
| ���� ���� | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0�� |
| 2 | �� | �� | �� | |||||
| 3 | �� | �� | �� | �� | �� | |||
| 4 | �� |
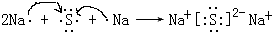 ���û������������ӻ��������ۡ������ӡ�����
���û������������ӻ��������ۡ������ӡ�����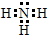 ���û����ﺬ�Ļ�ѧ�������ǹ��ۼ���
���û����ﺬ�Ļ�ѧ�������ǹ��ۼ��� ���� ��Ԫ�������ڱ��е�λ�ÿ�֪������C������N������O������Na������Al������S������Cl������Ar������K��
��1��ϡ�����廯ѧ�����ȶ���
��2��ͬ����Ԫ�ص�ԭ�ӣ�����������������Ӧˮ�������������ǿ������������ͬ����Ԫ�ص�ԭ�ӣ����µ�������������Ӧˮ�������������ǿ������������
��3�������������Ӻ�������֮��ͨ�����Ӽ��γɵ����ӻ����
��4��NԪ�غ���Ԫ�ذ�ԭ�Ӹ�����1��3�γɵĻ�����ΪNH3��Nԭ�Ӻ�3��Hԭ�Ӽ��Թ��ۼ���ϣ�
��� �⣺��Ԫ�������ڱ��е�λ�ÿ�֪������C������N������O������Na������Al������S������Cl������Ar������K��
��1������ЩԪ���У���ѧ�������ȶ�����ϡ������Ar���ʴ�Ϊ��Ar��
��2������Ԫ���У�Cl�ķǽ�������ǿ����������ǿ�Ļ�����ΪHClO4��K�Ľ�������ǿ��������ǿ����KOH���ʴ�Ϊ��KOH��
��3�������������Ӻ�������֮��ͨ�����Ӽ��γɵ����ӻ�����γɹ���Ϊ��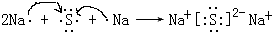 ��
��
�ʴ�Ϊ��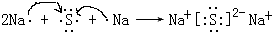 �����ӣ�
�����ӣ�
��4��NH3���ڹ��ۻ����Nԭ�Ӻ�Hԭ�Ӽ�Ϊ���ۼ��������д���3�Թ��õ��Ӷԣ���ԭ�������Ϊ8�����ӣ������ĵ���ʽΪ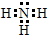 ���ʴ�Ϊ��
���ʴ�Ϊ��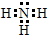 �����ۼ���
�����ۼ���
���� ���⿼��λ�á��ṹ�����ʣ�Ϊ��Ƶ���㣬�漰Ԫ�����ڱ���Ԫ�������ɵȣ���Ŀ�ѶȲ���ע�����ʽ����д�Լ��õ���ʽ��ʾ�γɹ��̵IJ�ͬ��


 ��ĩ���100�ִ��½����ȫ�Ծ�ϵ�д�
��ĩ���100�ִ��½����ȫ�Ծ�ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
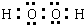 ��[Fe��C2O4��3]3-������������������������ӣ���������������ӣ�������������������
��[Fe��C2O4��3]3-������������������������ӣ���������������ӣ��������������������鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | �����ܶȱ�Һ̬ˮ���ܶ�С | B�� | NH3��Һ�� | ||
| C�� | NH3��PH3�����ȶ� | D�� | �Ҵ��������������ˮ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | H2O2 | B�� | N2 | C�� | C2H4 | D�� | BeCl2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ��pH=1��Һ�У�NH4+��Fe2+��NO3-��Cl- | |
| B�� | ��ˮ�����c��H+��=10-12 mol•L-1����Һ��AlO2-��K+��CO32-��Na+ | |
| C�� | ʹ��̪��Һ������Һ��Na+��Cl-��SO42-��CO32- | |
| D�� | ��������ΪNa2SO4����Һ��K+��CO32-��NO3-��Al3+ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
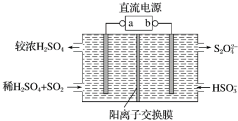 ����ͼ����ʾװ�ã��缫��Ϊ���Ե缫��������SO2�����������ų�����Һ����NO2������˵����ȷ���ǣ�������
����ͼ����ʾװ�ã��缫��Ϊ���Ե缫��������SO2�����������ų�����Һ����NO2������˵����ȷ���ǣ�������| A�� | aΪֱ����Դ�ĸ��� | |
| B�� | һ��ʱ�����������Һ��pH��С | |
| C�� | �����ĵ缫��ӦʽΪSO2+2H2O-2e-=SO42-+4H+ | |
| D�� | ���ʱ��H+��������ͨ�������ӽ���Ĥ�������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����

| A�� | ����������ڷ����廯���� | |
| B�� | 1mol�������������6molNaOH��Ӧ | |
| C�� | �������������ˮ | |
| D�� | ��������ķ���ʽΪC26H24O8Cl6 |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com