

| A������ʳ��ˮ | B����ʯ�� | C��Ũ���� | D��ˮ |
| ʵ�鲽�� | ʵ������ | ʵ����� |
| �� | | ��ҺX�к���Na+ |
| �� | | ��ҺX�к���Cl�� |
 3H2��+NCl3��2�֣���ƽ��������״̬��ȫ��1�֣�
3H2��+NCl3��2�֣���ƽ��������״̬��ȫ��1�֣� | ʵ�鲽�� | ʵ������ | ʵ����� |
| ������˿պȡ��ҺX���ھƾ��ƻ��������գ�1�֣� | ����ʻ�ɫ��1�֣� | ��ҺX�к���Na+ |
| ��ȡ������ҺX����������ϡHNO3��1�֣����ٵμ����� AgNO3��Һ��1�֣� | ���ְ�ɫ������1�֣� | ��ҺX�к���Cl�� |
 NCl3+3H2������2������������ˮ����������Ҳ������ˮ����ʳ��ˮ�����ܳ�ȥ������Ҳ�ܳ�ȥ�������ȣ���AD��������ʯ�ҵ���Ҫ�ɷ����������ƺ������ƣ��������������ƺ������ƾ�����Ӧ�����Ƕ����������ʯ���ܷ�Ӧ����B����������Ũ�����ܷ�Ӧ����������炙�������泥����������Ȳ�����Ũ���ᷴӦ����C��ȷ����3���ٶ������Ⱦ���ǿ�����ԣ����Խ�KI����ΪI2������Һ��ܿ������ղ���Ķ����������塢ʹ��ƿ����ѹǿ��ȣ���Ϊ��������������ˮ��ƽ����ƿ����ѹ����ʹ������������˳��ͨ�룻�����ڶ������ȡ��������ܽ�KI����ΪI2����������ԭΪ�����ӣ����ݻ��ϼ�����������ɡ�ԭ���غ�ԭ���ɵã�2ClO2+10I��+8H+=2Cl��+5I2+4H2O���۵ζ�ǰ��ƿ����Һ�к���I2�͵��ۣ��������ⵥ�ʱ����������һ�ε���ʱ����Һ����ɫ��Ϊ��ɫ���Ұ�����ڲ��ָ�Ϊ��ɫ�����ﵽ�ζ��յ㣻������c?V=n�����������ƻ������������ӵ����ʵ���Ϊc��V��10��3mol������I2+2S2O32��=2I��+S4O62����I2��S2O32�������ʵ���֮�ȵ���ϵ��֮�ȿ���I2�����ʵ���Ϊc��V��10��3mol��1/2��Ȼ����2ClO2+10I��+8H+=2Cl��+5I2+4H2O��ClO2��I2�����ʵ���֮�ȵ���ϵ��֮�ȿ���ClO2�����ʵ���Ϊc��V��10��3mol��1/2��2/5�������nM=m����������ȵ�����Ϊc��V��10��3mol��1/2��2/5��67.5g/mol=1.35cV��10��2g����4��NaClO2��NCl3��H2O������Ӧ�����ݻ��ϼ���������ԭ���غ���ƽ�ɵã�6NaClO2+NCl3+3H2O=6ClO2��+NH3��+X�������ơ��ȡ���ԭ�Ӹ����غ�ɵ�XΪ3NaCl��3NaOH������X��Һ����������ʱ������˿պȡ��ҺX���ھƾ��ƻ��������գ�����ʻ�ɫ������X��Һ����������ʱ���������ų����������Ӷ������Ӽ���ĸ��ţ���ȡ������ҺX����������ϡHNO3���ٵμ�����AgNO3��Һ�����ְ�ɫ������˵��X��Һ�к���Cl����
NCl3+3H2������2������������ˮ����������Ҳ������ˮ����ʳ��ˮ�����ܳ�ȥ������Ҳ�ܳ�ȥ�������ȣ���AD��������ʯ�ҵ���Ҫ�ɷ����������ƺ������ƣ��������������ƺ������ƾ�����Ӧ�����Ƕ����������ʯ���ܷ�Ӧ����B����������Ũ�����ܷ�Ӧ����������炙�������泥����������Ȳ�����Ũ���ᷴӦ����C��ȷ����3���ٶ������Ⱦ���ǿ�����ԣ����Խ�KI����ΪI2������Һ��ܿ������ղ���Ķ����������塢ʹ��ƿ����ѹǿ��ȣ���Ϊ��������������ˮ��ƽ����ƿ����ѹ����ʹ������������˳��ͨ�룻�����ڶ������ȡ��������ܽ�KI����ΪI2����������ԭΪ�����ӣ����ݻ��ϼ�����������ɡ�ԭ���غ�ԭ���ɵã�2ClO2+10I��+8H+=2Cl��+5I2+4H2O���۵ζ�ǰ��ƿ����Һ�к���I2�͵��ۣ��������ⵥ�ʱ����������һ�ε���ʱ����Һ����ɫ��Ϊ��ɫ���Ұ�����ڲ��ָ�Ϊ��ɫ�����ﵽ�ζ��յ㣻������c?V=n�����������ƻ������������ӵ����ʵ���Ϊc��V��10��3mol������I2+2S2O32��=2I��+S4O62����I2��S2O32�������ʵ���֮�ȵ���ϵ��֮�ȿ���I2�����ʵ���Ϊc��V��10��3mol��1/2��Ȼ����2ClO2+10I��+8H+=2Cl��+