
(14·Ö)ŅŃ ÖŖ“ęŌŚČēĻĀ·“Ó¦£ŗ
ÖŖ“ęŌŚČēĻĀ·“Ó¦£ŗ
ĘäÖŠAŗĶCĪŖ³£¼ū½šŹōµ„ÖŹ£»BŗĶDĪŖŃõ»ÆĪļĒŅB¾ßÓŠ“ÅŠŌ£»EĪŖĪŽÉ«ĘųĢåµ„ÖŹ”£
¾Ż“Ė»Ų“š£ŗ
£Ø1£©DæÉČÜÓŚĒæĖįŗĶĒæ¼ī£¬Š“³öDÓėĒāŃõ»ÆÄĘČÜŅŗ·“Ó¦µÄĄė×Ó·½³ĢŹ½
ӣ
£Ø2£©ĪļÖŹEŹĒ £ØĢī»ÆѧŹ½£©£»ŌŚ·“Ó¦¢ŁÖŠ£¬1mol B·¢Éś·“Ó¦Ź±×ŖŅʵē×ÓµÄĪļÖŹµÄĮæŹĒ ”£
£Ø3£©½«µ„ÖŹAŗĶCÓƵ¼ĻßĮ¬½Ó£¬ÓėĻ”ĮņĖį¹¹³ÉŌµē³Ų£¬ŌņøĆŌµē³ŲµÄøŗ¼«ŹĒ £ØĢīŌŖĖŲ·ūŗÅ£©£¬Õż¼«µÄµē¼«·“Ó¦Ź½ŹĒ ”£
£Ø4£©BÓė¹żĮæĻ”ĻõĖįĻąÓö£¬æÉÉś³ÉŅ»ÖÖĪŽÉ«ĘųĢ壬Š“³ö“Ė·“Ó¦µÄĄė×Ó·½³ĢŹ½
£»
Čō½«BČÜÓŚŃĪĖįÖŠæÉŠĪ³ÉČÜŅŗF£¬FÖŠ“ęŌŚµÄĮ½ÖÖ½šŹōŃōĄė×ÓŹĒ £ØĢīĄė×Ó·ūŗÅ£©”£
ĻÖÓŠČē ĻĀČżÖÖ¼ģŃéFÖŠµÄµĶ¼Ū½šŹōŃōĄė×ӵďµŃé·½°ø£Ø°üĄØŹµŃé²½Öč¼°Ō¤É菵ŃéĻÖĻ󣩣ŗ
ĻĀČżÖÖ¼ģŃéFÖŠµÄµĶ¼Ū½šŹōŃōĄė×ӵďµŃé·½°ø£Ø°üĄØŹµŃé²½Öč¼°Ō¤É菵ŃéĻÖĻ󣩣ŗ
·Ö±šČ”ÉŁĮæFČÜŅŗÓŚČżÖ§½ą¾»µÄŹŌ¹ÜÖŠ£¬
¢ŁĻņµŚŅ»Ö§ŹŌ¹ÜÖŠµĪ¼ÓKSCN ČÜŅŗ£¬ČÜŅŗ²»±äÉ«£¬ŌŁµĪ¼ÓĀČĖ®£¬ČÜŅŗ±äŗģÉ«£»
ČÜŅŗ£¬ČÜŅŗ²»±äÉ«£¬ŌŁµĪ¼ÓĀČĖ®£¬ČÜŅŗ±äŗģÉ«£»
¢ŚĻņµŚ¶žÖ§ŹŌ¹ÜÖŠČÜŅŗµÄµ×²æÓƵĪ¹Ü×¢Čė NaOHČÜŅŗ£¬Éś³É°×É«³Įµķ£¬Č»ŗóŃøĖŁ±äĪŖ»ŅĀĢÉ«£¬×īÖÕ±äĪŖŗģŗÖÉ«£»
NaOHČÜŅŗ£¬Éś³É°×É«³Įµķ£¬Č»ŗóŃøĖŁ±äĪŖ»ŅĀĢÉ«£¬×īÖÕ±äĪŖŗģŗÖÉ«£»
¢ŪĻņµŚČżÖ§ŹŌ¹ÜÖŠµĪ¼ÓĖįŠŌKMnO4ČÜŅŗ£¬Õńµ“£¬KMnO4ČÜŅŗ×ĻÉ«ĶŹČ„”£
ÉĻŹöČżÖÖ·½°øÖŠŗĻĄķµÄŹĒ £ØĢī¢Ł”¢¢Ś»ņ¢Ū£©£»
