
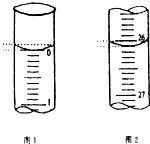
 ijѧ��������֪���ʵ���Ũ�ȵ��������ⶨδ֪���ʵ���Ũ�ȵ�����������Һʱ��ѡ�������ָʾ��������д���пհף�
ijѧ��������֪���ʵ���Ũ�ȵ��������ⶨδ֪���ʵ���Ũ�ȵ�����������Һʱ��ѡ�������ָʾ��������д���пհף�| �ζ����� | ��������������Һ�����/mL | 0.1000mol•L-1��������/mL | ||
| �ζ�ǰ�̶� | �ζ���̶� | ��Һ���/mL | ||
| ��һ�� | 25.00 | 0.00 | 26.11 | 26.11 |
| �ڶ��� | 25.00 | 1.56 | 30.30 | 28.74 |
| ������ | 25.00 | 0.22 | 26.31 | 26.09 |
���� ��1�����ְ�����ʽ�ζ��ܵĻ���������ҡ����ƿ���۾��۾�Ҫע����ƿ����Һ����ɫ�仯���ζ��յ�ʱ��Һ��ɫ�ɺ�ɫͻ��Ϊ��ɫ��
��2������c�����⣩=$\frac{c������V������}{V�����⣩}$��������������V��������Ӱ�죬�Դ��ж�Ũ�ȵ���
��3����ʽ�ζ��ܵ�С�̶����Ϸ���ȷ��Ϊ0.01mL��
��4���ȸ������ݵ���Ч�ԣ���ȥ��2�����ݣ�Ȼ�����1��3��ƽ������V�����ᣩ�����Ÿ��������NaOH��Ӧ���c��NaOH����
��� �⣺��1������к͵ζ�ʱ���۾�Ҫע����ƿ����Һ����ɫ�仯���ζ��յ�ʱ��Һ��ɫ�ɺ�ɫͻ��Ϊ��ɫ���Ұ��������ɫ���䣬
�ʴ�Ϊ����ƿ����Һ��ɫ�ı仯�����������ɫ���䣻
��2��A����ʽ�ζ���δ�ñ�������Һ��ϴ��ֱ��ע���������Һ����Һ��ϡ�ͣ����V������ƫ����c�����⣩=$\frac{c������V������}{V�����⣩}$��֪���ⶨc��NaOH��ƫ��A����
B���ζ�ǰʢ������������Һ����ƿ������ˮϴ����û�и������Һ�����ʵ������䣬��V��������Ӱ�죬����c�����⣩=$\frac{c������V������}{V�����⣩}$��֪���ⶨc��NaOH����Ӱ�죬��B����
C����ʽ�ζ����ڵζ�ǰ�����ݣ��ζ���������ʧ�����V������ƫ����c�����⣩=$\frac{c������V������}{V�����⣩}$��֪���ⶨc��NaOH��ƫ��C����
D����ȡ�������ʱ����ʼ���Ӷ������ζ�����ʱ���Ӷ��������V������ƫС������c�����⣩=$\frac{c������V������}{V�����⣩}$��֪���ⶨc��NaOH��ƫС����D��ȷ��
�ʴ�Ϊ��D��
��3����ͼ��֪����ʼ����Ϊ0.00mL���յ����Ϊ26.10mL������������Һ�����Ϊ26.10mL-0.00mL=26.10 mL��
�ʴ�Ϊ��0.00��26.10��26.10��
��4���������ݵ���Ч�ԣ���ȥ��2�����ݣ���1��3�����ݿ�֪ƽ������V�����ᣩ=$\frac{26.11+26.09}{2}$mL=26.10mL�����ݷ�Ӧ����ʽ��HCl+NaOH=NaCl+H2O��n��HCl��=n��NaOH��������0.0261L��0.1000mol•L-1=0.025L��c��NaOH�������c��NaOH��=$\frac{0.0261L��0.1000mol/L}{0.025L}$=0.1044mol/L��
�ʴ�Ϊ��0.1044mol/L��
���� ���⿼�����ʺ����ⶨʵ�飬Ϊ��Ƶ���㣬����������ʹ�á��к͵ζ���Ӧ�õ�Ϊ���Ĺؼ������ط�����ʵ�������Ŀ��飬ע�⣨4�������ݴ�������Ŀ�ѶȲ���


 53���ò�ϵ�д�
53���ò�ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
