
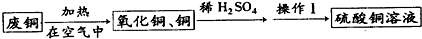



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��.��һС����г��ϵĵ��ν������о����ڵ��������KIO3��ʽ���ڡ�������������⣺
��1����ȷʳ�õ��β�����������ã���С�����ըʳƷ�Ƿ����˷ŵ�����˿����о���
����Э���ⶨ�������ƣ�______________________________��
���о��ɹ���������ʽ�ǣ�______________________________��
��2��������������к��е⣿���ɹ�ѡ����Լ��У�ϡ���ᡢϡ���ᡢKCl��KI������Һ��ʯ����Һ�������ӷ�Ӧ����ʽ��ʾ______________________________��
��.�ڶ�С�����÷�ͭ��ȡCuSO4��
��1����ѧ������������ʵ�飺ȡ��6.4 gͭ�ķ�ͭ��10 mL 18 mol��L-1Ũ�������Բ����ƿ�й��ȣ�ֱ����Ӧ��ϣ��������ƿ�л���ͭƬʣ�ࡣ�������Լ���ѧ�Ļ�ѧ֪ʶ����Ϊ��ƿ�л��н϶������ʣ�ࡣ����Ϊ�����ж��Ƿ���ȷ___________����ǡ������������ǣ�_____________________________________________________��
��2����ͬѧ��Ϊ����Ƶ�ʵ�鷽�����ã����Լ���Ƶ�˼·�ǣ�
2Cu+O2![]() 2CuO��CuO+H2SO4====CuSO4+H2O���Աȼķ�������ͬѧ�������ŵ��ǣ�
2CuO��CuO+H2SO4====CuSO4+H2O���Աȼķ�������ͬѧ�������ŵ��ǣ�
_____________________________________________________________________��
��3��ij���зḻ�ĵ���Դ��Ϊ�˾߱���ͬѧ���ŵ㣬���ܲ��ܸ��ݸõص���Դ����Ҳ���һ����ͭ��������ȡCuSO4�ķ�����������������ѡ��_______________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ��ʴ���
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2009���㽭ʡ�߿���ѧģ���Ծ����ģ��������棩 ���ͣ������
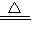 2CuO��CuO+H2SO4=CuSO4+H2O��
2CuO��CuO+H2SO4=CuSO4+H2O���鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com