
�����������һ����Ҫ�Ļ�����Ʒ��ij��ȤС�����Ʊ���������ƾ���(Na2S2O3��5H2O)��
��.���������ϡ�
(1)Na2S2O3��5H2O����ɫ�����壬������ˮ����ϡ��Һ��BaCl2��Һ����������ɡ�
(2)��Na2CO3��Na2S�����Һ��ͨ��SO2���Ƶ�Na2S2O3�����ò�Ʒ����������Na2SO3��Na2SO4��
(3)Na2SO3�ױ�������BaSO3������ˮ��������ϡ���ᡣ
��.���Ʊ���Ʒ��
ʵ��װ����ͼ��ʾ(ʡ�Լг�װ��)��
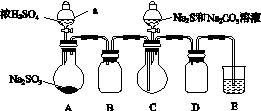
ʵ�鲽�裺
(1)���װ�������ԣ���ͼʾ�����Լ���
����a��������________��E�е��Լ���________(ѡ��������ĸ���)��
A��ϡH2SO4
B��NaOH��Һ
C������NaHSO3��Һ
(2)����C����ƿ����Na2S��Na2CO3�Ļ����Һ������A����ƿ�μ�ŨH2SO4��
(3)��Na2S��Na2CO3��ȫ���ĺ�����Ӧ������C�еĻ�����Һ��________(��д��������)���ᾧ�����ˡ�ϴ�ӡ�����õ���Ʒ��
��.��̽���뷴˼��
(1)Ϊ��֤��Ʒ�к���Na2SO3��Na2SO4����С�����������ʵ�鷽�����뽫��������������(�����Լ���ϡHNO3��ϡH2SO4��ϡ���ᡢ����ˮ��ѡ��)
ȡ������Ʒ���ϡ��Һ���μ�����BaCl2��Һ���а�ɫ�������ɣ�____________________________________��������δ��ȫ�ܽ⣬���д̼�����ζ��������������ȷ����Ʒ�к���Na2SO3��Na2SO4��
(2)Ϊ����װ��C������Na2SO4�������ڲ��ı�ԭ��װ�õĻ����϶�ʵ�鲽��(2)�����˸Ľ����Ľ���IJ�����______________________________________________________________________________��
(3)Na2S2O3��5H2O���ܽ�����¶����������������ò�Ʒͨ��________�����ᴿ��
��.(1)�ٷ�Һ©������B
(3)����
��.(1)���ˣ�������ˮϴ�ӳ�����������м�������ϡ���ᡡ(2)����A����ƿ�μ�ŨH2SO4�����������彫װ���еĿ����ž�������C����ƿ����Na2S��Na2CO3�����Һ
(3)�ؽᾧ
[����] ��.(1)��aΪ��Һ©������E���Լ���������SO2β������ΪNaOH��Һ��(3)����Na2S2O3��5H2O����ɫ�����壬������ˮ����Ҫ����Һ�еõ���������ƾ��壬�辭������Ũ������ȴ�ᾧ�����ˣ�ϴ�ӣ�����õ���Ʒ����. (1)Na2S2O3��5H2O��ϡ��Һ��BaCl2����������ɣ���ʵ��������а�ɫ�������ɣ������һ����֤�������ɫ�����еμ�ϡ���ᣬ������δ��ȫ�ܽ⣬���д̼�����ζ��������������ȷ����Ʒ�к���Na2SO3��Na2SO4��(2)Na2SO3�����ױ������е�����������Na2SO4��Ϊ�˼���Na2SO4���ɵ��������ſ�װ���еĿ�����(3)Na2S2O3��5H2O���ܽ�����¶������������ᾧʱ���������������ʣ����ͨ���ؽᾧ�ķ����ᴿ��


 ��ս�п�����ϵ�д�
��ս�п�����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��ʯ����Ҫ�ɷ�Ϊ�����Σ���ǰ20��Ԫ���е�������ɣ���ѧʽΪXYZ3W8�����У�ֻ��W�Ը��ۡ�X��Y������������֮����Z�������������ȡ�Y3����W�������Ӿ�����ͬ�ĵ��Ӳ�ṹ��X��W��������֮�͵���Y��Z��������֮�͡�����˵���������(����)21��cn��jy��com
A��X�����Ӱ뾶>Y�����Ӱ뾶
B��Z���⻯���ȶ���<W���⻯���ȶ���
C��Y����������������ᣬ������NaOH��Һ��Ӧ
D��X2W2��Y2W3���ֻ����ﺬ�еĻ�ѧ��������ȫ��ͬ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
