


 ��ĩ1�����ʽ���������ϵ�д�
��ĩ1�����ʽ���������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| | ���� | ���� | ���� |
| A�� | �μ���ˮ��CCl4�������� | �²���Һ����ɫ | ԭ��Һ����I- |
| B�� | �μ�BaCl2��Һ | ���ɰ�ɫ���� | ԭ��Һ��һ����SO42- |
| C�� | �ýྻ��˿պȡ��Һ������ɫ��Ӧ | ����ʻ�ɫ | ԭ��Һ����Na+����K+ |
| D�� | �μ�ϡNaOH��Һ����ʪ���ɫʯ����ֽ�����Թܿ� | ��ֽ������ | ԭ��Һ��һ����NH4+ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��ֽ�ϲ�����ͨ������ֽ��Ϊ����֧�����ֽ��ά�ϵ��ǻ�������ˮ�ԣ�����������ˮ���̶��� |
| B���ؽᾧʱ�����ʵ��ܽ��Խ����Һ��ȴ�ٶ�Խ�����õ��ľ������Խ�� |
| C���������ҵĽ���Һ�м����������ˮ���Ա�֤I����ȫ����ΪI2 |
| D����3~4�����ͷ����ˮ�У�Ƭ�̺�ȡ������Һ���Թ��У���AgNO3��Һ��ϡ�����NaNO2��Һ�������ְ�ɫ������˵��������Ԫ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
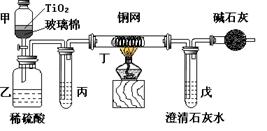
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A���屽���壩����NaOH��Һ����Һ |
| B��MgCl2��Һ(Fe3+)����MgO���壬���� |
| C�����ᣨˮ������������ʯ�ң����� |
| D��Ӳ֬�ᣨ���ᣩ�������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
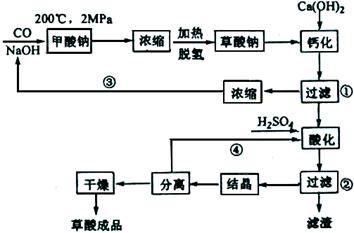
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��ϴ�� | B����ȡ | C������ | D������ |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com