ČōĻņČÜŅŗFÖŠ¼ÓČėÉŁĮæĶ·Ū£¬Õńµ“ŗó£¬Ķ·ŪÖš½„Čܽā£¬ĒėŠ“³ö“Ė¹ż³ĢÖŠ·¢Éś·“Ó¦µÄĄė×Ó·½³ĢŹ½ 
 ӣ
ӣ

| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŌĶĮĄķ½ā
| ŹµŃé²½Öč | ĪŹĢā |
| 1“ÓUŠĶ¹Ü×ó¶Ė¼ÓČėĻ”ĻõĖįÖ±ÖĮ³äĀśUŠĶ¹ÜÓŅ¶Ė | ”Ī/ |
| 2ÓĆø½ÓŠĶĖæµÄ½ŗČūČū×”UŠĶ¹ÜÓŅ¶Ė£¬¹Ū²ģĻÖĻó | ĻÖĻóŹĒ ÓŠĪŽÉ«ĘųĢå²śÉś£¬ÓŅ±ßČÜŅŗÖš½„±ä³ÉĀĢÉ« ÓŠĪŽÉ«ĘųĢå²śÉś£¬ÓŅ±ßČÜŅŗÖš½„±ä³ÉĀĢÉ« |
| 3“ż·“Ó¦Ķ£Ö¹ŗó“ņæŖ½ŗČū£¬¹Ū²ģŹµŃéĻÖĻó | ĻÖĻóŹĒ ĪŽÉ«ĘųĢåÓėæÕĘų½Ó“„ŗóĮ¢¼“±ä³Éŗģ×ŲÉ« ĪŽÉ«ĘųĢåÓėæÕĘų½Ó“„ŗóĮ¢¼“±ä³Éŗģ×ŲÉ« |
| 10-3a©qV +0.5 |
| 0.14 |
| 10-3a©qV +0.5 |
| 0.14 |
| 1 |
| 2 |
|
|
| 1 |
| 2 |
|
|
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2013½ģ½ĖÕŹ”ŃĪ³ĒŹŠĆ÷“ļ֊ѧøßČżÉĻѧʌѧĒéµ÷ŃŠæ¼ŹŌ»ÆѧŹŌ¾ķ£Ø“ų½āĪö£© ĢāŠĶ£ŗĢīæÕĢā
(14 ·Ö)¹żŃõ»ÆĒāŗĶ³ōŃõ¶¼ŹĒ³£¼ūµÄĀĢÉ«Ńõ»Æ¼Į£¬ŌŚ¹¤ŅµÉś²śÖŠÓŠ×ÅÖŲŅŖµÄÓĆĶ¾”£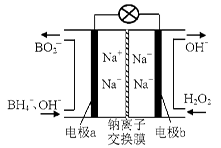
£Ø1£©¾Ż±ØµĄŅŌÅšĒā»ÆŗĻĪļNaBH4£ØBµÄ»ÆŗĻ¼ŪĪŖ+3¼Ū£©ŗĶH2O2×÷ŌĮĻµÄČ¼ĮĻµē³Ų£¬æÉÓĆ×÷ĶØŠÅĪĄŠĒµēŌ“”£øŗ¼«²ÄĮĻ²ÉÓĆPt/C£¬Õż¼«²ÄĮĻ²ÉÓĆMnO2£¬Ę乤×÷ŌĄķČēÓŅĶ¼ĖłŹ¾”£øƵē³Ų·ÅµēŹ±Õż¼«µÄµē¼«·“Ó¦Ź½ĪŖ£ŗ £»
ŅŌMnO2×öÕż¼«²ÄĮĻ£¬æÉÄÜŹĒŅņĪŖ ”£
£Ø2£©»š¼ż·¢Éä³£ŅŌŅŗĢ¬ėĀ£ØN2H4£©ĪŖČ¼ĮĻ£¬ŅŗĢ¬¹żŃõ»ÆĒā
ĪŖÖśČ¼¼Į”£ŅŃÖŖ£ŗ