| ���� | ������ƽ | С�ձ� | ����ǯ | ������ | ҩ�� | ��Ͳ |
| ���� |  |  |  |  |  | |
| ��� | a | b | c | d | e | f |
| ʵ�� ���� | ��ʼ�¶�t1/�� | ��ֹ�¶� t2/�� | �¶Ȳ�ƽ��ֵ ��t2-t1��/�� | ||
| H2SO4 | NaOH | ƽ��ֵ | |||
| 1 | 26.2 | 26.0 | 26.1 | 30.1 | |
| 2 | 27.0 | 27.4 | 27.2 | 33.3 | |
| 3 | 25.9 | 25.9 | 25.9 | 29.8 | |
| 4 | 26.4 | 26.2 | 26.3 | 30.4 | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
 ������̼�IJ�����������Դ�����һ����Ҫս�Է���
������̼�IJ�����������Դ�����һ����Ҫս�Է��� ��������ѧ���������ǹ��ۼ���
��������ѧ���������ǹ��ۼ����鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
| ʵ����� | ��һ�� | �ڶ��� | ������ |
| ����ʳ�����������/mL | 0.02 | 0.03 | 0.00 |
| ����ʳ������ն���/mL | 25.01 | 25.04 | 25.02 |
| �������Ʊ�Һ�����������/mL | 0.01 | 0.03 | 0.04 |
| �������Ʊ�Һ������ն���/mL | 12.52 | 12.55 | 12.58 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
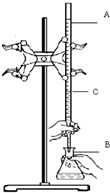 ��ʵ��������֪Ũ�ȵ�����ζ�ijδ֪Ũ�ȵ�NaOH��Һ��װ�úͲ�������ͼ��ʾ����ش�
��ʵ��������֪Ũ�ȵ�����ζ�ijδ֪Ũ�ȵ�NaOH��Һ��װ�úͲ�������ͼ��ʾ����ش��鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
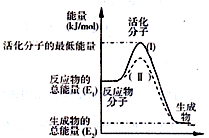 ��֪��Ӧ��3I-��aq��+S2O82-��aq���TI3-��aq��+2SO42-��aq��+Q
��֪��Ӧ��3I-��aq��+S2O82-��aq���TI3-��aq��+2SO42-��aq��+Q| ʵ���� | �� | �� | �� | �� | �� |
| c��I-��/mol•L-1 | 0.040 | 0.080 | 0.080 | 0.160 | 0.160 |
| c��S2O82-��/mol•L-1 | 0.040 | 0.040 | 0.080 | 0.080 | 0.040 |
| t/s | 88 | 44 | 22 | 11 | t1 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | 1 mol�����к���1.204��1024����ԭ�ӣ��ڱ�״����ռ�����22.4 L | |
| B�� | 1 mol������1.5 mol����������ͬ����ԭ���� | |
| C�� | ��He��H2��O2����������¶Ⱥ��ܶȶ���ͬʱ�����ǵ�ѹǿ��С��p��O2����p��He����p��H2�� | |
| D�� | �����ʵ����ĸɱ��������ǣ�����ʽΪC6H12O6��������̼ԭ����֮��Ϊ1��6����ԭ����֮��Ϊ1��3 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ��������ķ�Һ����ˮ�ۣ���ˮ������ˮ�� | |
| B�� | ��ҽ�þƾ�������������ɵõ����ߴ��ȵľƾ���Һ | |
| C�� | ���ȸ�����ع�����ȡ���ռ�����������Ӧ����ֹͣ���� | |
| D�� | ����ʵ��ʱ����ˮӦ���Ͽڽ��¿ڳ� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com