
øßĢśĖį¼ŲŹĒŅ»ÖÖøߊ§µÄ¶ą¹¦ÄÜĖ®“¦Ąķ¼Į£¬¾ßÓŠŃõ»Æ”¢Īüø½”¢ŠõÄż”¢”¢ÖśÄż”¢É±¾ś”¢³ż³ōµČ×÷ÓĆ£¬øßĢśĖį¼ŲµÄæŖ·¢ŗĶĄūÓĆÕżČÕŅęĻŌŹ¾³öĘä¹ćĄ«µÄÓ¦ÓĆĒ°¾°”£ĘäÖʱøĀ·ĻßČēĻĀ£ŗ
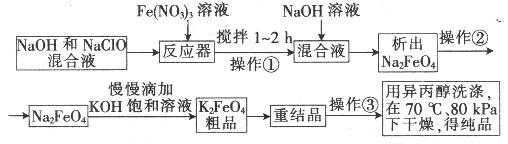
ŹŌ»Ų“šĻĀĮŠĪŹĢā
£Ø1£©²Ł×÷¢Ł½Į°čµÄÄæµÄŹĒ£ŗ ”£
£Ø2£©²Ł×÷¢ŚÓė²Ł×÷¢ŪĻąĶ¬£¬øĆ²Ł×÷Ćū³ĘŹĒ£ŗ___________________”£
£Ø3£©·“Ó¦Ę÷ÖŠ£¬ NaOH”¢NaClO¼°Fe(NO3)3·“Ó¦µÄĄė×Ó·½³ĢŹ½ĪŖ£ŗ _______________________”£
£Ø4£©“ĪĀČĖįÄĘÅØ¶Č¶ŌøßĢśĖį¼Ų²śĀŹÓŠŅ»¶ØÓ°Ļģ£¬µ±øßĢśĖį¼Ų²śĀŹ×īøߏ±£¬NaClOµÄĪļÖŹµÄĮæÅضČŌ¼ĪŖ_________”££Ø¼ĘĖć½į¹ū±£ĮōŅ»Ī»Š”Źż£©

£Ø5£©“Ó»·¾³±£»¤µÄ½Ē¶Čæ“£¬ÖʱøÖŠ¼ä²śĪļNa2FeO4½ĻŗƵķ½·ØĪŖµē»Æѧ·Ø£¬Ęä×°ÖĆČēĶ¼ŅŅĖłŹ¾£¬ŌņŃō¼«µÄµē¼«·“Ó¦Ź½ĪŖ ”£
 Č«ÓŵćĮ·µ„ŌŖ¼Ę»®ĻµĮŠ“š°ø
Č«ÓŵćĮ·µ„ŌŖ¼Ę»®ĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ


²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2014½ģɽ¶«Ź”ĪĵĒŹŠøßČżÉĻѧʌʌ֊Ķ³æ¼Ąķ×Ū»ÆѧŹŌ¾ķ£ØB£©£Ø½āĪö°ę£© ĢāŠĶ£ŗĢīæÕĢā
øßĢśĖį¼ŲŹĒŅ»ÖÖøߊ§µÄ¶ą¹¦ÄÜĖ®“¦Ąķ¼Į£¬¾ßÓŠŃõ»Æ”¢Īüø½”¢ŠõÄż”¢”¢ÖśÄż”¢É±¾ś”¢³ż³ōµČ×÷ÓĆ£¬øßĢśĖį¼ŲµÄæŖ·¢ŗĶĄūÓĆÕżČÕŅęĻŌŹ¾³öĘä¹ćĄ«µÄÓ¦ÓĆĒ°¾°”£ĘäÖʱøĮ÷³ĢČēĻĀ£ŗ

ŹŌ»Ų“šĻĀĮŠĪŹĢā
£Ø1£©·“Ó¦Ę÷ÖŠ£¬NaOH”¢NaClO”¢Fe(NO3)3·¢Éś·“Ӧɜ³ÉNa2FeO4£¬Ķź³É²¢ÅäĘ½ĻĀĮŠĄė×Ó·“Ó¦·½³ĢŹ½£ŗ
”õFe3++”õClO©¤+”õOH©¤ ==”õFeO42©¤+”õCl©¤+”õ___________
£Ø2£©“ĪĀČĖįÄĘÅØ¶Č¶ŌøßĢśĖį¼Ų²śĀŹÓŠŅ»¶ØÓ°Ļģ£¬µ±NaClOÅضČĪŖ298g/LŹ±£¬øßĢśĖįÄĘµÄ²śĀŹ×īøߣ¬“ĖŹ±NaClOµÄĪļÖŹµÄĮæÅضČĪŖ_______________”£
£Ø3£©ÄćČĻĪŖ²Ł×÷¢ŁÄܹ»·¢Éś×Ŗ»ÆµÄŌŅņŹĒ__________£¬ĀżĀżµĪ¼ÓµÄŌŅņµÄŹĒ___________”£