N2H4(g) + O2(g) £½ N2(g) + 2H2O(g) ”÷H =" ØC" 534 kJ”¤mol-1
H2O2(l) = H2O(l) + 1/2O2(g) ”÷H =" ØC" 98.64 kJ”¤mol-1
H2O(l) = H2O(g) ”÷H=" +" 44kJ”¤mol-1
Ōņ·“Ó¦N2H4(g) + 2H2O2(l) = N2(g) + 4H2O(g) µÄ”÷H= ”£
£Ø3£©O3 æÉÓɳōŃõ·¢ÉśĘ÷£ØŌĄķČēÓŅĶ¼ĖłŹ¾£©µē½āĻ”ĮņĖįÖʵƔ£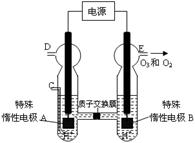
¢Ś Ķ¼ÖŠŅõ¼«ĪŖ £ØĢī”°A”±»ņ”°B”±£©”£
¢ŚČōC“¦ĶØČėO 2£¬ŌņA¼«µÄµē¼«·“Ó¦Ź½ĪŖ£ŗ ”£
¢ŪČōC“¦²»ĶØČėO 2 £¬D”¢E“¦·Ö±šŹÕ¼Æµ½15.68LŗĶÓŠ6.72LĘųĢå£Ø±ź×¼×“æöĻĀ£©£¬ŌņE“¦ŹÕ¼ÆµÄĘųĢåÖŠO2ŗĶO3µÄĢå»żÖ®±ČĪŖ £ØŗöĀŌ O 3 µÄ·Ö½ā£©”£
£Ø4£©ŠĀŠĶO3Ńõ»Æ¼¼Źõ¶ŌČ¼ĆŗŃĢĘųÖŠµÄNOxŗĶSO2ĶŃ³żŠ§¹ūĻŌÖų£¬¹ųĀÆŃĢĘųÖŠµÄNOx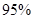 ŅŌÉĻŹĒŅŌNOŠĪŹ½“ęŌŚµÄ£¬æÉ·¢Éś·“Ó¦NO(g£©+ O3 (g)
ŅŌÉĻŹĒŅŌNOŠĪŹ½“ęŌŚµÄ£¬æÉ·¢Éś·“Ó¦NO(g£©+ O3 (g)  NO2(g)+ O2 (g)”£ŌŚŅ»¶ØĢõ¼žĻĀ£¬½«NOŗĶO3ĶØČė¾ųČČŗćČŻĆܱÕČŻĘ÷ÖŠ·¢ÉśÉĻŹö·“Ó¦ £¬Õż·“Ó¦ĖŁĀŹĖꏱ¼ä±ä»ÆµÄŹ¾ŅāĶ¼£ØČēĻĀĶ¼£©ĖłŹ¾”£ÓÉĶ¼æÉµĆ³öµÄÕżČ·Ėµ·ØŹĒ
NO2(g)+ O2 (g)”£ŌŚŅ»¶ØĢõ¼žĻĀ£¬½«NOŗĶO3ĶØČė¾ųČČŗćČŻĆܱÕČŻĘ÷ÖŠ·¢ÉśÉĻŹö·“Ó¦ £¬Õż·“Ó¦ĖŁĀŹĖꏱ¼ä±ä»ÆµÄŹ¾ŅāĶ¼£ØČēĻĀĶ¼£©ĖłŹ¾”£ÓÉĶ¼æÉµĆ³öµÄÕżČ·Ėµ·ØŹĒ 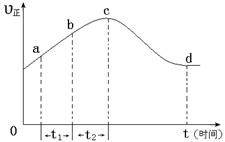
a£®·“Ó¦ŌŚcµć“ļµ½Ę½ŗāדĢ¬
b£®·“Ó¦ĪļÅØ¶Č£ŗbµćŠ”ÓŚcµć
