
æĘѧ¼ŅŅ»Ö±ÖĀĮ¦ŃŠ¾æ³£ĪĀ”¢³£Ń¹ĻĀ”°ČĖ¹¤¹ĢµŖ”±µÄŠĀ·½·Ø”£ŌųÓŠŹµŃé±ØµĄ£ŗŌŚ³£ĪĀ”¢³£Ń¹”¢¹āÕÕĢõ¼žĻĀ£¬N2ŌŚ“߻ƼĮ£Ø²ōÓŠÉŁĮæFe2O3µÄTiO2£©±ķĆęÓėĖ®·¢Éś·“Ó¦£¬Éś³ÉµÄÖ÷ŅŖ²śĪļĪŖNH3£¬·“Ó¦µÄ»Æѧ·½³ĢŹ½ČēĻĀ£ŗN2(g)+ 3H2O(l) ![]() 2NH3(g)+ O2(g)”£»Ų“šĻĀĮŠĪŹĢā£ŗ
2NH3(g)+ O2(g)”£»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©½ųŅ»²½ŃŠ¾æNH3Éś³ÉĮæÓėĪĀ¶ČµÄ¹ŲĻµ£¬²æ·ÖŹµŃ鏿¾Ż¼ūĻĀ±ķ£Ø¹āÕÕ”¢N2Ń¹Į¦1.0”Į105 Pa”¢·“Ó¦Ź±¼ä3 h£©£¬ŌņøĆ·“Ó¦µÄÕż·“Ó¦ĪŖ ·“Ó¦£ØĢī”°ĪüČČ”±»ņ”°·ÅČČ”±£©
| T/K | 303 | 313 | 323 |
| NH3Éś³ÉĮæ/£Ø10-6 mol£© | 4.8 | 5.9 | 6.0 |
£Ø2£©ÓėÄæĒ°¹ć·ŗŹ¹ÓĆµÄ¹¤ŅµŗĻ³É°±·½·ØĻą±Č£¬øĆ·½·ØÖŠ¹ĢµŖ·“Ó¦ĖŁĀŹĀż”£ĒėĢį³öæÉĢįøßĘä·“Ó¦ĖŁĀŹĒŅŌö“óNH3Éś³ÉĮæµÄ½ØŅé£ŗ”””” ”£
£Ø3£©ŗĻ³É°±¹¤ŅµÖŠŌĮĻĘųN2æÉ“ÓæÕĘųÖŠ·ÖĄėµĆµ½£¬H2æÉÓĆ¼×ĶéŌŚøßĪĀĻĀÓėĖ®ÕōĘų·“Ó¦ÖʵƔ£¼×ĶéŌŚøßĪĀĻĀÓėĖ®ÕōĘų·“Ó¦·“Ó¦·½³ĢŹ½ĪŖ£ŗCH4(g)£«H2O(g)£½CO(g)£«3H2(g)”£²æ·ÖĪļÖŹµÄČ¼ÉÕČČŹż
¾ŻČēĻĀ£ŗ
H2(g) £ŗ”÷H =£285.8 kJ・mol£1£»
CO(g) £ŗ ”÷H =£283.0 kJ・mol£1£»
CH4(g) £ŗ”÷H =£890.3 kJ・mol£1 ”£
ŅŃÖŖ1mol H2O(g)×Ŗ±äĪŖ1mol H2O(l)Ź±·Å³ö44.0 kJČČĮ攣Š“³öCH4ŗĶH2OŌŚøßĪĀĻĀ·“Ó¦µÄČČ»Æѧ·½³ĢŹ½__________________________________”£
£Ø4£©ÓŠČĖÉčĻėŃ°ĒóŹŹŗĻµÄ“߻ƼĮŗĶµē¼«²ÄĮĻ£¬ŅŌN2”¢H2ĪŖµē¼«·“Ó¦Īļ£¬ŅŌHCl”Ŗ”ŖNH4ClĪŖµē½āÖŹČÜŅŗÖĘ³ÉŠĀŠĶČ¼ĮĻµē³Ų£¬ĒėŠ“³öøƵē¼«µÄÕż¼«·“Ó¦Ź½
£Ø5£©Éś³ÉµÄNH3æÉÓĆÓŚÖĘļ§Ģ¬µŖ·Ź£¬Čē(NH4)2SO4”¢NH4Cl£¬ÕāŠ©·ŹĮĻĻŌ ŠŌ£¬ŌŅņŹĒ£ØÓĆĄė×Ó·½³ĢŹ½±ķŹ¾£©___________________________£¬Ź¹ÓĆŹ±±ÜĆāÓė________________ĪļÖŹŗĻŹ©”£
 ÓÅÉśĄÖŌ°ĻµĮŠ“š°ø
ÓÅÉśĄÖŌ°ĻµĮŠ“š°ø ŠĀ±ąŠ”ѧµ„ŌŖ×Ō²āĢāĻµĮŠ“š°ø
ŠĀ±ąŠ”ѧµ„ŌŖ×Ō²āĢāĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŌĶĮĄķ½ā
| T/K | 303 | 313 | 323 |
| NH3Éś³ÉĮæ/£Ø10-6mol£© | 4.8 | 5.9 | 6.0 |
| H | + 4 |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2012-2013ѧğÉĻŗ£ŹŠĒąĘÖĒųøßæ¼Ņ»Ä£»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗĢīæÕĢā
æĘѧ¼ŅŅ»Ö±ÖĀĮ¦ÓŚ”°ČĖ¹¤¹Ģ°±”±µÄŠĀ·½·ØŃŠ¾æ”£ÄæĒ°ŗĻ³É°±¼¼ŹõŌĄķĪŖ£ŗ
N2(g) +3H2(g)  2NH3(g)+92.4 kJ/mol
2NH3(g)+92.4 kJ/mol
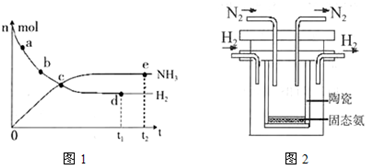
673K£¬30MPaĻĀ£¬ÉĻŹöŗĻ³É°±·“Ó¦ÖŠn(NH3)ŗĶn(H2)Ėꏱ¼ä±ä»ÆµÄ¹ŲĻµČēÓŅĶ¼ĖłŹ¾”£

