
����������CO2���ڻ��ۣ���в����̬����������������ܵ�ȫ�����ע��
��1����ҵ�ϳ��ø�Ũ�ȵ�K2CO3��Һ����CO2������ҺX�������õ�ⷨʹK2CO3��Һ��������װ��ʾ��ͼ���£�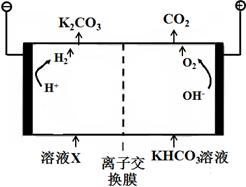
�������������ķ�Ӧ���� ��H ++ HCO3-=H2O+CO2����
����CO32-��������������ԭ�� ��
��2������װ���в�����CO2��H2��һ�������·�Ӧ���ɼ״��Ȳ����ҵ�����ø÷�Ӧ�ϳɼ״���
��֪��25 �棬101 KPa�£�
H2(g)+1/2 O2(g)=H2O(g) �� H1=" -242" kJ/mol
CH3OH(g)+3/2 O2(g)=CO2 (g)+2 H2O(g) �� H2=" -676" kJ/mol
д��CO2��H2������̬�״��Ȳ�����Ȼ�ѧ����ʽ ��
�����ʾ�ϳɼ״��ķ�Ӧ�������仯ʾ��ͼ��������ȷ���� ������ĸ��ţ���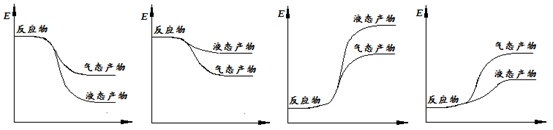
a b c d
��3������ȼ�ϵ����һ���������ォ��ѧ��ֱ��ת���ɵ��ܵ�װ�á���֪ij�ּ״�
����ȼ�ϵ���У��������ҺΪ���ԣ�ʾ��ͼ���£�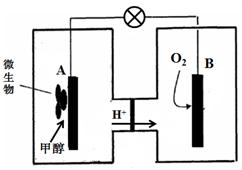
�õ�����·���ӵ���������Ϊ ����д����A��B����B��A������
����������B�缫����Һ��pH�빤��ǰ��Ƚ� ����д����������С�����䡱����Һ����仯���Բ��ƣ���
A�缫�����״������ĵ缫��ӦʽΪ ��
��1����4OH--4e-=2H2O+O2����2�֣�
�ڴ�1��HCO3 �C���ڵ���ƽ�⣺HCO3 �C H++CO32- ��1�֣�������H+�ŵ�Ũ�ȼ�Сƽ�����ƣ�1�֣�CO32-����
H++CO32- ��1�֣�������H+�ŵ�Ũ�ȼ�Сƽ�����ƣ�1�֣�CO32-����
��2������H+�ŵ�OH-Ũ������1�֣���OH-��HCO3 �C��Ӧ����CO32-��1�֣�CO32-����
��2����CO2(g)+3H2(g)=CH3OH(g)+H2O(g) ��H="-50" kJ/mol��2�֣� �� a��2�֣�
��3���ٴ�A��B��1�֣� �ڲ��䣨1�֣�
��CH3OH+H2O - 6e-=6H++ CO2����2�֣�
���������������1����������H2O�������OH?ʧ���ӣ��缫����ʽΪ��4OH - - 4e-=2H2O+O2��
��������H+�ŵ緢���õ��ӷ�Ӧ:2H++2e?=H2��,HCO3?���ڵ���ƽ�⣺HCO3? CO32?+H+��H+Ũ�ȼ�С��ʹHCO3?����ƽ�������ƶ���CO32?Ũ�������������
CO32?+H+��H+Ũ�ȼ�С��ʹHCO3?����ƽ�������ƶ���CO32?Ũ�������������
��2��������д��CO2��H2��Ӧ���ɼ״��Ļ�ѧ����ʽ����ע��״̬��Ȼ����ݸ�˹������?H=3?H1��?H2= -50 kJ?mol?1�������Ȼ�ѧ����ʽΪ��CO2(g)+3H2(g)=CH3OH(g)+H2O(g) ��H="-50" kJ?mol?1��
�ڸ÷�ӦΪ���ȷ�Ӧ��������ΪҺ̬���ų����������࣬����a��ͼ����ȷ��
��3�� �ټ״�ʧȥ���ӣ�Ϊ��صĸ��������Ըõ�����·���ӵ���������Ϊ��A��B��
��B�缫��O2�õ�������H+��ͬʱ��Һ�е�H+����B�缫�ң�����B�缫����Һ��pH�빤��ǰ��Ƚ����䡣
��CH3OHʧ���ӣ�����CO2��H+�����ݻ��ϼ۱仯��Ԫ���غ���ƽ����ʽ��CH3OH+H2O - 6e-=6H++ CO2��
���㣺���⿼����ԭ����ԭ���ԭ��������ʽ����д��ƽ���ƶ����Ȼ�ѧ����ʽ����д��


 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��Դ�Ŀ����������������Ŀɳ�����չϢϢ��ء�
��.��֪��Fe2O3(s)��3C(s)=2Fe(s)��3CO(g) ��H1��a kJ��mol��1
CO(g)��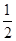 O2(g)=CO2(g)�� ��H2��b kJ��mol��1
O2(g)=CO2(g)�� ��H2��b kJ��mol��1
4Fe(s)��3O2(g)=2Fe2O3(s)�� ��H3��c kJ��mol��1
��C��ȼ���Ȧ�H��________ kJ��mol��1��
��.(1)����ԭ��صĹ���ԭ�������л�ѧ��Ӧ�������Ͽ�����Ƴ�ԭ��ص���________(�����)��
A��C(s)��CO2(g)=2CO(g)
B��NaOH(aq)��HCl(aq)=NaCl(aq)��H2O(l)
C��2H2O(l)=2H2(g)��O2(g)
D��2CO(g)��O2(g)=2CO2(g)
�������ڵ�K2CO3��CO2Ϊ��Ӧ�Ļ�����������ѡ��Ӧ��Ƴ�һ��ԭ��أ���д����ԭ��صĸ�����Ӧ��______________________________________
_______________________________________________________________��
(2)ijʵ��С��ģ�ҵ�ϳɰ���ӦN2(g)��3H2(g) 2NH3(g)����H����92.4 kJ��mol��1����ʼ���ǽ�N2��H2�������20 mol(�����1��1)����5 L�ϳ����У���ӦǰѹǿΪp0����Ӧ������ѹǿ��p��ʾ����Ӧ������
2NH3(g)����H����92.4 kJ��mol��1����ʼ���ǽ�N2��H2�������20 mol(�����1��1)����5 L�ϳ����У���ӦǰѹǿΪp0����Ӧ������ѹǿ��p��ʾ����Ӧ������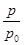 ��ʱ��t�Ĺ�ϵ��ͼ��ʾ��
��ʱ��t�Ĺ�ϵ��ͼ��ʾ��
��ش��������⣺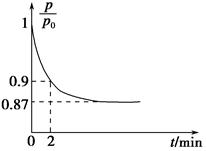
�ٷ�Ӧ��ƽ��ı�־��(����ĸ����)________��
A��ѹǿ���ֲ���
B�������ܶȱ��ֲ���
C��NH3������������N2���������ʵ�2��
��0��2 min�ڣ���c(N2)�仯��ʾ��ƽ����Ӧ����Ϊ________��
�������N2��ת���ʣ��ɲ�ȡ�Ĵ�ʩ��________��
A������ϵ�а������1��1�ٳ���N2��H2
B�������NH3
C�������¶�
D�����뺤��ʹѹǿ����
E������һ������N2
(3)25 ��ʱ��BaCO3��BaSO4���ܶȻ������ֱ���8��10��9��1��10��10��ij����BaCO3����������Һ�У�c(CO32��)��0.2 mol��L��1���������������Na2SO4��Һ����Ҫ����BaSO4����������Na2SO4��Һ�����ʵ���Ũ����С��________ mol��L��1��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ҵ�ϳ�����CO��H2�ϳɿ�������Դ�״���
��1����֪CO(g)��CH3OH(l)��ȼ���ȷֱ�Ϊ283.0 kJ��mol-1��726.5 kJ��mol-1����CH3OH(l)����ȫȼ������CO(g)��H2O(l)���Ȼ�ѧ����ʽΪ ��
��2���ϳɼ״��ķ���ʽΪCO(g)+2H2(g) CH3OH(g) ��H <0��
CH3OH(g) ��H <0��
��230 ��?270 ����Ϊ������Ϊ�о��ϳ�������ʵ���ʼ��ɱ�n(H2):n(CO)���ֱ���230 �桢250 ���270 �����ʵ�飬���������ͼ��ʾ������270 ���ʵ��������Ӧ��������_____������ĸ����������X��Y��Z��Ӧ��Ͷ�ϱȴﵽ��ͬ��COƽ��ת����ʱ����Ӧ�ķ�Ӧ�¶���Ͷ�ϱȵĹ�ϵ�� ��
��3����Ͷ�ϱ�Ϊ1��1���¶�Ϊ230 �棬ƽ���������У�CH3OH�����ʵ�������Ϊ ������1λС������ƽ��ʱCO��ת���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�Դ�����Ⱦ��SO2��NOx�����о�������Ҫ�������塣��ش��������⣺
��1��Ϊ����SO2���ŷţ�����ȡ�Ĵ�ʩ�ǽ�úת��Ϊ�������ȼ�ϡ�
��֪��H2��g���� O2��g����H2 O��g�� ��H����241.8kJ��mol��1
O2��g����H2 O��g�� ��H����241.8kJ��mol��1
C��s���� O2��g����CO��g�� ��H����110.5kJ��mol��1
O2��g����CO��g�� ��H����110.5kJ��mol��1
д����̿��ˮ������Ӧ���Ȼ�ѧ����ʽ�� ��
��2����֪����������NO�ķ�ӦΪ��N2��g����O2��g�� 2NO��g�� ��H
2NO��g�� ��H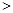 0����1.0 mol������0.80 mol N2��0.20 mol O2��1300oCʱ��1.0 L�ܱ������ھ���5s��Ӧ�ﵽƽ�⣬���NOΪ8.0��10��4 mol��
0����1.0 mol������0.80 mol N2��0.20 mol O2��1300oCʱ��1.0 L�ܱ������ھ���5s��Ӧ�ﵽƽ�⣬���NOΪ8.0��10��4 mol��
��5s�ڸ÷�Ӧ��ƽ�����ʦͣ�NO�� �� ������2λ��Ч���֣�����1300oC ʱ���÷�Ӧ��ƽ
�ⳣ������ʽK�� ��
�����������������¶�Խ�ߣ���λʱ����NO�ŷ���Խ��ԭ���� ��
��3������β����NO��CO��ת��������������һ��ʱ�������������ı���������ѧ��Ӧ���ʡ���ͼ��ʾ��������������ʱ����Ӧ2NO��g����2CO��g�� 2CO2��g����N2��g�� �У�NO��Ũ��
2CO2��g����N2��g�� �У�NO��Ũ��
c��NO�����¶ȣ�T���������������S����ʱ�䣨t���ı仯���ߡ�
�ٸ÷�Ӧ�ġ�H 0 ���������������
���������ı����S1��S2������ͼ�л���c��NO�� ��T1��S2�����´ﵽƽ������еı仯���ߣ�������Ӧ��ע����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���ڹ�����ռ����Ҫ��λ��
��1����ҵ�ϳɰ�ʱ���ϳ�����ÿ����1 mol NH3���ų�46.1 kJ��������
�� ��ҵ�ϳɰ����Ȼ�ѧ����ʽ�� ��
�� ��֪��
N2 (g)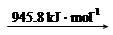 2N (g)
2N (g)
H2 (g)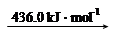 2H (g)
2H (g)
��Ͽ�1 mol N��H�������������_______kJ��
��2���±��ǵ���Ӧ���а�n(N2):n(H2)=1:3Ͷ�Ϻ���200�桢400�桢600���£���Ӧ�ﵽƽ��ʱ���������NH3�����ʵ���������ѹǿ�ı仯���ߡ�
�� ����a��Ӧ���¶��� ��
�� ���ڹ�ҵ�ϳɰ��ķ�Ӧ������������ȷ���� ������ĸ����
A. ��ʱ�����NH3�������H2��ƽ��ת����
B. �Ӵ����ܼӿ췴Ӧ���������H2��ƽ��ת����
C. ��ͼ��M��N��Q��ƽ�ⳣ��K�Ĵ�С��ϵ��K(M)=" K(Q)" >K(N)
�� M���Ӧ��H2ת������ �� ��3������һ��DZ�ڵ������Դ������������ȼ�ϵ�ص�ȼ�ϡ���ص��ܷ�ӦΪ��
��3������һ��DZ�ڵ������Դ������������ȼ�ϵ�ص�ȼ�ϡ���ص��ܷ�ӦΪ��
4NH3(g) + 3O2(g) = 2N2(g) + 6H2O(g)��
���ȼ�ϵ�صĸ�����Ӧʽ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
I����֪����Ӧ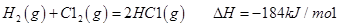
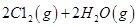

��ش�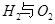 ��Ӧ������̬ˮ���Ȼ�ѧ����ʽ__________________________
��Ӧ������̬ˮ���Ȼ�ѧ����ʽ__________________________
II��ʵʩ�Լ�����Դ�˷Ѻͽ��ͷ����ŷ�Ϊ�������ݵĽ��ܼ������ߣ���Ӧ��ȫ���������⡢������Դ��Լ�͡������Ѻ������ı�Ȼѡ����ҵ�ķ�չ������Ϲ��ҽ��ܼ��ŵ�����Ҫ����������ѧ֪ʶ������������⣺
��1����֪ij��Ӧ��ƽ�����ʽΪ��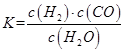
������Ӧ�Ļ�ѧ����ʽΪ��_________________________________________________
��2����֪��400��ʱ����Ӧ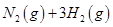
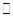
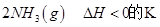 =0��5����400��ʱ����0��5L�ķ�Ӧ�����н��кϳɰ���Ӧ��һ��ʱ����N2��H2��NH3�����ʵ����ֱ�Ϊ2mol��1mol��2mol�����ʱ��Ӧ
=0��5����400��ʱ����0��5L�ķ�Ӧ�����н��кϳɰ���Ӧ��һ��ʱ����N2��H2��NH3�����ʵ����ֱ�Ϊ2mol��1mol��2mol�����ʱ��Ӧ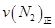 ____________