c£®øĆ·“Ó¦ĪŖ·ÅČČ·“Ó¦
d£®¦¤t1£½¦¤t2Ź±£¬NOµÄ×Ŗ»ÆĮæ£ŗa”«b¶ĪŠ”ÓŚb”«c¶Ī
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2013½ģ½Ī÷Ź”ø߶žĻĀѧʌµŚ¶ž“ĪŌĀæ¼»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗĢīæÕĢā
£ØĆææÕ2·Ö£¬¹²14·Ö£©A”¢B”¢C”¢D¾łĪŖŗ¬±½»·µÄ»ÆŗĻĪļ£¬ĒŅĻą¶Ō·Ö×ÓÖŹĮæB£¾A£¾C£¬ŅŃÖŖÓŠ»śĪļAµÄ½į¹¹¼ņŹ½ĪŖ£ŗ 
(1)A¼ČÄÜŗĶNaOHČÜŅŗ·“Ó¦£¬ÓÖÄÜŗĶNaHCO3ČÜŅŗ·“Ó¦£¬Š“³öAŗĶ NaHCO3ČÜŅŗ·“Ó¦µÄ»Æѧ·½³ĢŹ½______ __________”£
(2)¢ŁÓŠ»śĪļBŌŚÅØĮņĖį“ęŌŚĢõ¼žĻĀ£¬Ļą»„×÷ÓĆÉś³ÉŅ»ÖÖ»·×“õ„(ČēĻĀĶ¼)ŌņÓŠ»śĪļBµÄ½į¹¹¼ņŹ½ĪŖ _”£

¢ŚµČĪļÖŹµÄĮæBÓėNa”¢NaOH”¢NaHCO3³ä·Ö·“Ó¦£¬ĻūŗÄNa”¢NaOH”¢NaHCO3µÄĪļÖŹµÄĮæÖ®±ČĪŖ___ _____”£
(3) 1molA”¢CĶźČ«Č¼ÉÕ£¬ĻūŗÄO2µÄÖŹĮæĻąµČ,ĒŅ1molCÄÜŗĶ1molNaĶźČ«·“Ó¦£¬Š“³öŗ¬Ģ¼Ō×ÓŹż×īÉŁµÄCµÄ½į¹¹¼ņŹ½_____ ___£¬Š“³öCÓėÅØäåĖ®µÄ·“Ó¦µÄ»Æѧ·½³ĢŹ½ ”£
(4)DŗĶB»„ĪŖĶ¬·ÖŅģ¹¹Ģ壬ĒŅÖŖĘä±½»·ÉĻĮ¬ÓŠČżøöČ”“ś»ł£¬±½»·ÉĻµÄŅ»ĀČ“śĪļÖ»ÓŠĮ½ÖÖ£¬D²»ÓėNaHCO3ČÜŅŗ·“Ó¦£¬ÄÜÓėNaŗĶNaOH·“Ó¦£¬µČÖŹĮæµÄDĻūŗÄNaŗĶNaOHµÄĪļÖŹµÄĮæÖ®±ČĪŖ2”Ć3£¬ŌņDµÄ½į¹¹¼ņŹ½ĪŖ ”¢ ”£(Ö»Š“¶žÖÖ)
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2010-2011ѧğ°²»ÕŹ”ŗĻ·ŹŹŠøßČżµŚ¶ž“Ī½Ģѧ֏Įæ¼ģ²āĄķ×Ū»Æѧ²æ·Ö ĢāŠĶ£ŗĢīæÕĢā