���и����е��������������Ժ��ֱ�����ϣ�ֻҪ�����������ʵ���һ��������ȫȼ��ʱ����ˮ������������O2��������������� �� ��
A��C3H6��C4H6O2 B ��C2H4��C2H6O C��C2H2��C6H6 D ��CH4O��C3H4O3
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
������ͭ�Ǻϳ��������������в��ϡ�����������ͭ����Ҫǰ����֮һ������������һ��ʵ���Һϳ�·�ߣ�
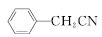 ��H2O��H2SO4
��H2O��H2SO4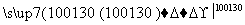
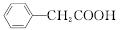 ��NH4HSO4
��NH4HSO4
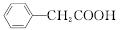 | |||
 | |||
��Cu(OH)2�D�� ��H2O
�Ʊ��������װ��ʾ��ͼ����(���Ⱥͼг�װ�õ���)��
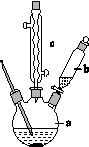
��֪����������۵�Ϊ76.5 �棬������ˮ�������Ҵ���
�ش��������⣺
(1)��250 mL����ƿa�м���70 mL 70%���ᡣ���ƴ�����ʱ����������ˮ��Ũ������Ⱥ�˳����______________________________��
(2)��a�е���Һ������100 �棬�����μ�40 g�����浽������Һ�У�Ȼ��������130 �������Ӧ����װ���У�����b��������__________������c��������__________����������____________��
��Ӧ�������������ˮ���ٷ�����������Ʒ��������ˮ��Ŀ����____________�����������п����ڷ��뱽�����Ʒ����____________(����)��
A����Һ©����B��©����C���ձ���D��ֱ�������ܡ�E��������
(3)�ᴿ�ֱ�����ķ�����____________�����յõ�44 g��Ʒ��������IJ�����____________��
(4)��CuCl2��2H2O��NaOH��Һ�Ʊ�����Cu(OH)2�����������������ˮϴ�ӳ������жϳ���ϴ�ɾ���ʵ�������������__________________��
(5)����������뵽�Ҵ���ˮ�Ļ���ܼ��У�����ܽ����Cu(OH)2����30 min�����ˣ���Һ����һ��ʱ�䣬����������ͭ���壬����ܼ����Ҵ���������________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
���й�������Ӧ�ú���ɵ�˵����ȷ����(����)
A��P2O5�����ڸ���Cl2��NH3
B������ȼ������Ҫ�ɷ��Ǽ����ˮ
C��CCl4�����ڼ�����ˮ�͵�ˮ
D��Si��SiO2������������ά
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
Ϊʵ������ʵ��Ŀ�ģ������±��ṩ����Ҫ�����������Լ���������(����)
| ѡ�� | ʵ��Ŀ�� | ��Ҫ���� | �Լ� |
| A | ����Br2��CCl4�Ļ���� | ��Һ©�����ձ� | Br2��CCl4�Ļ�������ˮ |
| B | ���������Ǻ����� | �Թܡ��ձ����ƾ��� | ��������Һ��������Һ��������Һ |
| C | ʵ������ȡH2 | �Թܡ������ܵ���Ƥ�� | п����ϡHNO3 |
| D | �ⶨNaOH��Һ��Ũ�� | �ζ��ܡ���ƿ���ձ� | NaOH��Һ�� 0��100 0 mol��L��1���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����������������۷���Ϣ�صijɷ�֮һ�������㽶����ζ��ʵ�����Ʊ������������ķ�Ӧ��װ��ʾ��ͼ���й��������£�
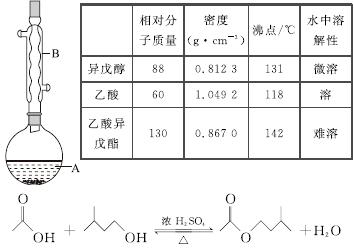
ʵ�鲽�裺
