
ĪŖŃŠ¾æĶÓėÅØĮņĖįµÄ·“Ó¦£¬Ä³»ÆѧŠĖȤŠ”×é½ųŠŠČēĻĀŹµŃ锣
ŹµŃé¢ń£ŗ·“Ó¦²śĪļµÄ¶ØŠŌĢ½¾æ”£
ŹµŃé×°ÖĆČēĶ¼ĖłŹ¾£ŗ£Ø¹Ģ¶Ø×°ÖĆŅŃĀŌČ„£©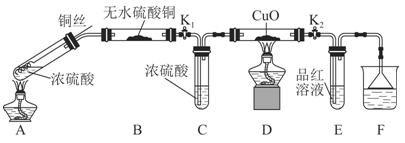
£Ø1£©AÖŠ·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ ”£
£Ø2£©FÉÕ±ÖŠµÄČÜŅŗĶس£ŹĒ ”£
£Ø3£©ŹµŃé¹ż³ĢÖŠ£¬ÄÜÖ¤Ć÷ÅØĮņĖįÖŠĮņŌŖĖŲµÄŃõ»ÆŠŌĒæÓŚĒāŌŖĖŲµÄĻÖĻóŹĒ
ӣ
£Ø4£©ŹµŃé½įŹųŗó£¬Ö¤Ć÷A×°ÖĆŹŌ¹ÜÖŠ·“Ó¦ĖłµĆ²śĪļŹĒ·ńŗ¬ÓŠĶĄė×ӵIJŁ×÷·½·ØŹĒ ”£
£Ø5£©ĪŖĖµĆ÷ÅØĮņĖįÖŠµÄĖ®ŹĒ·ńÓ°ĻģB×°ÖĆĻÖĻóµÄÅŠ¶Ļ£¬»¹Šė½ųŠŠŅ»“ĪŹµŃ锣ŹµŃé·½°øĪŖ ”£
ŹµŃé¢ņ£ŗ·“Ó¦²śĪļµÄ¶ØĮæĢ½¾æ
£Ø6£©ŌŚĶÓėÅØĮņĖį·“Ó¦µÄ¹ż³ĢÖŠ£¬·¢ĻÖÓŠŗŚÉ«ĪļÖŹ³öĻÖ£¬¾²éŌÄĪÄĻ×»ńµĆĻĀĮŠ×ŹĮĻ”£
׏ĮĻ1£ŗ
| ĮņĖį/mol”¤L£1 | ŗŚÉ«ĪļÖŹ³öĻÖµÄĪĀ¶Č/”ę | ŗŚÉ«ĪļÖŹĻūŹ§µÄĪĀ¶Č/”ę |
| 15 | Ō¼150 | Ō¼236 |
| 16 | Ō¼140 | Ō¼250 |
| 18 | Ō¼120 | ²»ĻūŹ§ |
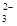 £«I2===S4O
£«I2===S4O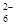 £«2I££©
£«2I££© £Ø1£©
£Ø2£©ĒāŃõ»ÆÄĘČÜŅŗ£Ø»ņĖįŠŌøßĆĢĖį¼ŲČÜŅŗ£©
£Ø3£©D×°ÖĆÖŠŗŚÉ«¹ĢĢåŃÕÉ«ĪŽ±ä»Æ£¬EÖŠČÜŅŗĶŹÉ«
£Ø4£©½«A×°ÖĆÖŠŹŌ¹ÜÄŚĄäČ“µÄ»ģŗĻĪļŃŲ±±Ś£Ø»ņ²£Į§°ō£©»ŗ»ŗµ¹ČėŹ¢ÓŠĖ®µÄÉÕ±ÖŠ£¬²¢²»¶Ļ½Į°č£¬æ“ČÜŅŗŹĒ·ń±äĄ¶
£Ø5£©Ź¹ÓĆÉĻŹö×°ÖĆ£¬²»·ÅČėĶĖæ½ųŠŠŹµŃ飬¹Ū²ģĪŽĖ®ĮņĖįĶŹĒ·ń±äĄ¶
£Ø6£©Abd
£Ø7£©64bV/a%
½āĪöŹŌĢā·ÖĪö£ŗ£Ø1£©¾ßÓŠĒæŃõ»ÆŠŌ£¬Ņņ“ĖÅØĮņĖįæÉŅŌÓėĶ·“Ӧɜ³ÉĮņĖįĶ£¬¶žŃõ»ÆĮņŗĶĖ®”£
£Ø2£©×°ÖĆÖŠ»įÉś³É¶žŃõ»ÆĮņĪŪČ¾ŠŌĘųĢ壬²»ÄÜÅŷŵ½æÕĘųÖŠ£¬ŠčŅŖÓĆĒāŃõ»ÆÄĘČÜŅŗĪüŹÕ£»
