




| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��11�֣���ⷨ���ռ��ԭ���DZ���ʳ��ˮ�����ڴ����к�����ɳ��Ca2+��Mg2+��Fe3+��SO42-���ʣ������ϵ��Ҫ����˱��뾭�����ơ�ijУʵ��С�龫�ƴ���ˮ��ʵ��������£�

��ش��������⣺
��1������a��������____________��
��2���ڢ��У���������Լ������������ִ����ij��������Լ���Ϊ________��Һ���Լ���Ϊ ������FΪ ��
��3���ڵڢ����У������Լ���ֱ����Һ�����Ա仯ʱ��д���˹��̵Ļ�ѧ����ʽ �� _ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010������ʡ�������и�һ��ѧ��10���¿�����ѧ�� ���ͣ������
��11�֣���ⷨ���ռ��ԭ���DZ���ʳ��ˮ�����ڴ����к�����ɳ��Ca2+��Mg2+��Fe3+��SO42-���ʣ������ϵ��Ҫ����˱��뾭�����ơ�ijУʵ��С�龫�ƴ���ˮ��ʵ��������£�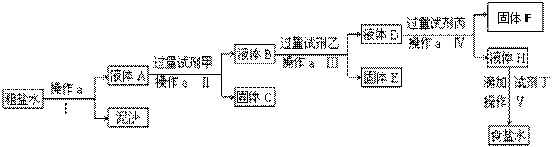
��ش��������⣺
��1������a��������____________��
��2���ڢ��У���������Լ������������ִ����ij��������Լ���Ϊ________��Һ���Լ���Ϊ ������FΪ ��
��3���ڵڢ����У������Լ���ֱ����Һ�����Ա仯ʱ��д���˹��̵Ļ�ѧ����ʽ �� _ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010������ʡ��һ��ѧ��10���¿�����ѧ�� ���ͣ������
��11�֣���ⷨ���ռ��ԭ���DZ���ʳ��ˮ�����ڴ����к�����ɳ��Ca2+��Mg2+��Fe3+��SO42- ���ʣ������ϵ��Ҫ����˱��뾭�����ơ�ijУʵ��С�龫�ƴ���ˮ��ʵ��������£�
��ش��������⣺
��1������a��������____________��
��2���ڢ��У���������Լ������������ִ����ij��������Լ���Ϊ________��Һ���Լ���Ϊ ������FΪ ��
��3���ڵڢ����У������Լ���ֱ����Һ�����Ա仯ʱ��д���˹��̵Ļ�ѧ����ʽ �� _ ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com