£Ø4£©²Ł×÷¢ŚČē¹ūĪĀ¶Č¹żøß»įŌģ³ÉøßĢśĖį¼ŲµÄ·Ö½ā£¬øßĢśĖį¼ŲŹÜČČ·Ö½āŹ±Éś³É½šŹōŃõ»ÆĪļŗĶŃõĘų£¬øĆ·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ__________________”£
£Ø5£©æÉŅŌÓƵē»Æѧ·ØÖĘČ”Na2FeO4£¬Ęä×°ÖĆČēĶ¼ĖłŹ¾£¬ŌņŃō¼«µÄµē¼«·“Ó¦Ź½ĪŖ £»
Ņõ¼«µÄµē¼«·“Ó¦Ź½ĪŖ______________£»µē½āŅ»¶ĪŹ±¼äŗóČÜOH©¤µÄÅØ¶Č»į_________(Ģī”°Éżøß”±”¢”°½µµĶ”±
»ņ”°²»±ä”±)”£

²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2011-2012ѧğ½ĖÕŹ”øßČż12ŌĀ½×¶ĪŠŌ²āŹŌ»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗĢīæÕĢā
øßĢśĖį¼ŲŹĒŅ»ÖÖøߊ§µÄ¶ą¹¦ÄÜĖ®“¦Ąķ¼Į£¬¾ßÓŠŃõ»Æ”¢Īüø½”¢ŠõÄż”¢”¢ÖśÄż”¢É±¾ś”¢³ż³ōµČ×÷ÓĆ£¬øßĢśĖį¼ŲµÄæŖ·¢ŗĶĄūÓĆÕżČÕŅęĻŌŹ¾³öĘä¹ćĄ«µÄÓ¦ÓĆĒ°¾°”£ĘäÖʱøĀ·ĻßČēĻĀ£ŗ
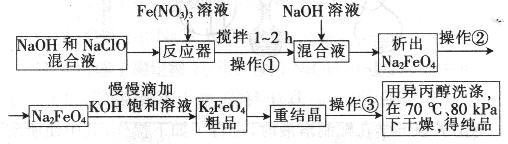
ŹŌ»Ų“šĻĀĮŠĪŹĢā
(1)²Ł×÷¢ŁŗóµÄ»ģŗĻŅŗÖŠ¼ÓČėNaOHČÜŅŗµÄ×÷ÓĆŹĒ£ŗ ”£
×īŗóÓĆŅģ±ū“¼Ļ“µÓ²śĘ·µÄÄæµÄŹĒ ”£
(2) ²Ł×÷¢ŚÓė²Ł×÷¢ŪĻąĶ¬£¬øĆ²Ł×÷Ćū³ĘŹĒ£ŗ___________________”£
(3)·“Ó¦Ę÷ÖŠ£¬NaOH”¢NaClO¼°Fe(NO3)3·“Ó¦µÄĄė×Ó·½³ĢŹ½ĪŖ£ŗ _____________________”£
(4)“ĪĀČĖįÄĘÅØ¶Č¶ŌøßĢśĖį¼Ų²śĀŹÓŠŅ»¶ØÓ°Ļģ£ØĻĀĶ¼¼×£©£¬µ±øßĢśĖį¼Ų²śĀŹ×īøߏ±£¬NaClOµÄĪļÖŹµÄĮæÅضČŌ¼ĪŖ_________”££Ø¼ĘĖć½į¹ū±£ĮōŅ»Ī»Š”Źż£©
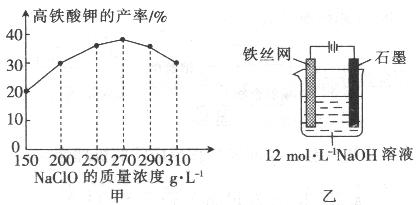
(5)“Ó»·¾³±£»¤µÄ½Ē¶Čæ“£¬ÖʱøÖŠ¼ä²śĪļNa2FeO4½ĻŗƵķ½·ØĪŖµē»Æѧ·Ø£¬Ęä×°ÖĆČēĶ¼ŅŅĖłŹ¾£¬ŌņŃō¼«µÄµē¼«·“Ó¦Ź½ĪŖ ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2010-2011ѧğ½ĖÕŹ”ĖÕĪż³£ÕņĖÄŹŠøßČżµ÷ŃŠ²āŹŌ£ØŅ»£©»ÆѧŹŌ¾ķ ĢāŠĶ£ŗĢīæÕĢā