£Ø3£©Ö¤Ć÷ÅØĮņĖįÖŠĮņŌŖĖŲµÄŃõ»ÆŠŌĒæÓŚĒāŌŖĖŲ£¬ĄūÓĆŌŖĖŲ»ÆŗĻ¼Ū±ä»ÆÉś³ÉµÄ²śĪļ·ÖĪöÅŠ¶Ļ£¬ČōÉś³ÉĒāĘų£¬D×°ÖĆ»įŗŚÉ«±ä»ÆĪŖŗģÉ«£¬ČōÉś³É¶žŃõ»ÆĮņE×°ÖĆÖŠĘ·ŗģ»įĶŹÉ«·ÖĪö£»
£Ø4£©Ńé֤ɜ³ÉµÄČÜŅŗÖŠŗ¬ÓŠĶĄė×Ó£¬ŠčŅŖ°ŃŹŌ¹ÜÖŠµÄŅŗĢåµ¹ČėĖ®ÖŠČܽā£¬¹Ū²ģŹĒ·ń³öĻÖĄ¶É«ČÜŅŗ£¬½«A×°ÖĆÖŠĄäČ“µÄ»ģŗĻČÜŅŗŃŲÉÕ±ÄŚ±Ś»ŗ»ŗµ¹ČėŹ¢Ė®µÄÉÕ±ÖŠ£¬²¢²»¶Ļ½Į°č£¬³öĻÖĄ¶É«ČÜŅŗÖ¤Ć÷ŗ¬ÓŠĶĄė×Ó£¬
£Ø5£©ĄūÓĆ×°ÖĆÖŠĶ²»ÓėÅØĮņĖį½Ó“„·“Ó¦£¬²»·ÅČėĶĖæ·“Ó¦£¬¹Ū²ģB×°ÖĆŹĒ·ń±äĄ¶£¬Čō²»±äĄ¶Ö¤Ć÷ÅØĮņĖįÖŠµÄĖ®²»Ó°ĻģŹµŃ飬ČōĪŽĖ®ĮņĖįĶ³öĻֱ䥶£¬ĖµĆ÷ÅØĮņĖįÖŠµÄĖ®»įÓ°ĻģŹµŃ飬
£Ø6£©ŅĄ¾ŻĶ¼±ķŹż¾Ż·ÖĪöæÉÖŖ£¬ÅØ¶Č²»Ķ¬Ź±³öĻÖŗŚÉ«ĪļÖŹµÄŹ±¼äĖęÅضČŌö“ó¼õŠ”£¬ÅضČĪŖ18mol/LŹ±Éś³ÉµÄŗŚÉ«ĪļÖŹ²»ÄÜĻūŹ§£»
a£®·ÖĪö¹ż³ĢæÉÖŖ£®·¢ÉśµÄ·“Ó¦¹ż³ĢÖŠÉś³É¶žŃõ»ÆĮņŗĶĮņĖįĶµÄ·“Ó¦¹ż³ĢÖŠÓŠĘęĢø·“Ó¦·¢ÉśÉś³ÉŗŚÉ«ĪļÖŹ£¬ĶÓėÅØĮņĖį·“Ó¦Ź±ĖłÉę¼°µÄ·“Ó¦æÉÄܲ»Ö¹Ņ»øö£¬¹ŹA·ūŗĻ£»
b£®Ķ¼±ķŹż¾Ż·ÖĪöæÉÖŖĮņĖįÅضČŃ”ŌńŹŹµ±£¬æɱÜĆā×īŗó²śĪļÖŠ³öĻÖŗŚÉ«ĪļÖŹ£¬¹ŹB·ūŗĻ£»
c£®ÅØĮņĖįŗĶĶµÄ·“Ó¦ÅØ¶ČŠ”ÓŚ15mol/LŅ²æÉÄܳöĻÖŗŚÉ«ĪļÖŹ£¬³öĻֵď±¼ä³¤£¬øĆ·“Ó¦·¢ÉśµÄĢõ¼ž²»Ņ»¶ØŹĒĮņĖįÅØ¶Č”Ż15mol?L-1£¬¹ŹC²»·ūŗĻ£»
d£®Ķ¼±ķŹż¾ŻÖŠ£¬18mol/LµÄÅØĮņĖį£¬³öĻÖŗÖÉ«ĪļÖŹµÄŹ±¼ä¶Ģ£¬ŗŚÉ«ĪļÖŹÄŃŅŌĻūŹ§£¬ĖłŅŌĮņĖįÅضČŌ½“ó£¬ŗŚÉ«ĪļÖŹŌ½æģ³öĻÖ”¢Ō½ÄŃĻūŹ§£¬¹Źd·ūŗĻ£»
