
| n |
| V |


 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| A��ת�ƴ���Һ������ƿʱ��δϴ���ձ� |
| B���ζ����յ�ʱ���ζ��ܼ�������Һ�� |
| C���ζ�ʱ����Ӧ����ҡ��̫���ң�������Һ�彦�� |
| D������ʽ�ζ��ܿ�ʼʱ���ӣ����յ�ʱ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
 ij��ѧ��ȤС�����ʵ�飬��Ũ������ͭ��Ӧ�Ƶ�SO2���������ʵ��̽����
ij��ѧ��ȤС�����ʵ�飬��Ũ������ͭ��Ӧ�Ƶ�SO2���������ʵ��̽����| ʵ����� | ʵ����� | ���� | ��Ӧ�Ŀ������ |
| �� | �μ��������۵⻯����Һ���� | �� | |
| �� | �μ������غ�ɫ��KI3��Һ���� | �� | |
| �� | �������������KMnO4��Һ���� | ��Һ����ɫ | |
| �� | ���뼸С��CaCO3���� | �����ݲ��� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| W | �����ǵ���ɫ���� | ||
| X | �ڵؿ��еĺ����ӵڶ�λ | ||
| Y | ԭ�������������ǵ���������
| ||
| Z | ��������ԭ�Ӱ뾶��С�Ľ��� |
| A����̬�⻯������ȶ��ԣ�X��W |
| B������������Ӧˮ��������ԣ�Y��X |
| C�����Ӱ뾶��Z��W |
| D��Y���������к��зǼ��Թ��ۼ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
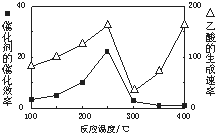 CH4��CO2��Ӧ���������ֵ���ߵĻ�ѧ��Ʒ��
CH4��CO2��Ӧ���������ֵ���ߵĻ�ѧ��Ʒ��| �� �� | CH4 | CO2 | CO | H2 |
| ƽ��Ũ�ȣ�mol?L-1�� | 0.5 | 0.5 | 2.0 | 2.0 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| A��1 | B��2 | C��13 | D��14 |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com