
���� ��1������ȼ���ȵĶ����𣬱�ʾȼ���ȵ��Ȼ�ѧ����ʽ�п�ȼ��Ϊ1mol������Ϊ�ȶ����������1g�Ҵ���ȫȼ������CO2��Һ̬ˮʱ����23kJ�����ȼ���ȵĶ���������1mol�Ҵ���ȫȼ�շų���������Ȼ��д���Ȼ�ѧ����ʽ��
��2������ÿ����23��NO2��Ҫ����16.95kJ����������2molNO2ȼ�շų�����������ϻ�ѧ����ʽ��д�Ȼ�ѧ����ʽ��ע�����ʵľۼ�״̬�ͷ�Ӧ�ȵĵ�λ�����⣮
��� �⣺��1��ȼ������ָ����25�桢101KPaʱ��1mol��������ȫȼ�������ȶ���������ų���������
1g�Ҵ���ȫȼ������CO2��Һ̬ˮʱ����23kJ����1mol�Ҵ�������Ϊ46g����ȫȼ�������ȶ���������ų�������Ϊ��46��23kJ=1058KJ��
��ȼ���ȵ��Ȼ�ѧ����ʽΪ��C2H5OH��l��+3O2��g���T2CO2��g��+3H2O��l����H=-1058 kJ•mol-1��
�ʴ�Ϊ��C2H5OH��l��+3O2��g���T2CO2��g��+3H2O��l����H=-10588 kJ•mol-1��
��2��������N2��O2��ȫ��Ӧ��ÿ����23��NO2��Ҫ����16.95 kJ����������ÿ����92��NO2��Ҫ����67.8kJ���������Ȼ�ѧ����ʽΪ��N2��g��+2O2��g��=2NO2��g����H=+67.8kJ/mol��
�ʴ�Ϊ��N2��g��+2O2��g���T2NO2��g����H=+67.8kJ/mol��
���� ���⿼����ȼ���ȵĸ����Լ�ȼ���ȵ��Ȼ�ѧ����ʽ����д��ע��ȼ���ȵ��Ȼ�ѧ����ʽ�п�ȼ������ʵ���Ϊ1mol������������ȶ����������Ŀ�ϼ������ڿ���ѧ���ķ��������ͼ���������


 ѧ���쳵�����ּ��������ҵ�½����������ϵ�д�
ѧ���쳵�����ּ��������ҵ�½����������ϵ�д� �����ѧСѧ�꼶�νӵ������㽭��ѧ������ϵ�д�
�����ѧСѧ�꼶�νӵ������㽭��ѧ������ϵ�д� Сѧ�����ҵ���ϴ�ѧ������ϵ�д�
Сѧ�����ҵ���ϴ�ѧ������ϵ�д� ���Ž�����ٰθ��νӹ㶫���������ϵ�д�
���Ž�����ٰθ��νӹ㶫���������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
| T��K�� | 938 | 1100 |
| K | 0.68 | 0.40 |
| ���� | CH3COONa | NaHCO3 | Na2CO3 | NaClO | NaCN | C6H5ONa |
| pH | 8.8 | 9.7 | 11.6 | 10.3 | 11.1 | 11.3 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
 ijѧ��������֪���ʵ���Ũ�ȵ��������ⶨδ֪���ʵ���Ũ�ȵ�NaOH��Һʱ��ѡ�������ָʾ��������д���пհף�
ijѧ��������֪���ʵ���Ũ�ȵ��������ⶨδ֪���ʵ���Ũ�ȵ�NaOH��Һʱ��ѡ�������ָʾ��������д���пհף�| �ζ����� | ����NaOH��Һ�����/mL | 0.100 0mol/L��������/mL | ||
| �ζ�ǰ�̶� | �ζ���̶� | ��Һ���/mL | ||
| ��һ�� | 25.00 | 0.10 | 26.11 | 26.01 |
| �ڶ��� | 25.00 | 1.56 | 30.30 | 28.74 |
| ������ | 25.00 | 0.32 | 26.31 | 25.99 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
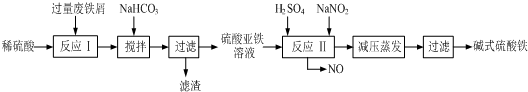
| ������ | Fe��OH��3 | Fe��OH��2 | Al��OH��3 |
| ��ʼ���� | 2.3 | 7.5 | 3.4 |
| ��ȫ���� | 3.2 | 9.7 | 4.4 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | �ܱ������У�9.6g ����� 11.2g ���ۻ�ϼ������������� 17.6g ʱ���ų� 19.12KJ ��������Fe��s��+S��s���TFeS��s����H=-95.6KJ?mol-1 | |
| B�� | ϡ������ 0.1mol?L-1NaOH ��Һ��Ӧ��H+��aq��+OH-��aq���TH2O��l����H=-57.3KJ?mol-1 | |
| C�� | ��֪��1molH2 ��ȫȼ������Һ̬ˮ���ų�������Ϊ 285.5KJ����ˮ�ֽ���Ȼ�ѧ����ʽΪ��2H2O��l���T2H2��g��+O2��g����H=-285.5KJ?mol-1 | |
| D�� | ��֪ 2C��s��+O2��g���T2CO��g����H=-221KJ?mol-1�����֪ C ��ȼ���ȡ�H=-110.5KJ?mol-1 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | 0.40mol | B�� | ��0.20mol | ||
| C�� | ����0.20mol | D�� | ��0.40mol������0.20mol |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | �ؿ��н���Ԫ�صĺ��� | B�� | ��������չ�� | ||
| C�� | �����ĵ����� | D�� | ������Լ�ұ�������׳̶� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com