐÈÈ»ŻŃ§·œłÌÊœÖĐČ»ŐęÈ·”ÄÊÇ(ĄĄĄĄ)
AŁźCH4(g)+2O2(g) CO2(g)+2H2O(l) Š€H ="-890" kJ/mol CO2(g)+2H2O(l) Š€H ="-890" kJ/mol |
BŁź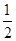 CH4(g)+O2(g) CH4(g)+O2(g) 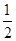 CO2(g)+H2O(l) Š€H="-445" kJ/mol CO2(g)+H2O(l) Š€H="-445" kJ/mol |
CŁź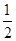 CO2(g)+H2O(l) CO2(g)+H2O(l) 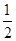 CH4(g)+O2(g) Š€H="+445" kJ/mol CH4(g)+O2(g) Š€H="+445" kJ/mol |
DŁźCH4(g)+2O2(g) CO2(g)+2H2O(l) Š€H="-55.625" kJ/mol CO2(g)+2H2O(l) Š€H="-55.625" kJ/mol |
Č鿎Žđ°žșÍœâÎö>>
żÆÄżŁșžßÖĐ»ŻŃ§ ÀŽÔŽŁș ÌâĐÍŁș”„ŃĄÌâ
žùŸĘÒÔÏÂ3žöÈÈ»ŻŃ§·œłÌÊœŁș
2H2S(g)Ł«3O2(g)=2SO2(g)Ł«2H2O(l)ĄĄ Š€HŁœŁa kJĄ€molŁ1
2H2S(g)Ł«O2(g)=2S(s)Ł«2H2O(l)ĄĄ Š€HŁœŁb kJĄ€molŁ1
2H2S(g)Ł«O2(g)=2S(s)Ł«2H2O(g) Š€HŁœŁc kJĄ€molŁ1(aĄąbĄącŸùÎȘŐęÊę)
¶ÔaĄąbĄącÈęŐߎóĐĄčŰÏ””ÄĆжÏŐęÈ·”ÄÊÇ(ĄĄĄĄ)
| AŁźa>b>c | BŁźa>c>b | CŁźc>b>a | DŁźb>a>c |
Č鿎Žđ°žșÍœâÎö>>
°Ù¶ÈÖÂĐĆ - Á·Ï°ČáÁбí - ÊÔÌâÁбí
șț±±ÊĄ»„ÁȘÍű΄·šșÍČ»ÁŒĐĆÏąŸÙ±šÆœÌš | ÍűÉÏÓĐșŠĐĆÏąŸÙ±šŚšÇű | ”çĐĆŐ©ÆŸÙ±šŚšÇű | ÉæÀúÊ·ĐéÎȚÖśÒćÓĐșŠĐĆÏąŸÙ±šŚšÇű | ÉæÆóÇÖÈšŸÙ±šŚšÇű
΄·šșÍČ»ÁŒĐĆÏąŸÙ±š”ç»°Łș027-86699610 ŸÙ±šÓÊÏäŁș58377363@163.com