
��ˮ![]() �ữ
�ữ![]() ����
����![]() �������塢�����������
�������塢�����������![]() ����
����![]() ����
����![]() ��
��
����������⣺
��1��д��Cl2��������������Br-�ķ�Ӧ�����ӷ���ʽ________________________ ��
��2��ΪʲôҪ�����������¶������ڼ�����������Br-��
��3�����������ɵ��嵥��Ϊʲô�������ȿ���������
��4���ȿ���������Br2��SO2��ˮ���գ�д����һ��Ӧ�Ļ�ѧ����ʽ__________________��
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2010-2011�긣����Ϫһ�С��ݰ�һ�С�������ѧ�߶���ѧ����ĩ������ѧ�� ���ͣ������
��16�֣�
��1����֪298Kʱ��Fe(OH)3���ܶȻ����� =2.6��10-39�� Mg(OH)2���ܶȻ�����
=2.6��10-39�� Mg(OH)2���ܶȻ����� =5.6��
=5.6�� ��ȡ����Ũ�Ⱦ�Ϊ0.1mol/L��MgCl2��FeCl3���Һ����HCl��������һ������MgCO3�ﵽ������Һƽ�⣬���pH=4.00������¶��²�������Һ�е�c(Fe3+)=______________��
��ȡ����Ũ�Ⱦ�Ϊ0.1mol/L��MgCl2��FeCl3���Һ����HCl��������һ������MgCO3�ﵽ������Һƽ�⣬���pH=4.00������¶��²�������Һ�е�c(Fe3+)=______________��
��û��Mg(OH)2��������______________��С����ޡ�����������________________________________��
��2����HnA���B(OH)m��ȫ��Ӧ��������.
����HnAΪHCl���Ҹ�����Һ��pH��7�������ӷ���ʽ˵��ԭ��
������0.4mol��L��1��NaOH��Һ��0.2mol��L��1��HnA��Һ�������Ϻ�pH=10��
��HnAΪ ������ţ�.
a.һԪǿ�� b. һԪ���� c. ��Ԫǿ�� d. ��Ԫ����
��3��ijѧУ������ȤС��Ӻ�ˮɹ�κ����±(��Ҫ��Na+��Mg2+��Cl����Br����)��ģ�ҵ��������ȡþ����Ҫ�������£��ش��������⣺
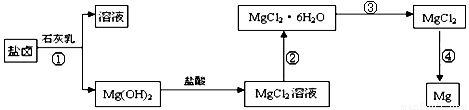
��.�ӹ��̢ٵõ���Mg(OH)2�����л���������Ca(OH)2 ����ȥ����Ca(OH)2�ķ������Ƚ��������뵽ʢ�� ��Һ���ձ��У���ֽ�����ˡ�ϴ�ӿɵô�����Mg(OH)2��
��.���̢۵�ת����Ҫ��HCl�����м��ȣ�HCl��������
��.д�����̢��з�����Ӧ�Ļ�ѧ����ʽ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��1����֪298Kʱ��Fe(OH)3���ܶȻ�����![]() =2.6��10-39�� Mg(OH)2���ܶȻ�����
=2.6��10-39�� Mg(OH)2���ܶȻ�����![]() =5.6��
=5.6��![]() ��ȡ����Ũ�Ⱦ�Ϊ0.1mol/L��MgCl2��FeCl3���Һ����HCl��������һ������MgCO3�ﵽ������Һƽ�⣬���pH=4.00������¶��²�������Һ�е�c(Fe3+)=______________��
��ȡ����Ũ�Ⱦ�Ϊ0.1mol/L��MgCl2��FeCl3���Һ����HCl��������һ������MgCO3�ﵽ������Һƽ�⣬���pH=4.00������¶��²�������Һ�е�c(Fe3+)=______________��
��û��Mg(OH)2��������______________��С����ޡ�����������________________________________��
��2����HnA���B(OH)m��ȫ��Ӧ��������.
����HnAΪHCl���Ҹ�����Һ��pH��7�������ӷ���ʽ˵��ԭ��
������0.4mol��L��1��NaOH��Һ��0.2mol��L��1��HnA��Һ�������Ϻ�pH=10��
��HnAΪ ������ţ�.