£Ø7£©2Cu2++4I-=2CuI+I2£¬2S2O32-+I2=S4O62-+2I-£¬·ÖĪö·“Ó¦æÉÖŖ£¬Cu2+”«I2”«22S2O32-£¬½«øĆ·“Ó¦ĖłµĆČÜŅŗÖŠŗĶŗóÅäÖĘ³É250.00mLČÜŅŗ£¬Č”øĆČÜŅŗ25.00mL¼ÓČė×ćĮæKIČÜŅŗÕńµ“£¬ŅŌµķ·ŪČÜŅŗĪŖÖøŹ¾¼Į£¬ÓĆb mol?L-1Na2S2O3ČÜŅŗµĪ¶ØÉś³ÉµÄI2£¬3“ĪŹµŃéĘ½¾łĻūŗÄøĆNa2S2O3ČÜŅŗV mL£®Čō·“Ó¦ĻūŗÄĶµÄÖŹĮæĪŖag£¬ŌņÉś³ÉĮņĖįĶµÄ²śĀŹæÉŅŌ¼ĘĖćĶŌŖĖŲµÄ²śĀŹ¼“ÓĆĶŌŖĖŲĪļÖŹµÄĮæ¼ĘĖć²śĀŹ£¬Éč25mlČÜŅŗÖŠÉś³ÉĶĄė×ÓĪļÖŹµÄĮæĪŖx£»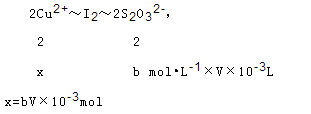
250mlČÜŅŗÖŠĶĄė×ÓĪļÖŹµÄĮæ=bV”Į10-2mol£»ĮņĖįĶµÄ²śĀŹ= 
æ¼µć£ŗæ¼²éĮĖÅØĮņĖįŠŌÖŹµÄ·ÖĪöÅŠ¶Ļ£¬ŹµŃé¹ż³ĢµÄÉč¼Ę·½·Ø£¬²śĪļŹµŃéŃéÖ¤µÄÉč¼Ę·½·ØŗĶĻÖĻóÅŠ¶Ļ



| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
ÓŠX”¢Y”¢ZČżÖÖŌŖĖŲ£¬ŅŃÖŖ£ŗ¢ŁX”¢Y”¢ZµÄµ„ÖŹŌŚ³£ĪĀĻĀ¾łĪŖĘųĢ壻¢ŚXµ„ÖŹæÉŌŚZµ„ÖŹÖŠČ¼ÉÕ£¬Éś³ÉĘųĢ¬XZ£»¢ŪXZ¼«Ņ×ČÜÓŚĖ®£¬µēĄė³öX+ŗĶZ££¬ĘäĖ®ČÜŅŗæÉŹ¹Ą¶É«ŹÆČļŹŌÖ½±äŗģ£»¢ÜĆæ2øöX2·Ö×ÓæÉÓė1øöY2·Ö×Ó»ÆŗĻÉś³É2øöX2Y·Ö×Ó£¬X2YŌŚ³£ĪĀĻĀĪŖĪŽÉ«ŅŗĢ壻¢ŻZµ„ÖŹČÜÓŚX2YÖŠ£¬ĖłµĆČÜŅŗ¾ßÓŠĘÆ°××÷ÓĆ”£
£Ø1£©ĶʶĻX£¬YĮ½ÖÖŌŖĖŲ£ŗX________£¬Y_________£ØĢīŌŖĖŲĆū³Ę£©”£
£Ø2£©Š“³ö¢Ż¹ż³ĢÖŠµÄ»Æѧ·“Ó¦µÄ·½³ĢŹ½ ”£
£Ø3£©Š“³ö¹¤ŅµÉĻÖĘČ”Zµ„ÖŹµÄ»Æѧ·½³ĢŹ½ ”£
£Ø4£©Š“Zµ„ÖŹÓėŹÆ»ŅČé·“Ó¦µÄ»Æѧ·½³ĢŹ½ ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĪŹ“šĢā
ĻõĖįŹĒ¼«ĘäÖŲŅŖµÄ»Æ¹¤ŌĮĻ£¬¹¤ŅµÉĻ½«²śÉśµÄNO2ŌŚĆܱÕČŻĘ÷ÖŠÓĆĖ®¶ą“Ī·“ø“Ń»·ĪüŹÕÖʱøĻõĖį”£
£Ø1£©¹¤ŅµÉĻÓĆĖ®ĪüŹÕNO2Éś²śHNO3£¬Éś³ÉµÄĘųĢ徶ą“ĪŃõ»Æ”¢ĪüŹÕµÄŃ»·²Ł×÷³ä·Ö×Ŗ»ÆĪŖĻõĖį£Ø¼Ł¶ØÉĻŹö¹ż³ĢĪŽĘäĖūĖšŹ§£©”£ŹŌŠ“³öÉĻŹö·“Ó¦µÄ»Æѧ·½³ĢŹ½£ŗ___________________________________________”£