£Ø12·Ö£©øßĢśĖį¼ŲŹĒŅ»ÖÖøߊ§µÄ¶ą¹¦ÄܵÄĖ®“¦Ąķ¼Į”£¹¤ŅµÉĻ³£²ÉÓĆNaClOŃõ»Æ·ØÉś²ś£¬ŌĄķĪŖ£ŗ
3NaClO + 2Fe(NO3)3 + 10NaOH£½2Na2FeO4”ż+ 3NaCl + 6NaNO3 + 5H2O
Na2FeO4£«2KOH£½K2FeO4£«2NaOH
Ö÷ŅŖµÄÉś²śĮ÷³ĢČēĻĀ£ŗ
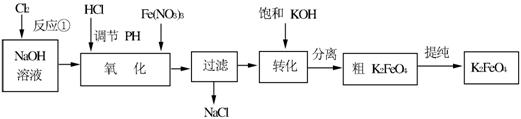
£Ø1£©Š“³ö·“Ó¦¢ŁµÄĄė×Ó·½³ĢŹ½_____________________________”£
£Ø2£©Į÷³ĢĶ¼ÖŠ”°×Ŗ»Æ”±ŹĒŌŚÄ³µĶĪĀĻĀ½ųŠŠµÄ£¬ĖµĆ÷“ĖĪĀ¶ČĻĀKsp(K2FeO4) _________ Ksp(Na2FeO4)£ØĢī”°£¾”±»ņ”°£¼”±»ņ”°£½”±£©”£
£Ø3£©·“Ó¦µÄĪĀ¶Č”¢ŌĮĻµÄÅضČŗĶÅä±Č¶ŌøßĢśĖį¼ŲµÄ²śĀŹ¶¼ÓŠÓ°Ļģ”£
Ķ¼1ĪŖ²»Ķ¬µÄĪĀ¶ČĻĀ£¬Fe(NO3)3²»Ķ¬ÖŹĮæÅØ¶Č¶ŌK2FeO4Éś³ÉĀŹµÄÓ°Ļģ£»
Ķ¼2ĪŖŅ»¶ØĪĀ¶ČĻĀ£¬Fe(NO3)3ÖŹĮæÅضČ×ī¼ŃŹ±£¬NaClOÅØ¶Č¶ŌK2FeO4Éś³ÉĀŹµÄÓ°Ļģ”£
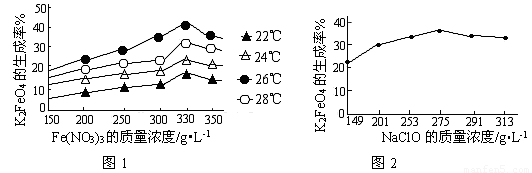
¢Ł¹¤ŅµÉś²śÖŠ×ī¼ŃĪĀ¶ČĪŖ_______”ę£¬“ĖŹ±Fe(NO3)3ÓėNaClOĮ½ÖÖČÜŅŗ×ī¼ŃÖŹĮæÅضČÖ®±ČĪŖ_______”£
¢ŚČōNaClO¼ÓČė¹żĮ棬Ńõ»Æ¹ż³ĢÖŠ»įÉś³ÉFe(OH)3£¬Š“³öøĆ·“Ó¦µÄĄė×Ó·½³ĢŹ½£ŗ
____________________________________________________ӣ
ČōFe(NO3)3¼ÓČė¹żĮ棬ŌŚ¼īŠŌ½éÖŹÖŠK2FeO4ÓėFe3+·¢ÉśŃõ»Æ»¹Ō·“Ӧɜ³ÉK3FeO4£¬“Ė·“Ó¦µÄĄė×Ó·½³ĢŹ½£ŗ____________________________________”£
£Ø4£©K2FeO4 ŌŚĖ®ČÜŅŗÖŠŅ×Ė®½ā£ŗ4FeO42”Ŗ+10H2O 4Fe(OH)3+8OH”Ŗ+3O2”ü”£ŌŚ”°Ģį“æ”±K2FeO4ÖŠ²ÉÓĆÖŲ½į¾§”¢Ļ“µÓ”¢µĶĪĀŗęøɵķ½·Ø£¬ŌņĻ“µÓ¼Į×īŗĆŃ”ÓĆ_______ČÜŅŗ£ØĢīŠņŗÅ£©”£
4Fe(OH)3+8OH”Ŗ+3O2”ü”£ŌŚ”°Ģį“æ”±K2FeO4ÖŠ²ÉÓĆÖŲ½į¾§”¢Ļ“µÓ”¢µĶĪĀŗęøɵķ½·Ø£¬ŌņĻ“µÓ¼Į×īŗĆŃ”ÓĆ_______ČÜŅŗ£ØĢīŠņŗÅ£©”£
A£®H2O B£®CH3COONa”¢Ņģ±ū“¼ C£®NH4Cl”¢Ņģ±ū“¼ D£®Fe(NO3)3”¢Ņģ±ū“¼
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com