a.һԪǿ�� b. һԪ���� c. ��Ԫǿ�� d. ��Ԫ����
��3��ijѧУ������ȤС��Ӻ�ˮɹ�κ����±(��Ҫ��Na+��Mg2+��Cl����Br����)��ģ�ҵ��������ȡþ����Ҫ�������£��ش��������⣺
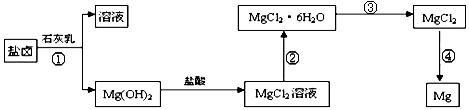
��.�ӹ��̢ٵõ���Mg(OH)2�����л���������Ca(OH)2 ����ȥ����Ca(OH)2�ķ������Ƚ��������뵽ʢ�� ��Һ���ձ��У���ֽ�����ˡ�ϴ�ӿɵô�����Mg(OH)2��
��.���̢۵�ת����Ҫ��HCl�����м��ȣ�HCl��������
��.д�����̢��з�����Ӧ�Ļ�ѧ����ʽ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��16�֣�
��1����֪298Kʱ��Fe(OH)3���ܶȻ�����![]() =2.6��10-39�� Mg(OH)2���ܶȻ�����
=2.6��10-39�� Mg(OH)2���ܶȻ�����![]() =5.6��
=5.6��![]() ��ȡ����Ũ�Ⱦ�Ϊ0.1mol/L��MgCl2��FeCl3���Һ����HCl��������һ������MgCO3�ﵽ������Һƽ�⣬���pH=4.00������¶��²�������Һ�е�c(Fe3+)=______________��
��ȡ����Ũ�Ⱦ�Ϊ0.1mol/L��MgCl2��FeCl3���Һ����HCl��������һ������MgCO3�ﵽ������Һƽ�⣬���pH=4.00������¶��²�������Һ�е�c(Fe3+)=______________��
��û��Mg(OH)2��������______________��С����ޡ�����������________________________________��
��2����HnA���B(OH)m��ȫ��Ӧ��������.
����HnAΪHCl���Ҹ�����Һ��pH��7�������ӷ���ʽ˵��ԭ��
������0.4mol��L��1��NaOH��Һ��0.2mol��L��1��HnA��Һ�������Ϻ�pH=10��
��HnAΪ ������ţ�.
a.һԪǿ�� b. һԪ���� c. ��Ԫǿ�� d. ��Ԫ����
��3��ijѧУ������ȤС��Ӻ�ˮɹ�κ����±(��Ҫ��Na+��Mg2+��Cl����Br����)��ģ�ҵ��������ȡþ����Ҫ�������£��ش��������⣺
��.�ӹ��̢ٵõ���Mg(OH)2�����л���������Ca(OH)2 ����ȥ����Ca(OH)2�ķ������Ƚ��������뵽ʢ�� ��Һ���ձ��У���ֽ�����ˡ�ϴ�ӿɵô�����Mg(OH)2��
��.���̢۵�ת����Ҫ��HCl�����м��ȣ�HCl��������
��.д�����̢��з�����Ӧ�Ļ�ѧ����ʽ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010��2011�긣����Ϫһ�С��ݰ�һ�С�������ѧ�߶���ѧ����ĩ������ѧ�� ���ͣ������
��16�֣�
��1����֪298Kʱ��Fe(OH)3���ܶȻ����� =2.6��10-39��Mg(OH)2���ܶȻ�����
=2.6��10-39��Mg(OH)2���ܶȻ����� =5.6��
=5.6�� ��ȡ����Ũ�Ⱦ�Ϊ0.1mol/L��MgCl2��FeCl3���Һ����HCl��������һ������MgCO3�ﵽ������Һƽ�⣬���pH=4.00������¶��²�������Һ�е�c(Fe3+)=______________��
��ȡ����Ũ�Ⱦ�Ϊ0.1mol/L��MgCl2��FeCl3���Һ����HCl��������һ������MgCO3�ﵽ������Һƽ�⣬���pH=4.00������¶��²�������Һ�е�c(Fe3+)=______________��
��û��Mg(OH)2��������______________��С����ޡ�����������________________________________��
��2����HnA���B(OH)m��ȫ��Ӧ��������.
����HnAΪHCl���Ҹ�����Һ��pH��7�������ӷ���ʽ˵��ԭ��
������0.4mol��L��1��NaOH��Һ��0.2mol��L��1��HnA��Һ�������Ϻ�pH=10��
��HnAΪ ������ţ�.
a.һԪǿ�� b. һԪ���� c. ��Ԫǿ�� d. ��Ԫ����
��3��ijѧУ������ȤС��Ӻ�ˮɹ�κ����±(��Ҫ��Na+��Mg2+��Cl����Br����)��ģ�ҵ��������ȡþ����Ҫ�������£��ش��������⣺
��.�ӹ��̢ٵõ���Mg(OH)2�����л���������Ca(OH)2 ����ȥ����Ca(OH)2�ķ������Ƚ��������뵽ʢ�� ��Һ���ձ��У���ֽ�����ˡ�ϴ�ӿɵô�����Mg(OH)2��
�� .���̢۵�ת����Ҫ��HCl�����м��ȣ�HCl��������
.���̢۵�ת����Ҫ��HCl�����м��ȣ�HCl��������
��.д�����̢��з�����Ӧ�Ļ�ѧ����ʽ
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com