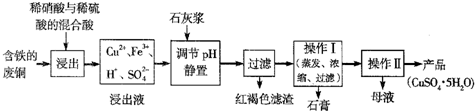
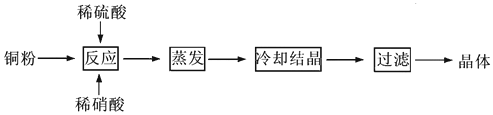
CuSO4��5H2O��ͭ����Ҫ��������Ź㷺��Ӧ�á�������CuSO4��5H2O��ʵ�����Ʊ�����ͼ��
�����������������գ�
��1�������ϣ�Ϊ���Ƶô�����CuSO4��5H2O���壬��Ҫ����ϡ���ᡢϡ�����������ʵ���֮��Ϊ__________��������Ӧ�����ӷ���ʽΪ____________________��
��2��Ϊ�ӿ�����ٶȣ��ɲ���__________���������ƣ����ò�����ϴ�ӳ���ʱ����Ҫע��ϴ�Ӽ���ѡ�����������Ӧ____________________
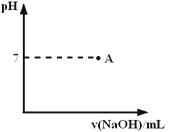
��3��ʵ�����������ж�����ϡ�����Ũ�ȿ���Ҫ��Ƚϸߣ�ͨ���ñ�����������Һ���ζ����ζ����������÷�̪��ָʾ�����յ�������____________________��������ͼ�л����ζ���������Һ��pH�����μ�����������Һ����ı仯������ͼ��Ҫ���A�㣩��
��4������ʹ�õı�����������Һ�Ѿ�ͨ�������ʵı궨������������ͨ���������궨��Һ�Ļ�������__________ ��
A������ B������ C�������� D������
��5���ö��Ե缫���һ��Ũ�ȵ�����ͭ��Һ��ͨ��һ��ʱ��������õ���Һ�м���0.1mol Cu2(OH)2CO3��ǡ�ûָ������ǰ��Ũ�Ⱥ�pH(������CO2���ܽ�)����������й�ת�Ƶ��ӵ����ʵ���Ϊ__________��

 �ִʾ�ƪ��ͬ�����Ĵ��ϵ�д�
�ִʾ�ƪ��ͬ�����Ĵ��ϵ�д� �߽�������ϵ�д�
�߽�������ϵ�д