£Ø2£©ĪŖĮĖÖ¤Ć÷NOŅ²æÉŅŌÓėŃõĘųŗĶĖ®¹²Ķ¬·“Ӧɜ³ÉHNO3£¬Ä³Ń§ÉśÉč¼ĘĮĖČēĶ¼ĖłŹ¾×°ÖĆ£ØÓŠ¹Ų¼Š³Ö×°ÖĆŅŃĀŌČ„£©”£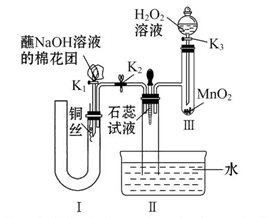
¢Ł¼ģ²é×°ÖĆĘųĆÜŠŌĮ¼ŗĆŗó£¬ĪŖ¹Ū²ģµ½NOĘųĢåÉś³É£¬“ņæŖK1£¬¹Ų±ÕK2£¬Ó¦“ÓUŠĪ¹ÜµÄ³¤¹ÜæŚ×¢ČėĻ”ĻõĖįÖĮ____________ŗó£¬ŃøĖŁ¹Ų±ÕK1£¬¹Ū²ģµ½UŠĪ¹ÜÄŚµÄĻÖĻóŹĒ_______________________________________________________________”£
¢Ś×°ÖĆ¢óÖŠ·¢Éś·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ__________________________________”£
¢ŪÕŗNaOHČÜŅŗµÄĆŽ»ØĶŵÄ×÷ÓĆŹĒ_______________________________________”£
¢Ü“ņæŖK2£¬ÖĮ×°ÖĆ¢ņÖŠ³¤²£Į§¹ÜÖŠµÄĘųĢå³ŹĪŽÉ«ŗ󣬓ņæŖK3£¬·“Ó¦Ņ»¶ĪŹ±¼äŗ󣬳¤²£Į§¹ÜÖŠ²¢Ī“³äĀśŅŗĢ唣Éč¼Ę¼ņµ„·½·Ø¼ģŃ鳤²£Į§¹ÜÖŠµÄĘųĢåŹĒ·ńŗ¬NO________________________________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŹµŃéĢā
£Ø16£©Ä³æĪĶā»ī¶ÆŠ”×éÉč¼ĘĮĖŅŌĻĀŹµŃé·½°øŃéÖ¤CuÓėÅØĻõĖį·“Ó¦µÄ¹ż³ĢÖŠæÉÄܲśÉśNO”£Ę䏵ŃéĮ÷³ĢĶ¼ČēĻĀ£ŗ
£Ø1£©²ā¶ØĻõĖįµÄĪļÖŹµÄĮæ·“Ó¦½įŹųŗ󣬓ÓĻĀĶ¼B×°ÖĆÖŠĖłµĆ100mLČÜŅŗÖŠČ”³ö25.00mLČÜŅŗ£¬ÓĆ0.1mol”¤L-1µÄNaOHČÜŅŗµĪ¶Ø£¬ÓĆ·ÓĢŖ×÷ÖøŹ¾¼Į£¬µĪ¶ØĒ°ŗóµÄµĪ¶Ø¹ÜÖŠŅŗĆęµÄĪ»ÖĆČēÓŅÉĻĶ¼ĖłŹ¾”£ŌŚBČŻĘ÷ÖŠÉś³ÉĻõĖįµÄĪļÖŹµÄĮæĪŖ mol”£
£Ø2£©²ā¶ØNOµÄĢå»ż
¢Ł“ÓÉĻĶ¼ĖłŹ¾µÄ×°ÖĆÖŠ£¬ÄćČĻĪŖӦєÓĆ ×°ÖĆ½ųŠŠCuÓėÅØĻõĖį·“Ó¦ŹµŃé£¬Ń”ÓƵĥķÓÉŹĒ ”£
¢ŚŃ”ÓĆÉĻĶ¼ĖłŹ¾ŅĒĘ÷×éŗĻŅ»Ģ×æÉÓĆĄ“Ķź³ÉŹµŃé²¢²ā¶ØÉś³ÉNOĢå»żµÄ×°ÖĆ£¬ĘäŗĻĄķµÄĮ¬½ÓĖ³ŠņŹĒ£ØĢīø÷µ¼¹ÜæŚ±ąŗÅ£© ”£
