
�赥�ʼ��仯����Ӧ�úܹ㡣��ش��������⣺
(1)�Ʊ���뵼����ϱ����ȵõ��ߴ��衣���ȼ���(SiHCl3)��ԭ���ǵ�ǰ�Ʊ��ߴ������Ҫ��������������ʾ��ͼ���£�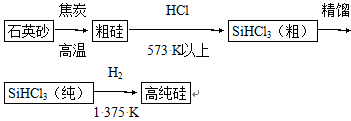
��д���ɴ�SiHCl3�Ʊ��ߴ���Ļ�ѧ��Ӧ����ʽ____________________��
�������Ʊ����̱����ϸ������ˮ��������SiHCl3��ˮ���ҷ�Ӧ����H2SiO3��HCl����һ�����ʣ�д����ƽ�Ļ�ѧ��Ӧ����ʽ____________________��H2��ԭSiHCl3������������O2����������ĺ����____________________��
(2)�����йع���ϵ�˵����ȷ���� (����)��
A�����ʹ軯ѧ�����ȶ��������Ա�ǿ����Һ��ʴ
B�����������跴Ӧ���ʲ�������Ϊ��Һ�ⵥ����
C����ͨ�������ɴ��ʯ��ʯ��ʯӢɰ�Ƴɵģ����۵�ܸ�
D�����ά����Ҫ�ɷ���SiO2
(3)������ˮ��Һ�׳�ˮ������ȡ������������Һ���Թ��У���μ������ᣬ��д��ʵ�����������(�û�ѧ����ʽ˵��)_________________________ ____��
(4)�����������ܵ�����ʱ����Ҫʹ��һ���������ǽ���������ֲ�����ڣ����ֲ�����________(����ĸ)��
A�����½ṹ�մ� ��B�������մɡ� C�������մ�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��16�֣�
��1����ͼΪʵ�����Ʊ����ռ�����HCl��װ�á�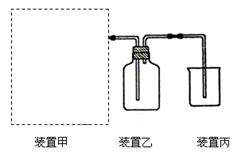
���Ʊ�HCl����ҩƷΪŨ�����Ũ���ᣬ������װ��Ӧѡ����ͼ�е� ��
�� ���������Ũ�����Ũ�����Ʊ�HCl�����ԭ�� ��
�� װ�ñ��������չ�����HCl���壬Ϊ��ֹ���������ձ���Ӧ��װ��ˮ�� ��
��2������Ϊ����ȡ����Ӧ��̽��ʵ�顣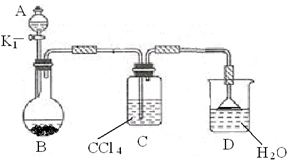
�� ����ͼ��ʾ��װ��ͼ���Ӻø�������
�� ����װ�õ������ԡ�
�� ��A�м��������ı���Һ��Ļ��Һ�壬��B�м����������ۣ�������Ƥ������K1����ʼ���з�Ӧ�������������Һ�ر�K1��
д��B�з�����Ӧ�Ļ�ѧ����ʽ ��
װ��C�������� ��
�� ��Ӧ���������Թ�ȡ����D�е���Һ������
�������Լ���������֤��B�е�ȡ����Ӧ�Ѿ�������
�� ��B�еĹ�Һ���������ˣ������Һ���������ͼ�������ƺ��б�������屽��
�����Լ�Ϊ ����������Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij��ȤС���ͬѧ��ͬ�������ͼ��ʾ�Ķ��ʵ��װ�á���װ�üȿ�������ȡ���壬�ֿ�������֤���ʵ����ʡ�
(1)��������ҩƷ��п����ͭƬ��ŨH2SO4��ŨHNO3������ˮ��NaOH��Һ����K1���ر�K2������ȡ ���塣
(2)ijͬѧ��ʯ��ʯ�����ᡢ��������Һ��ҩƷ�������һʵ�顣����ʵ��Ŀ���� ����K2���ر�K1����A��Һ�����B�к۲쵽��ʵ������Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�������ƣ�NaClO2����һ����Ҫ�ĺ�������������Ҫ����ˮ�������Լ�ɰ�ǡ���֬��Ư����ɱ�����������������ֽ⡣�������ƣ�NaClO3����Ϊԭ���Ʊ��������ƵĹ����������£�
��1����ߡ���Ӧ1����Ӧ���ʵĴ�ʩ�� �����ش�һ�㼴�ɣ�
��2������Ӧ1�� �����ӷ���ʽ�� ��
��3������Ӧ2������������ ���÷�Ӧ�Ļ�ѧ����ʽΪ ��
��4����ȡ����ѹ�����������á���ѹ��������ԭ���� ��
��5��ijѧϰС���õ������ⶨ�ֲ�Ʒ���������Ƶĺ���,ʵ�����£�
a��ȷ��ȡ��������������Ʒmg��С�ձ��У�������������ˮ�����ĵ⻯�ؾ��壬�ٵ���������ϡ���ᣬ��ַ�Ӧ�������û��Һ���250mL������Һ��(��֪��ClO2-+4I-+4H+=2H2O+2I2+Cl-)
