
��ˮ�Ƿḻ����Դ���⣬�Ӻ�ˮ����ȡԪ���ǻ�ѧ��ҵ����Ҫ��ɲ��֡�
��1�����ξ��ƾ��dz�ȥ���е�Ca2+��Fe3+��SO42-����ɳ�����ʣ��������Լ��У���Na2CO3��Һ ��HCl�����ᣩ ��Ba��OH��2��Һ���������Լ�������˳����_________������ţ���
��2�����������С���ˮ���塱����ȡ�嵥�ʷ�Ӧ�����ӷ���ʽΪ��__________��
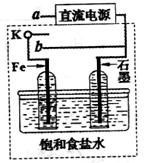
��3��ijͬѧ�������ͼװ�ý������µ绯ѧʵ�顣
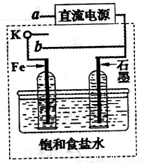
�ٵ�����K��a����ʱ�������������ݲ�����������Ϊ_______����
��һ��ʱ���ʹ����K��a�Ͽ�����b����ʱ�����߿��ڵ�װ�ÿɳ�Ϊ__________����д����ʱFe�缫�ϵĵ缫��Ӧʽ_________________��
��4��ij������ʢ��CaSO4����Һ�ķ�Ӧ����ͨ�백������ȡ���ʣ�NH4��2SO4��Ч�����á���ͨ��CO2��������������NH4��2SO4���������ԭ��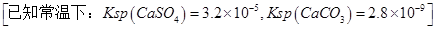 ��
��
��1���ۢ٢ڣ�2�֣�
��2��Cl2��2Br��=2Cl����Br2��2�֣�
��3����Fe����������2�֣�
��ԭ��أ�2�֣���Fe��2e����2OH��= Fe(OH)2����3�֣�
��4��CO2���ڰ�ˮ��������CO32-��CO2+2NH3��H2O =��NH4��2CO3+ H2O��1�֣� ��CO32-������Ʒ�Ӧ�����ܶȻ�������С��̼��Ƴ�����CaSO4��s����CO32-��aq��= CaCO3 ��s����SO42-��aq����2�֣���ʹ����Ƶij����ܽ�ƽ�����ܽ�ķ����ƶ���3�֣����Ӷ�����������NH4�� 2SO4������3�֣�
��������
�����������1��Na2CO3�ȳ�ȥCa2+������ȥ������Ba��OH��2��������Na2CO3���������������˳��Ϊ���ۢ٢ڡ�
��3������Ϊ���缫�������ݲ�����Fe�缫ֻ����������
�ڿ���K��a�Ͽ�����b����ʱ���γ�ԭ��ء�
��4���������Ա�Ksp���Կ���CaCO3�����ܣ�����ͨ��CO2�백ˮ���ɣ�NH4��2CO3��CO32‾ʹCaSO4ת��Ϊ��NH4�� 2SO4��
���㣺 ���⿼���˳����Լ���ѡ�����ӷ���ʽ����д���绯ѧ�ͳ����ܽ�ƽ�⡣


 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ��2013?�Ͳ���ģ����ˮ�Ƿḻ����Դ���⣬�Ӻ�ˮ����ȡԪ���ǻ�ѧ��ҵ����Ҫ��ɲ��֣�
��2013?�Ͳ���ģ����ˮ�Ƿḻ����Դ���⣬�Ӻ�ˮ����ȡԪ���ǻ�ѧ��ҵ����Ҫ��ɲ��֣��鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013-2014ѧ�����ʡ�����и�����һ��ģ�⿼�����ۻ�ѧ�Ծ��������棩 ���ͣ������
������һ���ḻ����Դ���⣬ͨ����ˮ���ۺ����ÿɻ���������ʹ�����ʹ�á�
��1�� ��ˮ���εĿ������ã�
��.��ˮ����Ŀǰ�����Ϊ�������������ѡ��Զ�뽭���뺣�ڣ�������꣬��ϫ��������ƽ̹�տ��ĺ�̲�����������Ϊ��ˮ�ء������غ�_______�ء�
II.Ŀǰ��ҵ�ϲ��ñȽ��Ƚ������ӽ���Ĥ���۷������ȼҵ�������ڵ����������ӽ���Ĥֻ����������ͨ������ֹ�����Ӻ�����ͨ������˵���ȼ������������ӽ���Ĥ������____________________________________________����дһ�㼴�ɣ�
��2�� ���������ǽ�������չ������һ�ֽϺõĺ�ˮ������������ԭ������ͼ��ʾ����ش��������⣺
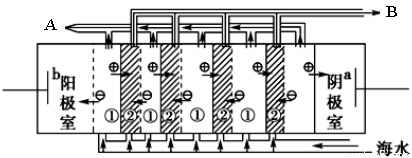
��.��ˮ����ֱ��ͨ�뵽��װ���У�������_____________________________________________��
��. B���ų�����________(������ˮ������Ũˮ��)��
��3�� �ÿ�±����Na+��K+��Mg2+��Cl-��Br-�����ӣ�����ȡ�壬�������������£�
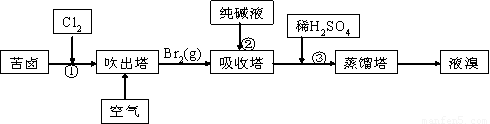
��.���������е���Һ��BrO3�������������з�Ӧ�����ӷ���ʽΪ�ߣߣߣߣߣߣߣߣߣߣߣߣ�
_________________________________________��
��.ͨ�����Ȼ��ѻ�ú�Br2����Һ��Ϊ�λ��辭�����������ա��ữ�����»�ú�Br2����Һ��_____________________________________________________________________��
��.����������ͨ��ˮ�������ȣ������¶���900C���ҽ��������ԭ����___________________________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��ˮ�Ƿḻ����Դ���⣬�Ӻ�ˮ����ȡԪ���ǻ�ѧ��ҵ����Ҫ��ɲ��֡�
��1�����ξ��ƾ��dz�ȥ���е�Ca2+��Fe3+��SO42-����ɳ�����ʣ��������Լ��У���Na2CO3��Һ ��HCl�����ᣩ ��Ba��OH��2��Һ���������Լ�������˳����_________������ţ���
��2�����������С���ˮ���塱����ȡ�嵥�ʷ�Ӧ�����ӷ���ʽΪ��__________��
��3��ijͬѧ���������ͼװ�ý������µ绯ѧʵ�顣
�ٵ�����K��a����ʱ�������������ݲ�����������Ϊ_______����
��һ��ʱ���ʹ����K��a�Ͽ�����b����ʱ�����߿��ڵ�װ�ÿɳ�Ϊ__________����д����ʱFe�缫�ϵĵ缫��Ӧʽ_________________��
��4��ij������ʢ��CaSO4����Һ�ķ�Ӧ����ͨ�백������ȡ���ʣ�NH4��2SO4��Ч�����á���ͨ��CO2��������������NH4��2SO4���������ԭ��![]() ��
��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013��ɽ��ʡ�Ͳ��и߿���ѧ��ģ�Ծ��������棩 ���ͣ������
 ��
��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com