(14·Ö)X”¢Y”¢Z”¢WŹĒŌŖĖŲÖÜĘŚ±ķĒ°ĖÄÖÜĘŚÖŠµÄ³£¼ūŌŖĖŲ£¬ĘäŌ×ÓŠņŹżŅĄ“ĪŌö“ó”£XŌŖĖŲµÄŅ»ÖÖŗĖĖŲµÄÖŹĮæŹżĪŖ12,ÖŠ×ÓŹżĪŖ6;YŌŖĖŲŹĒ¶ÆÖ²ĪļÉś³¤²»æÉȱɣ”¢¹¹³Éµ°°×ÖŹµÄÖŲŅŖ×é³ÉŌŖĖŲ;ZµÄ»łĢ¬Ō×ÓŗĖĶā9øö¹ģµĄÉĻĢī³äĮĖµē×Ó,ĒŅŗĖĶāÓŠ2øöĪ“³É¶Ōµē×Ó£¬ÓėY²»Ķ¬×å;WŹĒŅ»ÖÖ³£¼ūŌŖĖŲ,æÉŅŌŠĪ³É3ÖÖŃõ»ÆĪļ,ĘäÖŠŅ»ÖÖŃõ»ÆĪļŹĒ¾ßÓŠ“ÅŠŌµÄŗŚÉ«¾§Ģ唣
(1) ³£ĪĀ³£Ń¹ĻĀZµ„ÖŹŹĒ_______¾§Ģå£ØĢīĄąŠĶ£©£¬Ī¢Į£¼äĶعż______ŠĪ³É¾§Ģå(ĢīĪ¢Į£¼äĻą»„×÷ÓƵĥąŠĶ£©”£Y2·Ö×ÓÖŠ“ęŌŚ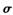 ¼üŗĶ
¼üŗĶ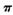 ¼üøöŹżÖ®±ČĪŖ____________”£
¼üøöŹżÖ®±ČĪŖ____________”£
(2) X-HŗĶY”ŖHŹōÓŚ¼«ŠŌ¹²¼Ū¼ü,ĘäÖŠ¼«ŠŌ½ĻĒæµÄ¼üŹĒ______(X”¢YÓĆŌŖĖŲ·ūŗűķŹ¾£©”£XµÄµŚŅ»µēĄėÄܱČYµÄ______(Ģī”°“ó”±»ņ”°Š””°£©”£
(3) Š“³öXµ„ÖŹÓėZµÄ×īøß¼ŪŃõ»ÆĪļ¶ŌÓ¦µÄĖ®»ÆĪļµÄÅØČÜŅŗ·“Ó¦µÄ»Æѧ·½³ĢŹ½____________________________________________________________
(4) WµÄ»łĢ¬Ō×ÓµÄĶāĪ§µē×ÓÅŲ¼Ź½ŹĒ____________”£
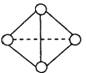
(5) ŅŃÖŖŅ»ÖÖ·Ö×ÓY4·Ö×Ó½į¹¹ČēÉĻĶ¼ĖłŹ¾£ŗ¶ĻĮŃ1molY”ŖYĪüŹÕ167KJµÄČČĮæ,Éś³É1molY Y·Å³ö942KJČČĮ攣ŹŌŠ“³öÓÉY4ĘųĢ¬·Ö×Ó±ä³ÉY2ĘųĢ¬·Ö×ÓµÄČČ»Æѧ·½³ĢŹ½________________________
Y·Å³ö942KJČČĮ攣ŹŌŠ“³öÓÉY4ĘųĢ¬·Ö×Ó±ä³ÉY2ĘųĢ¬·Ö×ÓµÄČČ»Æѧ·½³ĢŹ½________________________