¢ŪŌŚ²ā¶ØNOµÄĢå»żŹ±£¬ČōĮæĶ²ÖŠĖ®µÄŅŗĆę±Č¼ÆĘųĘæµÄŅŗĆęŅŖµĶ£¬“ĖŹ±Ó¦½«ĮæĶ²µÄĪ»ÖĆ £Ø”°ĻĀ½µ”±»ņ”°Éżøß”±£©£¬ŅŌ±£Ö¤ĮæĶ²ÖŠµÄŅŗĆęÓė¼ÆĘųĘæÖŠµÄŅŗĆę³ÖĘ½”£
£Ø3£©ĘųĢå³É·Ö·ÖĪö£ŗČōŹµŃé²āµĆNOµÄĢå»żĪŖ112.0mL£ØŅŃÕŪĖćµ½±ź×¼×“æö£©£¬ŌņCuÓėÅØĻõĖį·“Ó¦µÄ¹ż³ĢÖŠ £ØĢī”°ÓŠ”±»ņ”°Ć»ÓŠ”±£©NO²śÉś£¬×÷“ĖÅŠ¶ĻµÄŅĄ¾ŻŹĒ ”£
£Ø4£©ŹµŃéĒ°£¬ÓĆĶŠÅĢĢģĘ½³ĘČ”µÄĶʬÖĮÉŁÓ¦ĪŖ g”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŹµŃéĢā
£Ø14·Ö£©Ä³»ÆѧŠĖȤŠ”×éĶ¬Ń§Õ¹æŖ¶ŌĘÆ°×¼ĮŃĒĀČĖįÄĘ£ØNaClO2£©µÄŃŠ¾æ”£
ŹµŃé¢ń£ŗÖĘČ”NaClO2¾§Ģå
ŅŃÖŖ£ŗNaClO2±„ŗĶČÜŅŗŌŚĪĀ¶ČµĶÓŚ38”ꏱĪö³öµÄ¾§ĢåŹĒNaClO2?3H2O£¬øßÓŚ38”ꏱĪö³ö¾§ĢåµÄŹĒNaClO2£¬øßÓŚ60”ꏱNaClO2·Ö½ā³ÉNaClO3ŗĶNaCl”£Ba(ClO)2æÉČÜÓŚĖ®”£
ĄūÓĆĻĀĶ¼ĖłŹ¾×°ÖĆ½ųŠŠŹµŃ锣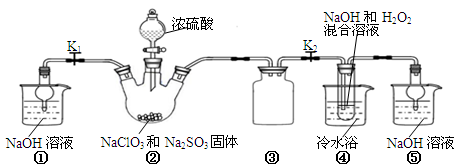
£Ø1£©×°ÖĆ¢ŁµÄ×÷ÓĆŹĒ ×°ÖĆ¢ŪµÄ×÷ÓĆŹĒ ”£
£Ø2£©×°ÖĆ¢ŚÖŠ²śÉśClO2µÄ»Æѧ·½³ĢŹ½ĪŖ ”£
£Ø3£©“Ó×°ÖĆ¢Ü·“Ó¦ŗóµÄČÜŅŗ»ńµĆ¾§ĢåNaClO2µÄ²Ł×÷²½ÖčĪŖ£ŗ
¢Ł¼õŃ¹£¬55”ęÕō·¢½į¾§£»¢Ś³ĆČČ¹żĀĖ£»¢Ū £»¢ÜµĶÓŚ60”ęøÉŌļ£¬µĆµ½³ÉĘ·”£
£Ø4£©Éč¼ĘŹµŃé¼ģŃéĖłµĆNaClO2¾§ĢåŹĒ·ńŗ¬ÓŠŌÓÖŹNa2SO4£¬²Ł×÷ÓėĻÖĻóŹĒ£ŗȔɣĮ澧ĢåČÜÓŚÕōĮóĖ®£¬ ”£
ŹµŃé¢ņ£ŗ²ā¶ØijŃĒĀČĖįÄĘѳʷµÄ“æ¶Č”£
Éč¼ĘČēĻĀŹµŃé·½°ø£¬²¢½ųŠŠŹµŃé£ŗ
¢Ł×¼Č·³ĘČ”ĖłµĆŃĒĀČĖįÄĘѳʷm gÓŚÉÕ±ÖŠ£¬¼ÓČėŹŹĮæÕōĮóĖ®ŗĶ¹żĮæµÄµā»Æ¼Ų¾§Ģ壬ŌŁµĪČėŹŹĮæµÄĻ”ĮņĖį£¬³ä·Ö·“Ó¦£ØŅŃÖŖ£ŗClO2£+ 4I£+4H+ =2H2O+2I2+Cl££©”£½«ĖłµĆ»ģŗĻŅŗÅä³É100mL“ż²āČÜŅŗ”£
