
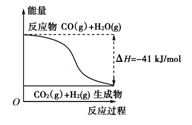
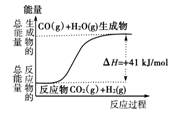
| A�����Ȼ�ѧ����ʽΪ��CO(g)��H2O(g)===CO2(g)��H2(g)������H����41 kJ/mol |
| B���÷�ӦΪ���ȷ�Ӧ |
| C���÷�ӦΪ���ȷ�Ӧ |
| D����H2OΪҺ̬ʱ���䷴Ӧ��ֵС��41 kJ/mol |
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
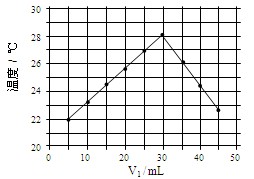
| A������ʵ��ʱ�����¶�Ϊ22�� | B����ʵ�������ѧ�ܿ���ת��Ϊ���� |
| C��NaOH��Һ��Ũ��ԼΪ1.0mol/L | D����ʵ�������ˮ���ɵķ�Ӧ���Ƿ��ȷ�Ӧ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��ʯ��ʯ���·ֽ� | B������ˮ��Ӧ |
| C������������ˮ��Ӧ | D�����ȷ�Ӧ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����-��ת�� | B����-��ת�� |
| C����-��ѧ��ת�� | D����-��������ת�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A���ؼ������ȷ�Ӧ�������Է���Ӧ |
| B����֪C(ʯī��s)��C(���ʯ��s) ��H��0������ʯ��ʯī�ȶ� |
| C��ʹ�ô����Ȳ���ı䷴Ӧ�����ֲ���ı䷴Ӧ�ʱ� |
| D����֪2C(s)��2O2(g)��2CO2(g)) ��H1 2C(s)��O2(g)��2CO(g) ��H2�����H1����H2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
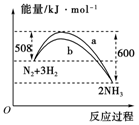
 ���һ����ѧ��Ӧ����Ӧ����������������������������ͼ��ʾ�Ĺ�ϵ����÷�Ӧ����__________��Ӧ��
���һ����ѧ��Ӧ����Ӧ����������������������������ͼ��ʾ�Ĺ�ϵ����÷�Ӧ����__________��Ӧ��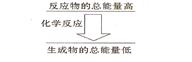
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
 2H2(g)+O2(g)�����Զ����������Ǹ����о�����Ҫ�ȵ㡣����Ϊ�����о�����������
2H2(g)+O2(g)�����Զ����������Ǹ����о�����Ҫ�ȵ㡣����Ϊ�����о����������� | A��Ѱ�Ҹ���Ļ�ʯȼ�ϣ�ȼ�ղ�������ʹˮ�ֽ����H2 |
| B���跨��̫����۽����������£�ʹˮ�ֽ����H2 |
| C��Ѱ�Ҹ�Ч���ʵĴ������ڽ�����������ʹˮ�ֽ����H2 |
| D��Ѱ������Ļ�ѧ���ʣ����ڿ���������Դ���Էֽ�ˮ��ȡH2 |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com