____________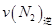 �������������������ȷ������
�������������������ȷ������
��ʹ�ø÷�Ӧ�Ļ�ѧ��Ӧ���ʼӿ죬ͬʱʹƽ��ʱNH3������������ӣ�
�ɲ�ȡ����ȷ��ʩ��____________������ţ�
A���Ӵ��� B�������¶� C����С�������ѹǿ D��ʹ����Һ������
��3����һ��������ܱ������У��������»�ѧ��Ӧ��
 ���仯ѧƽ�ⳣ��K���¶�T�Ĺ�ϵ���±���
���仯ѧƽ�ⳣ��K���¶�T�Ĺ�ϵ���±���
������������⣺
���жϸ÷�Ӧ�� ___________0�������������
___________0�������������
����һ�������£����жϸ÷�Ӧһ���ﻯѧƽ��״̬����___________������ţ���
A�� B��������ѹǿ���ֲ���
B��������ѹǿ���ֲ���
C��A��B��ת������� D�����������ܶȱ��ֲ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��1����ش��������⣺
����֪����CO(g) + 1/2O2(g) = CO2(g) ��H= ��283.0kJ��mol��1
��CH3OH(l) + 3/2O2(g) = CO2(g)+2H2O(l) ��H= ��726.5kJ��mol��1
��д���״�����ȫȼ������һ����̼��Һ̬ˮ���Ȼ�ѧ����ʽ��____________________________________________________________________��
����֪��1molH��H����1molCl��Cl����1molH��Cl���ֱ���Ҫ��������436kJ��243kJ��432kJ����Ӧ��Ӧ��H2(g)+ Cl2(g)=2HCl (g) �ġ�H= ��
��2����֪25�桢101 kPa�£�ϡ��ǿ����ϡ��ǿ����Һ��Ӧ���к���Ϊ -57.3 kJ/mol��
�����ʾϡ������ϡ�ռ���Һ�кͷ�Ӧ���Ȼ�ѧ����ʽΪ�� ��
�ڲⶨ�к���ʵ��ʱ����IJ����������ձ�����Ͳ�� �� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��������ЧӦ����Դ��ȱ�����⣬��ν��ʹ����е�CO2���������Կ������ã������˸������ձ����ӡ�Ŀǰ��ҵ����һ�ַ�������CO2����ȼ�ϼ״���һ�������·�����Ӧ��
CO2(g)+3H2(g) CH3OH(g)+H2O(g)��ͼ1��ʾ�÷�Ӧ����������(��λΪkJ��mol��1)�ı仯��
CH3OH(g)+H2O(g)��ͼ1��ʾ�÷�Ӧ����������(��λΪkJ��mol��1)�ı仯��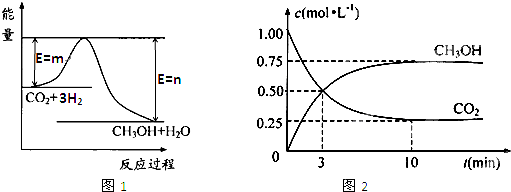
��1��д���÷�Ӧ���Ȼ�ѧ����ʽ ��
��2�����ڸ÷�Ӧ������˵���У���ȷ���� ��
A����H��0����S��0 B����H��0����S��0 C����H��0����S��0 D����H��0����S��0
��3���÷�Ӧ��ƽ�ⳣ��K�ı���ʽΪ�� ��
��4���¶Ƚ��ͣ�ƽ�ⳣ��K ������� �����䡱��С������
��5��Ϊ̽����Ӧԭ�����ֽ�������ʵ�飺�����Ϊ1 L���ܱ������У�����1 molCO2��3 molH2�����CO2��CH3OH(g)��Ũ����ʱ��仯��ͼ2��ʾ���ӷ�Ӧ��ʼ��ƽ�⣬������Ũ�ȱ仯��ʾ��ƽ����Ӧ����v (H2)�� ��
��6�����д�ʩ����ʹ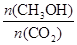 ������� ��
������� ��
A�������¶�
B���������
C����H2O(g)����ϵ�з���
D��������䣬����He(g)ʹ��ϵ��ѹǿ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��֪H2��g����CO��g����ȼ���ȡ�H�ֱ�Ϊ-285.8kJ��mol-1��-283.0kJ��mol��
��һ����̼��Һ̬ˮ��Ӧ�����ɶ�����̼���������Ȼ�ѧ����ʽΪ
________________________________________________________��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com