��A�м���4.4 g���촼��6.0 g���ᡢ����Ũ�����2��3Ƭ���Ƭ����ʼ��������A������50 min����ӦҺ�������º����Һ©���У��ֱ�������ˮ������̼��������Һ��ˮϴ�ӣ��ֳ��IJ������������ˮMgSO4���壬����Ƭ�̣����˳�ȥMgSO4���壬�����������ռ�140��143 ����֣�������������3.9 g��
�ش��������⣺
(1)����B��������________________��
(2)��ϴ�Ӳ����У���һ��ˮϴ����ҪĿ����____________________________________���ڶ���ˮϴ����ҪĿ����________________��
(3)��ϴ�ӡ���Һ�����У�Ӧ�����Ȼ���ã����ֲ��________(����)��
a��ֱ�ӽ������������ӷ�Һ©�����Ͽڵ���
b��ֱ�ӽ������������ӷ�Һ©�����¿ڷų�
c���Ƚ�ˮ��ӷ�Һ©�����¿ڷų����ٽ��������������¿ڷų�
d���Ƚ�ˮ��ӷ�Һ©�����¿ڷų����ٽ��������������Ͽڵ���
(4)��ʵ���м�����������Ŀ����___________________________________________��
(5)ʵ���м���������ˮMgSO4��Ŀ����________��
(6)����������У�����ѡ��װ����ȷ����________(����)��

������a��������������������������b.
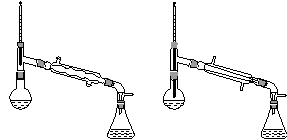
������c��������������������������d.
(7)��ʵ��IJ�����________(����)��
a��30% ��b��40% c��60% d��90%
(8)�ڽ����������ʱ������130 ��㿪ʼ�ռ���֣���ʹʵ��IJ���ƫ__________(��ߡ��͡�)����ԭ����______________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
H2O2��һ����ɫ������ԭ�Լ����ڻ�ѧ�о���Ӧ�ù㷺��
(1)ijС������ͬŨ��Fe3���Ĵ��£�̽��H2O2Ũ�ȶ�H2O2�ֽⷴӦ���ʵ�Ӱ�졣��ѡ�Լ���������30%H2O2��Һ��0.1 mol��L��1Fe2(SO4)3��Һ������ˮ����ƿ��˫������ˮ�ۡ����ܡ��������ܡ���Ͳ�����������ˮԡ�ۡ�ע������
��д����ʵ��H2O2�ֽⷴӦ����ʽ����������ת�Ƶķ������Ŀ��______________________________��
�����ʵ�鷽�����ڲ�ͬH2O2Ũ���£��ⶨ________(Ҫ������õ�������ֱ�����ַ�Ӧ���ʴ�С)��
�����ʵ��װ�ã����ͼ�е�װ��ʾ��ͼ��
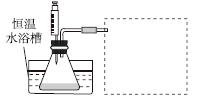
�ܲ����±���ʽ���ⶨʵ�������������ʵ�鷽��(�г���ѡ�Լ���������¼�Ĵ��������������ⶨ�����ݣ���������ĸ��ʾ)��
| ���������� ʵ����� ���� | V[0.1 mol��L��1 Fe2(SO4)3]/mL | ���� | |
| 1 | a | ���� | |
| 2 | a | ���� |
(2)����ͼ(a)��(b)�е���Ϣ����ͼ(c)װ��(��ͨ��A��Bƿ���ѳ���NO2����)����ʵ�顣�ɹ۲쵽Bƿ��������ɫ��Aƿ�е�__________(����dz��)����ԭ����____________________________��
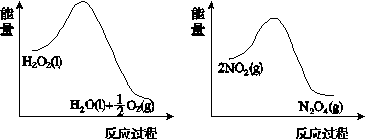
����������(a)������������������������(b)
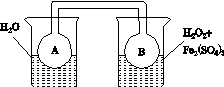
(c)
ͼ21
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
NA��ʾ�����ӵ�����������������ȷ���� ( )
A��1 mol Fel2������������Ӧʱת�Ƶĵ�����Ϊ2NA
B��2 L 0��5 mol/l�������Һ�����������������ΪNA
C��1 mol Na202�����к���������Ϊ4NA
D����ϩ�ͻ�������ɵ�42 g�����������ԭ�ӵĸ���Ϊ6NA
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com