¢ŚŅĘČ”25.00mL“ż²āČÜŅŗӌ׶ŠĪĘæÖŠ£¬ÓĆc mol?L-1 Na2S2O3±ź×¼ŅŗµĪ¶Ø£¬ÖĮµĪ¶ØÖÕµć”£ÖŲø“2“Ī£¬²āµĆĻūŗıź×¼ČÜŅŗµÄĢå»żµÄĘ½¾łÖµĪŖV mL£ØŅŃÖŖ£ŗI2 +2S2O32£=2I£+S4O62££©”£
£Ø5£©µĪ¶ØÖŠŹ¹ÓƵÄÖøŹ¾¼ĮŹĒ £¬“ļµ½µĪ¶ØÖÕµćŹ±µÄĻÖĻóĪŖ ”£
£Ø6£© ѳʷ֊NaClO2µÄÖŹĮæ·ÖŹżĪŖ £ØÓĆŗ¬m”¢c”¢VµÄ“śŹżŹ½±ķŹ¾£¬Ź½Įæ£ŗNaClO2 90.5£©”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŹµŃéĢā
£Ø16·Ö£©Ä³ŃŠ¾æŠŌѧĻ°Š”×éĪŖŃŠ¾æĢś·ŪÓėÅØĮņĖį·“Ó¦ĖłÉś³ÉĘųĢåµÄ³É·Ż²¢²ā¶Øø÷ĘųĢåµÄŗ¬Į棬½ųŠŠĮĖČēĻĀŹµŃé£ŗ”¾ŹµŃéĶĘ²ā”æ×ćĮæµÄĢś·ŪÓėÅØĮņĖį·“Ó¦ÄÜÉś³ÉSO2ŗĶH2Į½ÖÖĘųĢ唣
£Ø1£©øĆŠ”×é×÷³ö“ĖĶĘ²āµÄĄķÓÉŹĒ£ŗ ”£
”¾ŹµŃé×¼±ø”æa£®¹©Ń”ŌńµÄŅ©Ę·£ŗĢś·Ū”¢ÅØĮņĖį”¢Ńõ»ÆĶ·ŪÄ©”¢0.2 mol/LµÄH2C2O4±ź×¼ČÜŅŗ”¢0.1 mol/LµÄĖįŠŌKMnO4±ź×¼ČÜŅŗ”¢Ėį¼īÖøŹ¾¼Į”£
b£®ŹµŃé×°ÖĆÉč¼Ę¼°×é×°(¼ÓČČ¼°¼Š³Ö×°ÖĆ¾łŅŃĀŌČ„)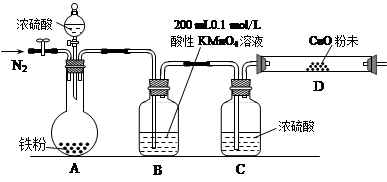
”¾ŹµŃé¹ż³Ģ¼°Źż¾Ż“¦Ąķ”æ
£Ø2£©BÖŠ·¢Éś·“Ó¦µÄĄė×Ó·½³ĢŹ½ĪŖ£ŗ ”£
£Ø3£©ŹµŃéĒ°ĻČĶØČėŅ»¶ĪŹ±¼äµÄN2£¬ĘäÄæµÄŹĒ ”£
£Ø4£©B”¢C”¢DÖŠĖłŹ¢ŹŌ¼Į¾ł×ćĮ棬ŌņÖ¤ŹµÉś³ÉµÄĘųĢåÖŠĶ¬Ź±“ęŌŚSO2ŗĶH2µÄĻÖĻóŹĒ
ӣ
£Ø5£©AÖŠ·“Ó¦½įŹųŗ󣬼ĢŠųĶØN2Ź¹AÖŠÉś³ÉµÄĘųĢåČ«²æøĻ³ö£¬“żB”¢DÖŠ·“Ó¦ĶźČ«ŗó£¬ĻČŗóČż“ĪČ”ÓĆBÖŠ·“Ó¦ŗóµÄČÜŅŗӌ׶ŠĪĘæÖŠ£¬Ćæ“ĪČ”ÓĆ25 mL£¬ÓĆH2C2O4±ź×¼ČÜŅŗ½ųŠŠ²ā¶Ø”£
¢ŁH2C2O4±ź×¼ČÜŅŗÓėĖįŠŌKMnO4ČÜŅŗ·“Ó¦µÄĄė×Ó·½³ĢŹ½ČēĻĀ£¬Ēė½«øĆ·½³ĢŹ½Ķź³É²¢ÅäĘ½”£
( )H2C2O4+( )MnO4- +( )H+ £½( )Mn2+ +( ) H2O+( ) ( )
¢Ś·“Ó¦ĶźČ«µÄĻÖĻóŹĒ ”£
¢ŪÖŲø“²ā¶ØĮ½“Ī£¬Ę½¾łĆæ“ĪŗÄÓĆH2C2O4±ź×¼ČÜŅŗ15.63 mL£¬ŌņĢśÓėÅØĮņĖį·“Ó¦²śÉśµÄSO2ĘųĢå