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2010ÄźŗŚĮś½Ź”ø߶žĻĀŃ§ĘŚĘŚÄ©æ¼ŹŌ»ÆѧŹŌĢā ĢāŠĶ£ŗŹµŃéĢā
(17·Ö)ijæĪĶāŠ”×éÉč¼ĘµÄŹµŃéŹŅÖĘČ”ŅŅĖįŅŅõ„µÄ×°ÖĆČēĶ¼14-2ĖłŹ¾£¬AÖŠ·ÅÓŠÅØĮņĖį£¬BÖŠ·ÅÓŠŅŅ“¼”¢ĪŽĖ®“×ĖįÄĘ£¬DÖŠ·ÅÓŠ±„ŗĶĢ¼ĖįÄĘČÜŅŗ”£

ŅŃÖŖ¢ŁĪŽĖ®ĀČ»ÆøĘæÉÓėŅŅ“¼ŠĪ³ÉÄŃČÜÓŚĖ®µÄCaCl2”¤6 C2H50H
¢ŚÓŠ¹ŲÓŠ»śĪļµÄ·Šµć:
|
ŹŌ¼Į |
ŅŅĆŃ |
ŅŅ“¼ |
ŅŅĖį |
ŅŅĖįŅŅõ„ |
|
·Šµć/”ę |
34.7 |
78.5 |
118 |
77.1 |
µŚ5Ņ³
Ēė»Ų“š£ŗ
£Ø1£©ÅØĮņĖįµÄ×÷ÓĆŹĒ £»ČōÓĆĶ¬Ī»ĖŲ180Ź¾×Ł·ØČ·¶Ø·“Ó¦²śĪļĖ®·Ö×ÓÖŠŃõŌ×ÓµÄĢį¹©Õߣ¬Š“³öÄܱķŹ¾180Ī»ÖƵĻÆѧ·½³ĢŹ½£ŗ ”£
£Ø2£©ĒņŠĪøÉŌļ¹ÜCµÄ×÷ÓĆŹĒ ”£Čō·“Ó¦Ē°ĻņDÖŠ¼ÓČė¼øµĪ·ÓĢŖ£¬ČÜŅŗ³ŹŗģÉ«£¬²śÉś“ĖĻÖĻóµÄŌŅņŹĒ(ÓĆĄė×Ó·½³ĢŹ½±ķŹ¾) £»·“Ó¦½įŹųÕšµ“¾²Ö¹ŗóDÖŠµÄĻÖĻóŹĒ £®
£Ø3£©“ÓDÖŠ·ÖĄė³öµÄŅŅĖįŅŅõ„ÖŠ³£ŗ¬ÓŠŅ»¶ØĮæµÄŅŅ“¼”¢ŅŅĆŃŗĶĖ®£¬Ó¦ĻČ¼ÓČėĪŽĖ®ĀČ»ÆøĘ£¬·ÖĄė³ö £»ŌŁ¼ÓČė(“ĖæÕ“ÓĻĀĮŠŃ”ĻīÖŠŃ”Ōń) £¬Č»ŗó½ųŠŠÕōĮó£¬ŹÕ¼Æ77”ę×óÓŅµÄĮó·Ö£¬ŅŌµĆ½Ļ“æ¾»µÄŅŅĖįŅŅõ„”£
A£®ĪåŃõ»Æ¶žĮ× B£®¼īŹÆ»Ņ C£®ĪŽĖ®ĮņĖįÄĘ D£®ÉśŹÆ»Ņ
£Ø4£©ÓŠĶ¬Ń§ÄāĶعżŗģĶā¹āĘ×ŅĒ¼ų¶ØĖłµĆ²śĪļÖŠŹĒ·ńŗ¬ÓŠ”°£CH2CH3”±£¬ Ą“Č·¶Øø±²śĪļÖŠ“ęŌŚŅŅĆŃ”£ĒėÄć¶ŌøĆĶ¬Ń§µÄ¹Ūµć½ųŠŠĘĄ¼Ū£ŗ ”£
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com