£Ø1£©ĻĀĮŠŠšŹöÕżČ·µÄŹĒ( )
A£®µćaµÄÕż·“Ó¦ĖŁĀŹ±ČµćbµÄ“ó
B£®µćc“¦·“Ó¦“ļµ½Ę½ŗā
C£®µćdŗĶµće“¦µÄn (N2)ĻąĶ¬
D£®773K£¬30MPa ĻĀ£¬·“Ó¦ÖĮt2Ź±æĢ“ļµ½Ę½ŗā£¬Ōņn(NH3)±ČĶ¼ÖŠeµćµÄÖµ“ó
£Ø2£©ŌŚČŻ»żĪŖ2.0 LŗćČŻµĆĆܱÕČŻĘ÷ÖŠ³äČė0.80 mol N2(g)ŗĶ1.60 mol H2(g)£¬673K”¢30MPaĻĀ“ļµ½Ę½ŗāŹ±£¬NH3µÄĢå»ż·ÖŹżĪŖ20%”£øĆĢõ¼žĻĀ£¬N2(g)+3H2(g)  2NH3(g)µÄĘ½ŗā³£ŹżÖµĪŖ£ŗ_____________”£
2NH3(g)µÄĘ½ŗā³£ŹżÖµĪŖ£ŗ_____________”£
£Ø3£©KÖµŌ½“󣬱ķĆ÷·“Ó¦“ļµ½Ę½ŗāŹ±( )”£
A. H2µÄ×Ŗ»ÆĀŹŅ»¶ØŌ½øß B£®NH3µÄ²śĮæŅ»¶ØŌ½“ó
C£®Õż·“Ó¦½ųŠŠµĆŌ½ĶźČ« D£®»Æѧ·“Ó¦ĖŁĀŹŌ½“ó
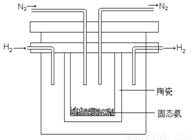
£Ø4£©1998ÄźĻ£Ą°ŃĒĄļĖ¹¶ąµĀ“óѧµÄĮ½Ī»æĘѧ¼Ņ²ÉÓĆøßÖŹ×Óµ¼µēŠŌµÄSCYĢÕ“É£ØÄÜ“«µŻH+£©£¬ŹµĻÖĮĖøßĪĀ”¢³£Ń¹ĻĀøß×Ŗ»ÆĀŹµÄµē½āŗĻ³É°±”£Ę䏵Ńé×°ÖĆČēĶ¼”£Ńō¼«µÄµē¼«·“Ó¦ĪŖ£ŗH2-2e 2H+£¬ŌņŅõ¼«µÄµē¼«·“Ó¦ĪŖ£ŗ_____________”£
2H+£¬ŌņŅõ¼«µÄµē¼«·“Ó¦ĪŖ£ŗ_____________”£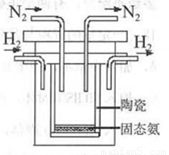
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2014½ģŗŚĮś½Ź”øßŅ»ĻĀŃ§ĘŚĘŚÄ©æ¼ŹŌ»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗĢīæÕĢā
(7·Ö) æĘѧ¼ŅŅ»Ö±ÖĀĮ¦ÓŚ”°ČĖ¹¤¹ĢµŖ”±µÄ·½·ØŃŠ¾æ”£
¢ÅÄæĒ°ŗĻ³É°±µÄ¼¼ŹõŌĄķĪŖ£ŗ
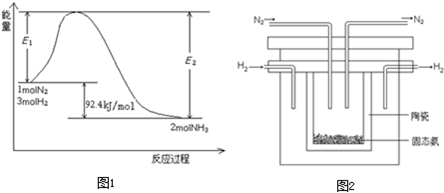
øĆ·“Ó¦µÄÄÜĮæ±ä»ÆČēĶ¼ĖłŹ¾”£