µÄĪļÖŹµÄĮæĪŖ £»¾³ĘĮ棬ŹµŃéĒ°ŗó×°ÖĆDµÄÖŹĮæ¼õÉŁ0.8 g£¬Ōņ²śÉśµÄĘųĢåÖŠSO2µÄĢå»ż·ÖŹżĪŖ ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŹµŃéĢā
(12·Ö)Ä³ŃŠ¾æŠ”×éÓūĢ½¾æSO2µÄ»ÆѧŠŌÖŹ£¬Éč¼ĘĮĖČēĻĀŹµŃé·½°ø”£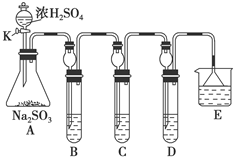
£Ø1£©ŌŚBÖŠ¼ģŃéSO2µÄŃõ»ÆŠŌ£¬ŌņBÖŠĖłŹ¢ŹŌ¼ĮæÉĪŖ________”£
£Ø2£©ŌŚC֊װFeCl3ČÜŅŗ£¬¼ģŃéSO2µÄ»¹ŌŠŌ£¬ŌņCÖŠ·“Ó¦µÄĄė×Ó·½³ĢŹ½ĪŖ__________________________”£
£Ø3£©ŌŚD֊װŠĀÖĘĘÆ°×·ŪÅØČÜŅŗ”£ĶØČėSO2Ņ»¶ĪŹ±¼äŗó£¬DÖŠ³öĻÖĮĖ“óĮæ°×É«³Įµķ”£Ķ¬Ń§ĆĒ¶Ō°×É«³Įµķ³É·Ö½ųŠŠĮĖĢ½¾æ”£Ēė»Ų“šĻĀĮŠĪŹĢā£ŗ
ĻŽŃ”µÄŅĒĘ÷ŗĶŹŌ¼Į£ŗ¹żĀĖ×°ÖĆ”¢ŹŌ¹Ü”¢µĪ¹Ü”¢“ųµ¼¹ÜµÄµ„æ×Čū”¢ÕōĮóĖ®”¢0£®5 mol”¤L£1ŃĪĖį”¢0£®5 mol”¤L£1H2SO4ČÜŅŗ”¢0£®5 mol”¤L£1BaCl2ČÜŅŗ”¢Ę·ŗģČÜŅŗ”¢ŠĀÖĘ³ĪĒåŹÆ»ŅĖ®”£
(¢”)¼ŁÉčŅ»£ŗøĆ°×É«³ĮµķĪŖCaSO3£»
¼ŁÉ趞£ŗøĆ°×É«³ĮµķĪŖ________£»
¼ŁÉčČż£ŗøĆ°×É«³ĮµķĪŖÉĻŹöĮ½ÖÖĪļÖŹµÄ»ģŗĻĪļ”£
(¢¢)»łÓŚ¼ŁÉčŅ»£¬ĢīŠ“ĻĀ±ķ£ŗ
| ŹµŃé²Ł×÷ | Ō¤ĘŚĻÖĻóŗĶ½įĀŪ |
| ½«DÖŠ³Įµķ¹żĀĖ£¬Ļ“µÓøɾ»±øÓĆ | |
| ÓĆĮķŅ»øɾ»ŹŌ¹ÜȔɣĮæ³Įµķѳʷ£¬¼ÓČė ______ | __________ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŹµŃéĢā
ijŠĖȤŠ”×éÉč¼Ę³öĻĀĶ¼ĖłŹ¾×°ÖĆĄ“øĽų½Ģ²ÄÖŠ”°ĶÓėĻõĖį·“Ó¦”±ŹµŃ飬ŅŌĢ½¾æ»ÆѧŹµŃéµÄĀĢÉ«»Æ”£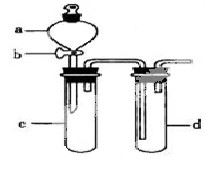
£Ø1£©ŹµŃéĒ°£¬¹Ų±Õ»īČūb£¬ŹŌ¹ÜdÖŠ¼ÓĖ®ÖĮ½žĆ»³¤µ¼¹ÜæŚ£¬Čū½ōŹŌ¹ÜcŗĶdµÄ½ŗČū£¬¼ÓČČc£¬ĘäÄæµÄŹĒ__________”£
