
����һ������ˮ��Һ��ֻ���ܺ������������е������֣�K+��NH4+��Cl����Mg2+��Ba2+��CO32����SO42������ȡ���ݸ�100mL��Һ��������ʵ�飺����֪��NH4+��OH�� �� NH3����H2O��
��һ�ݼ���AgNO3��Һ�г���������
�ڶ��ݼ�����NaOH��Һ���Ⱥ��ռ���0.08 mol���塣
�����ݼ�����BaCl2��Һ�õ��������12.54 g,����������ϴ�ӡ������������Ϊ4.66 g.����������ʵ�飬�ش��������⣺
��1���ɵ�һ�ݽ��е�ʵ���ƶϸû�����Ƿ�һ������Cl�� ��
��2���ɵڶ��ݽ��е�ʵ���֪�������Ӧ���� ���ӣ������ʵ���Ũ��Ϊ ��
��3���ɵ����ݽ��е�ʵ���֪12��54g�����ijɷ�Ϊ �������γɸó�����ԭ������и����ӵ����ʵ��� ��
��4���ۺ�����ʵ�飬����Ϊ���½�����ȷ����
A���û��Һ��һ�����У�K+��NH4+��CO32����SO42�������ܺ�Cl������n(K+) ��0.04mol
B���û��Һ��һ�����У�NH4+��CO32����SO42�������ܺ�K+��Cl��
C���û��Һ��һ�����У�NH4+��CO32����SO42�������ܺ�Mg2+��K+��Cl��
D���û��Һ��һ�����У�NH4+��SO42�������ܺ�Mg2+��K+��Cl��
 ���ʿ��ÿ��ֳɳ�ϵ�д�
���ʿ��ÿ��ֳɳ�ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2015-2016ѧ�긣��ʡ�߶������в��Ի�ѧ�Ծ��������棩 ���ͣ�ѡ����
������ʵ��������������ԭ�����͵���
A����ˮ����ƽ�⣺Cl2+H2O?HCl+HClO��������AgNO3��Һ����Һ��ɫ��dz
B����CO��g��+NO2��g��?CO2��g��+NO��g����ƽ����ϵ����ѹǿ��ʹ��ɫ����
C����2NO2(g) N2O4(g)��H<0�������¶�ƽ����ϵ��ɫ����
N2O4(g)��H<0�������¶�ƽ����ϵ��ɫ����
D��SO2��������SO3�ķ�Ӧ��������������Ŀ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015-2016ѧ�꺣��ʡ�߶������У��ģ���ѧ�Ծ��������棩 ���ͣ�ѡ����
����˵����ȷ����
A�����ϵ����滷����ˮ��Դ�ḻ�����ؽ�Լ��ˮ
B��ˮ�帻Ӫ��������Ϊˮ����ֲ��Ӫ�����ʹ���������������Ⱦ
C����ͨ���������ú�Ca2+Ũ�Ƚϸߵ�ˮ������
D���������ô���ˮ����ѧ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015-2016ѧ�꺣��ʡ��һ�����в��Ի�ѧ�Ծ��������棩 ���ͣ�ѡ����
��Na2SO4��Al2(SO4��3�Ļ����Һ�У����Al3+Ũ��Ϊ0.1mol/L��SO42��Ũ��Ϊ0.3mol/L��������Һ��Na+��Ũ��Ϊ
A��0.15mol/L B��0.45mol/L
C��0.6mol/L D��0.3mol/L
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015-2016ѧ�꺣��ʡ��һ�����в��Ի�ѧ�Ծ��������棩 ���ͣ�ѡ����
����˵����ȷ����
A���ڱ�״��ʱ��20mLNH3��60mLO2�����ķ��Ӹ���֮��Ϊ1:3
B����80gNaOH����1Lˮ�У�������Һ��NaOH�����ʵ���Ũ��Ϊ2mol/L
C��18gˮ�ڱ�״���µ����Ϊ22.4L
D����ͬ������CO��N2���������Ϊ1:1
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015-2016ѧ�����ɹŰ�ͷ���и�һ��12���¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
����ͬ�����µ�������Ӧ��2A +B2 =2B +A2��2C +A2 =2A +C2��2B +D2 =2D +B2��
���ж�
A�������ԣ�A2��B2��C2��D2
B����ԭ�ԣ�C����A����B����D��
C��2A��+ D2 = 2D��+A2���ܽ���
D��2C��+B2 =2B��+C2���ܽ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015-2016ѧ�����ɹŰ�ͷ���и�һ��12���¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
����ʵ��װ��һ�㲻���ڷ������ʵ���
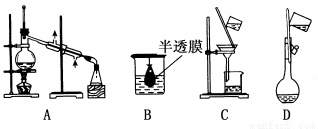
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015-2016ѧ����������ɽ��ѧ�߶��ϵڶ����¿���ѧ�Ծ��������棩 ���ͣ�ѡ����
�����ܴ����������������
A��NH4+��Mg2+��OH����Cl�� B��Ba2+��K+��SO42����NO3��
C��Al3+��Cu2+��SO42����Cl�� D��Na+��Al3+��Cl����AlO2��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2016������и�����ѧ�ڵ��Ĵ��¿����ۻ�ѧ�Ծ��������棩 ���ͣ�ʵ����
ij��ѧ�С���������ͼ��ʾ�����ּг�װ������ȥ��ʵ��װ�ã���̽����ʪ��Cl2��Na2CO3��Ӧ�õ��Ĺ������ʡ�
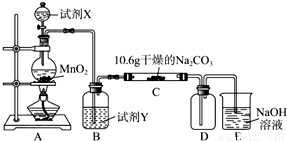
��1��д��װ��A�з�����Ӧ�Ļ�ѧ����ʽ��__________________________��
��2��д���Լ�Y�����ƣ�__________________________��
��3����֪��ͨ��һ�������������D��ֻ��һ�ֳ�����Ϊ�ƺ�ɫ�����壬��Ϊ�������������ȷ������C�к��е�����ֻ��һ�֣��Һ���NaHCO3���ֶ�C�ɷֽ��в����̽����
������������裮
����һ���������ֳɷ֣�NaHCO3��__________��
��������������ֳɷ֣�NaHCO3��___________��_____________��
����Ʒ���������ʵ�飮��д��ʵ�鲽���Լ�Ԥ������ͽ��ۣ��ɲ���������
��ѡʵ���Լ�������������ˮ��ϡ���ᡢBaCl2��Һ������ʯ��ˮ��AgNO3��Һ���Թܡ�С�ձ���
ʵ�鲽�� | Ԥ������ͽ��� |
����1��ȡC�е�����������Ʒ���Թ��У��μ���������ˮ�������ܽ⣬Ȼ��������Һ�ֱ�����A��B�Թ��У� | |
����2��_________________________ | ����_________________________ |
����3��B�Թ��еμӹ�����ϡHNO3��Һ���ٵμ�AgNO3��Һ | ����Һ����ǣ���ϲ���2�Т٣������1��������ϲ���2�Тڣ������2������ |
��4����֪C����0.1mol Cl2�μӷ�Ӧ��������һ����������֪C�з�Ӧ�Ļ�ѧ����ʽΪ
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com