¢ŁŌŚ·“Ó¦ĢåĻµÖŠ¼ÓČė“߻ƼĮ£¬·“Ó¦ĖŁĀŹŌö“ó£¬E2µÄ±ä»ÆŹĒ£ŗ ”££ØĢī”°Ōö“ó”±”¢”°¼õŠ””±»ņ”°²»±ä”±£©”£
¢Ś½«Ņ»¶ØĮæµÄN2(g)ŗĶH2(g)·ÅČė1LµÄĆܱÕČŻĘ÷ÖŠ£¬ŌŚ500”ę”¢2”Į107PaĻĀ“ļµ½Ę½ŗā£¬²āµĆN2ĪŖ0.1 mol£¬H2ĪŖ0.3 mol£¬NH3ĪŖ0.1 mol”£øĆĢõ¼žĻĀH2µÄ×Ŗ»ÆĀŹĪŖ ”£
¢ŪÓūĢįøߢŚČŻĘ÷ÖŠH2µÄ×Ŗ»ÆĀŹ£¬ĻĀĮŠ“ėŹ©æÉŠŠµÄŹĒ ”£
A£®ĻņČŻĘ÷ÖŠ°“Ō±ČĄżŌŁ³äČėŌĮĻĘų B£®ĻņČŻĘ÷ÖŠŌŁ³äČė¶čŠŌĘųĢå
C£®øı䷓ӦµÄ“߻ƼĮ D£®Ņŗ»ÆÉś³ÉĪļ·ÖĄė³ö°±
¢Ę1998ÄźĻ£Ą°ŃĒĄļŹæ¶ąµĀ“óѧµÄĮ½Ī»æĘѧ¼Ņ²ÉÓĆøßÖŹ×Óµ¼µēŠŌµÄSCYĢÕ“É£ØÄÜ“«µ¼H+£©£¬“Ó¶ųŹµĻÖĮĖøß×Ŗ»ÆĀŹµÄµē½ā·ØŗĻ³É°±”£Ę䏵Ńé×°ÖĆČēĶ¼ĖłŹ¾”£Ņõ¼«µÄµē¼«·“Ó¦Ź½ĪŖ ”£

¢Ēøł¾Ż×īŠĀ”°ČĖ¹¤¹ĢµŖ”±µÄŃŠ¾æ±ØµĄ£¬ŌŚ³£ĪĀ”¢³£ Ń¹”¢¹āÕÕĢõ¼žĻĀ£¬N2ŌŚ“߻ƼĮ£Ø²ōÓŠÉŁĮæFe2O3ŗĶTiO2£©±ķĆęÓėĖ®·¢ÉśĻĀĮŠ·“Ó¦£ŗ

½ųŅ»²½ŃŠ¾æNH3Éś³ÉĮæÓėĪĀ¶Č¹ŲĻµ£¬³£Ń¹ĻĀ“ļµ½Ę½ŗāŹ±²āµĆ²æ·ÖŹµŃ鏿¾ŻČēĻĀ£ŗ
|
T/K |
303 |
313 |
323 |
|
NH3Éś³ÉĮæ/£Ø10£6mol£© |
4.8 |
5.9 |
6.0 |
¢ŁŗĻ³É·“Ó¦µÄa_ 0”££ØĢī”°“óÓŚ”±”¢”°Š”ÓŚ”±»ņ”°µČÓŚ”±£©
¢ŚŅŃÖŖ