5I2+4H2O��ClO2��I2�����ʵ���֮�ȵ���ϵ��֮�ȿ���ClO2�����ʵ���Ϊc��V��10��3mol��1/2��2/5�������nM=m����������ȵ�����Ϊc��V��10��3mol��1/2��2/5��67.5g/mol=1.35cV��10��2g����4��NaClO2��NCl3��H2O������Ӧ�����ݻ��ϼ���������ԭ���غ���ƽ�ɵã�6NaClO2+NCl3+3H2O=6ClO2��+NH3��+X�������ơ��ȡ���ԭ�Ӹ����غ�ɵ�XΪ3NaCl��3NaOH������X��Һ����������ʱ������˿պȡ��ҺX���ھƾ��ƻ��������գ�����ʻ�ɫ������X��Һ����������ʱ���������ų����������Ӷ������Ӽ���ĸ��ţ���ȡ������ҺX����������ϡHNO3���ٵμ�����AgNO3��Һ�����ְ�ɫ������˵��X��Һ�к���Cl����

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A���ӵ�ˮ����ȡ���ʵ�ʱ����������ˮ�Ҵ�����CCl4 |
| B���������Ƶ�Cu(OH)2����Һ���������д��ڵĸ��� |
| C��ֽ����ʵ���У��뽫��ֽ�ϵ����������չ������ |
| D��ʵ�������ᴿ��������������Ҵ����ɲ����ȼ���ʯ�ң����˺�������ķ��� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��C2H6�л��е�C2H4�����Խ����������ͨ����ˮ��Ȼ���ü�ʯ�Ҹ��� |
| B��CaCO3�л��е�����NaHCO3�������ü��ȵķ�����ȥ |
| C����ϩ�л��е�SO2���壬����ͨ�����Ը��������Һ��ȥ |
| D��H2S�л��е�ˮ������������Ũ�����ȥ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
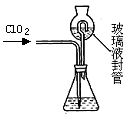
| A����ͼ1װ�ó�ȥC12��������HCl |
| B����ͼ2װ������NH4Cl������Һ��ȡNH4Cl���� |
| C����ͼ3װ����ȡ������CO2���� |
| D����ͼ4װ�÷���CCl4��ȡ��ˮ����л����ˮ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
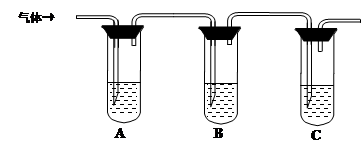
| �����Լ� | Ԥ������ͽ��� |
| �Թ�A�м������� �� �Թ�B�м�1%Ʒ����Һ�� �Թ�C�мӢ� �� | ��A����Һ����ɫ��B����Һ����ɫ��C����Һ����ǡ�������Һ���ֱ��ʣ� �� ������Һδ���ʣ� �� ������Һ��ȫ���ʡ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
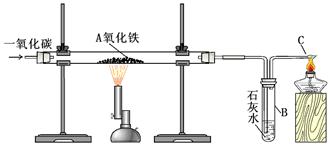
 2Fe3O4+CO2
2Fe3O4+CO2  4Fe+4CO2
4Fe+4CO2 | ʵ����� | Ԥ������ͽ��� |
| ����һ��ȡӲ�ʲ������й�����������ֱ���A��B�Թ��У���������1mol/LCuSO4��Һ�������ܽ⡣ | ��1����A�Թ��к�ɫ���岻�ܽ⣬����û�й۲쵽�����������ɫ����Ϊ ��2����B�Թ����к�ɫ������������˵����ɫ�����к���Fe�� |
| ����������Թ�B����Һ���ˣ������ù���ϴ�Ӹɾ�������1mol/L����������ηֱ��������0.01mol/L��ˮ������0.01mol/L KSCN��Һ | ��1������Һ�����ɫ���� ��2������Һ���ɫ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����ȼǰ��Ҫ���鴿�� | B��������ͬʱ�����ĵ�������ͬ |
| C��ȼ��ʱ����������� | D�����ȼ�գ�������������̼��ˮ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
A���������������ᣩ���ӱ���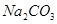 ��Һ��������ú�Һ ��Һ��������ú�Һ |
| B���Ҵ���ˮ��������������ʯ�ң����� |
| C�����ᣨ�Ҵ�������������ƣ����� |
D���屽���壩������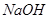 ��Һ��������ú�Һ ��Һ��������ú�Һ |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com