
| A��SO2��O3��PO43-��ClO4-��Ϊ�ȵ����� |
| B���������ʧȥһ��H�����γ�CH3-����̼ԭ�ӵ��ӻ����ͷ����˸ı� |
| C��Tiԭ�ӵĺ�������Ų�ʽΪ[Ar]3d34s2 |
| D��CS2��H2O��C2H2����ֱ���η��� |



 g/cm3��BN�۵�Ƚ��ʯ�͵�ԭ���뻯ѧ����ǿ���йأ�B��N���ļ�������C��C���ļ���������������ṹ��������ʯī���ƣ����B��Nԭ�Ӿ�����sp2�ӻ�����ƽ��ṹʾ��ͼ����ԭ�ӽ�����֡�
g/cm3��BN�۵�Ƚ��ʯ�͵�ԭ���뻯ѧ����ǿ���йأ�B��N���ļ�������C��C���ļ���������������ṹ��������ʯī���ƣ����B��Nԭ�Ӿ�����sp2�ӻ�����ƽ��ṹʾ��ͼ����ԭ�ӽ�����֡�


| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������


�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
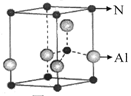


�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����Һ����AgNO3������������ɻ�ɫ���� |
| B����Һ����NaCl��������ⷢ����ѧ��Ӧ������ɫ�仯 |
| C��������������ԭ������������Ƥ�����ڳ�����֬�У�����ɫ�仯 |
| D��������������ԭ�������뺹Һ�еĵ�������ɫ�仯 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������


�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������


�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com