Ōņ
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2010-2011ѧğ±±¾©ĆÅĶ·¹µĒųøßȿ𼶳éŃł²āŹŌĄķæĘ×ŪŗĻÄÜĮ¦²āŹŌ»Æѧ²æ·Ö ĢāŠĶ£ŗĢīæÕĢā
£Ø14·Ö£©
æĘѧ¼ŅŅ»Ö±ÖĀĮ¦ÓŚ”°ČĖ¹¤¹ĢµŖ”±µÄ·½·ØŃŠ¾æ”£
¢ÅÄæĒ°ŗĻ³É°±µÄ¼¼ŹõŌĄķĪŖ£ŗ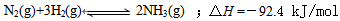
øĆ·“Ó¦µÄÄÜĮæ±ä»ÆČēĶ¼ĖłŹ¾”£

¢ŁŌŚ·“Ó¦ĢåĻµÖŠ¼ÓČė“߻ƼĮ£¬·“Ó¦ĖŁĀŹŌö“ó£¬E2µÄ±ä»ÆŹĒ£ŗ ”££ØĢī”°Ōö“ó”±”¢”°¼õŠ””±»ņ”°²»±ä”±£©”£
¢Ś½«Ņ»¶ØĮæµÄN2(g)ŗĶH2(g)·ÅČė1LµÄĆܱÕČŻĘ÷ÖŠ£¬ŌŚ500”ę”¢2”Į107PaĻĀ“ļµ½Ę½ŗā£¬²āµĆN2ĪŖ0.1 mol£¬H2ĪŖ0.3 mol£¬NH3ĪŖ0.1 mol”£øĆĢõ¼žĻĀH2µÄ×Ŗ»ÆĀŹĪŖ ”£
¢ŪÓūĢįøߢŚČŻĘ÷ÖŠH2µÄ×Ŗ»ÆĀŹ£¬ĻĀĮŠ“ėŹ©æÉŠŠµÄŹĒ ”£
A£®ĻņČŻĘ÷ÖŠ°“Ō±ČĄżŌŁ³äČėŌĮĻĘų
B£®ĻņČŻĘ÷ÖŠŌŁ³äČė¶čŠŌĘųĢå
C£®øı䷓ӦµÄ“߻ƼĮ
D£®Ņŗ»ÆÉś³ÉĪļ·ÖĄė³ö°±
¢Ę1998ÄźĻ£Ą°ŃĒĄļŹæ¶ąµĀ“óѧµÄĮ½Ī»æĘѧ¼Ņ²ÉÓĆøßÖŹ×Óµ¼µēŠŌµÄSCYĢÕ“É£ØÄÜ“«µ¼H+£©£¬“Ó¶ųŹµĻÖĮĖøß×Ŗ»ÆĀŹµÄµē½ā·ØŗĻ³É°±”£Ę䏵Ńé×°ÖĆČēĶ¼ĖłŹ¾”£Ņõ¼«µÄµē¼«·“Ó¦Ź½ĪŖ ”£
¢Ēøł¾Ż×īŠĀ”°ČĖ¹¤¹ĢµŖ”±µÄŃŠ¾æ±ØµĄ£¬ŌŚ³£ĪĀ”¢³£ Ń¹”¢¹āÕÕĢõ¼žĻĀ£¬N2ŌŚ“߻ƼĮ£Ø²ōÓŠÉŁĮæFe2O3ŗĶTiO2£©±ķĆęÓėĖ®·¢ÉśĻĀĮŠ·“Ó¦£ŗ

½ųŅ»²½ŃŠ¾æNH3Éś³ÉĮæÓėĪĀ¶Č¹ŲĻµ£¬³£Ń¹ĻĀ“ļµ½Ę½ŗāŹ±²āµĆ²æ·ÖŹµŃ鏿¾ŻČēĻĀ£ŗ
|
T/K |
303 |
313 |
323 |
|
NH3Éś³ÉĮæ/£Ø10£6mol£© |
4.8 |
5.9 |
6.0 |
¢Ł“ĖŗĻ³É·“Ó¦µÄa_ 0”££ØĢī”°“óÓŚ”±”¢”°Š”ÓŚ”±»ņ”°µČÓŚ”±£©
¢ŚŅŃÖŖ

¢Č NH4ClČÜŅŗ³ŹĖįŠŌ£¬ÕāŹĒÓÉÓŚNH Ė®½āµÄŌµ¹Ź”£ŌņNH4ClŌŚÖŲĖ®£ØD2O£©ÖŠĖ®½āµÄĄė×Ó·½³ĢŹ½ŹĒ
ӣ
Ė®½āµÄŌµ¹Ź”£ŌņNH4ClŌŚÖŲĖ®£ØD2O£©ÖŠĖ®½āµÄĄė×Ó·½³ĢŹ½ŹĒ
ӣ
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com