
£Ø18·Ö£©A”¢B”¢CŹĒ֊ѧ»Æѧ³£¼ūµÄČżÖÖĪļÖŹ£¬ĖüĆĒÖ®¼äµÄĻą»„×Ŗ»Æ¹ŲĻµČēĻĀ£Ø²æ·Ö·“Ó¦Ģõ¼ž¼°²śĪļĀŌČ„£©”£
¢Å ČōAŹĒŅ»ÖÖ»ĘÉ«µ„ÖŹ¹ĢĢ壬ŌņB”śCµÄ»Æѧ·½³ĢŹ½ĪŖ ”£
¢Ę ČōAŹĒŅ»ÖÖ»īĘĆ½šŹō£¬CŹĒµ»ĘÉ«¹ĢĢ壬ŌņCµÄĆū³ĘĪŖ £¬CæÉÓĆŌŚæóɽ”¢æÓµĄ”¢Ē±Ė®»ņÓīÖę·É“¬µČȱŃõµÄ³”ŗĻ£¬½«ČĖĆĒŗō³öµÄCO2ŌŁ×Ŗ»»³ÉO2£¬ŅŌ¹©ŗōĪüÖ®ÓĆ£¬ĒėÓĆ»Æѧ·½³ĢŹ½±ķŹ¾øĆ·“Ó¦ŌĄķ ”£
¢ĒČōAĪŖÄÜŹ¹ŹŖČóµÄŗģÉ«ŹÆČļŹŌÖ½±äĄ¶µÄĘųĢ壬CŹĒŗģ×ŲÉ«ĘųĢ壬ĒŅBŗĶCŅ×ŌŚæÕĘųŠĪ³É¹ā»ÆѧŃĢĪķ”£
¢ŁŌņAĪŖ £¬CĪŖ £¬(ĢīŠ“»ÆѧŹ½)
ŹµŃéŹŅÓĆŹģŹÆ»ŅÓėĀČ»Æļ§¹ĢĢå¹²ČČÖĘČ”A£¬Š“³ö“Ė»Æѧ·½³ĢŹ½£ŗ ”£
¢ŚŠ“³öCÓėĖ®·“Ó¦µÄ»Æѧ·½³ĢŹ½£ŗ ”£

| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ

| “߻ƼĮ |
| ¼ÓČČ |
| “߻ƼĮ |
| ¼ÓČČ |


²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ

| ||
| ”÷ |
| ||
| ”÷ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŌĶĮĄķ½ā
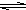 HS-+OH-
HS-+OH-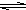 HS-+OH-
HS-+OH-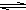 Al£ØOH£©3+3H+ÓŠĪŽÉ«ĘųĢåŗĶ°×É«Šõד³Įµķ²śÉś
Al£ØOH£©3+3H+ÓŠĪŽÉ«ĘųĢåŗĶ°×É«Šõד³Įµķ²śÉś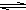 Al£ØOH£©3+3H+ÓŠĪŽÉ«ĘųĢåŗĶ°×É«Šõד³Įµķ²śÉś
Al£ØOH£©3+3H+ÓŠĪŽÉ«ĘųĢåŗĶ°×É«Šõד³Įµķ²śÉś²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
 ŅŃÖŖA”¢B”¢CŹĒ֊ѧ»ÆѧµÄ³£¼ūĪļÖŹ£¬ĖüĆĒŌŚŅ»¶ØĢõ¼žĻĀÓŠČēĻĀ×Ŗ»Æ¹ŲĻµ£ŗ
ŅŃÖŖA”¢B”¢CŹĒ֊ѧ»ÆѧµÄ³£¼ūĪļÖŹ£¬ĖüĆĒŌŚŅ»¶ØĢõ¼žĻĀÓŠČēĻĀ×Ŗ»Æ¹ŲĻµ£ŗ
| ||
| ”÷ |
| ||
| ”÷ |


²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŌĶĮĄķ½ā
| “߻ƼĮ |
| ”÷ |
| “߻ƼĮ |
| ”÷ |
| ||
| ||
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com