£Ø2£©ŌŚdÖŠ¼ÓŹŹĮæNaOHČÜŅŗ£¬cÖŠ·ÅŅ»Š”æéĶʬ£¬ÓÉ·ÖŅŗĀ©¶·aĻņcÖŠ¼ÓČė2mLÅØĻõĖį£¬cÖŠ·“Ó¦µÄ»Æѧ·½³ĢŹ½ŹĒ______________________”£
ŌŁÓÉaĻņcÖŠ¼Ó2mLÕōĮóĖ®£¬cÖŠµÄŹµŃéĻÖĻóŹĒ_____________”£
£Ø3£©ĻĀ±ķŹĒÖĘČ”ĻõĖįĶµÄČżÖÖ·½°ø£¬ÄÜĢåĻÖĀĢÉ«»ÆѧĄķÄīµÄ×ī¼Ń·½°øŹĒ_____£¬ĄķÓÉŹĒ_______”£
| ·½°ø | ·“Ó¦Īļ |
| ¼× | Cu”¢ÅØĻõĖį |
| ŅŅ | Cu”¢Ļ”ĻõĖį |
| ±ū | Cu”¢O2”¢Ļ”ĻõĖį |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŹµŃéĢā
¶žŃõ»ÆĆĢÓėÅØŃĪĖį»ģŗĻ¼ÓČȵƵ½ĀČĘų£¬ČēĶ¼ŹĒÖĘČ”²¢Ģ½¾æCl2»ÆѧŠŌÖŹµÄ×°ÖĆĶ¼”£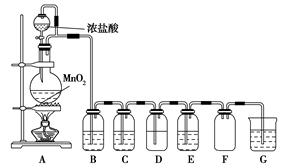
(1)Ō²µ×ÉÕĘæÖŠ·¢Éś·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ ”£
(2)ČōŅŖµĆµ½øÉŌļ“æ¾»µÄĘųĢ壬B”¢CÖŠÓ¦·Ö±šŹ¢·ÅµÄŹŌ¼ĮĪŖ ”¢ ”£
(3)EÖŠČō×°ÓŠFeCl2ČÜŅŗ£¬·“Ó¦µÄĄė×Ó·½³ĢŹ½ĪŖ £¬EÖŠČō×°ÓŠµķ·Ūµā»Æ¼ŲČÜŅŗ£¬ÄܹŪ²ģµ½µÄŹµŃéĻÖĻóŹĒ ”£
(4)ŹµŃéÖŠ·¢ĻÖ£ŗÅØŃĪĖįÓėMnO2»ģŗĻ¼ÓČČÉś³ÉĀČĘų£¬Ļ”ŃĪĖįÓėMnO2»ģŗĻ¼ÓČČ²»Éś³ÉĀČĘų”£Õė¶ŌÉĻŹöĻÖĻóij»ÆѧŠĖȤŠ”×é¶Ō”°Ó°ĻģĀČĘųÉś³ÉµÄŌŅņ”±½ųŠŠĮĖĢÖĀŪ£¬²¢Éč¼ĘĮĖŅŌĻĀŹµŃé·½°ø£ŗ
a£®Ļ”ŃĪĖįµĪČėMnO2ÖŠ£¬Č»ŗóĶØČėHClĘųĢå¼ÓČČ
b£®Ļ”ŃĪĖįµĪČėMnO2ÖŠ£¬Č»ŗó¼ÓČėNaCl¹ĢĢå¼ÓČČ
c£®Ļ”ŃĪĖįµĪČėMnO2ÖŠ£¬Č»ŗó¼ÓČėÅØĮņĖį¼ÓČČ
d£®MnO2ÓėNaClµÄÅØČÜŅŗ»ģŗĻ¼ÓČČ
e£®ÅØĮņĖįÓėNaCl¹ĢĢ唢MnO2¹ĢĢå¹²ČČ
¢ŁŹµŃébµÄÄæµÄŹĒ £¬ŹµŃécµÄÄæµÄŹĒ ”£
¢ŚŹµŃéĻÖĻó£ŗa”¢c”¢eÓŠ»ĘĀĢÉ«ĘųĢåÉś³É£¬b”¢dƻӊ»ĘĀĢÉ«ĘųĢåÉś³É”£ÓÉ“ĖµĆ³öÓ°ĻģĀČĘųÉś³ÉµÄŌŅņŹĒ ”£
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com