b����ȡ25.00mL������Һ����ƿ�У��Ӽ��ε�����Һ����c mol��L-1 Na2S2O3��Һ�ζ����յ㣬�ظ�2�Σ����ƽ��ֵΪV mL ��
(��֪��I2+2S2O32-=2I-+S4O62-)
�ٴﵽ�ζ��յ�ʱ������Ϊ ��
�ڸ���Ʒ��NaClO2����������Ϊ (�ú�m��c��V�Ĵ���ʽ��ʾ������������)
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
Ũ�����������A��F�����ʣ�A���ԣ�B�߷е��ѻӷ���C��ˮ�ԣ�D��ˮ�ԣ�Eǿ�����ԣ�F����ˮ�ų�������
(1)Ũ������ͭ���ȷ�����Ӧ�Ļ�ѧ����ʽΪ ��ʵ���������д�����ɫ�����������ɼ�Ũ�����ڸ�ʵ���б��ֵ������� ��(Ũ���������á�A������B������C������D������E������F����գ���ͬ)
(2)ʵ��֤��ͭ�����ڵ�������O2��Ӧ��Ҳ������ϡ���Ṳ�ȷ�����Ӧ������ҵ��ȴ�ǽ���ͭм�����ȵ�ϡ�����в�ͨ��������Ʊ�CuSO4��Һ��ͭм�ڴ�״̬�±��ܽ�Ļ�ѧ����ʽΪ �������ڸ÷�Ӧ�б��ֵ������� ��
(3)�ڹ���������ϡ����Ļ����Һ�м���ͭƬ�������¾�������ɫ��Һ��д���йط�Ӧ�Ļ�ѧ����ʽ�� ����(2)�з�Ӧ�Ƚϣ���Ӧ������ͬ��ԭ���� ��
(4)�����Ǿ����е�2��3��ˮ���ٵ���������Ũ���ᡣ���ּ�ˮ��������ڣ���ɫ���������������һ�����ɵĽ�̿�������д̼�����ζ�����������д�������д̼���ζ����Ļ�ѧ����ʽ�� ����ʵ����Ũ������ֵ������� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ش�����ʵ��������ȡ������й����⡣
(1)��ͼ����KMnO4��Ũ���ᷴӦ��ȡ���������ļ���װ�á�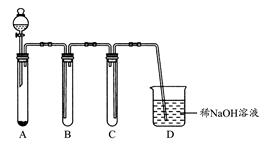
װ��B��C��D�����÷ֱ��ǣ�B ��C ��D ��
(2)��ʵ��������ȡ����NO���塣
����ͼ�����ʺ���ɸ�ʵ��ļ���װ���� (�����)��
�ڸ�����ѡ��װ������±���(����Ҫ�Ŀɲ���)
| | Ӧ��������� | ��������� |
| A | | |
| B | | |
| C | | |
| D | | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ʽ̼��ͭ������������;�㷺�Ļ���ԭ�ϡ�
��1����ҵ�Ͽ������Կ�ʴ��Һ����Ҫ�ɷ���Cu2+��Fe2+��Fe3+��H +��Cl?���Ʊ���ʽ̼��ͭ�����Ʊ��������£�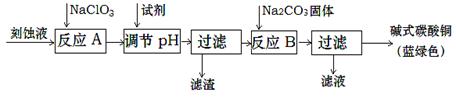
��֪��Cu2+��Fe2+��Fe3+���ɳ�����pH���£�
| ���� | Cu(OH)2 | Fe (OH)2 | Fe (OH)3 |
| ��ʼ����pH | 4.2 | 5.8 | 1.2 |
| ��ȫ����pH | 6.7 | 8.3 | 3.2 |

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���ͼ��ѡ�ñ�Ҫ��װ�ý��е�ⱥ��ʳ��ˮ��ʵ�飬Ҫ��ⶨ���������������������25mL���������������������ԡ�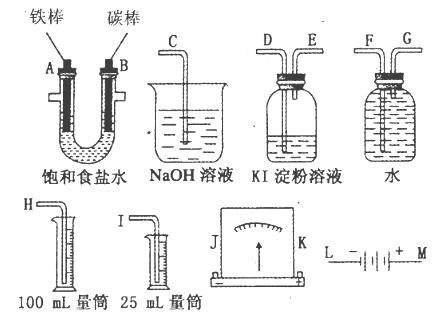
��1��A�������ĵ缫��Ӧʽ��_____________________��B�������ĵ缫��Ӧʽ��_______________________����ⱥ��ʳ��ˮ�Ļ�ѧ����ʽ��______________________��
��2����Դ��M��Ӧ���ӵĵ缫Ϊ____________��A��B������
��3�������������ʵ��װ��ʱ�����ӿڵ���ȷ����˳��Ϊ��______________________________
__________________________________����ӿڱ�ţ���
��4��ʵ���У���ʢ��KI������Һ�������з�����Ӧ�����ӷ���ʽΪ________________��
��5����֪����ʳ��ˮ50mL��ijʱ�̲��H2���Ϊ56mL����״��������ʱ��ҺpHԼΪ___________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������һ����Ҫ�Ĺ�ҵ��Ʒ��Ҳ��һ����Ҫ�Ļ���ԭ�ϡ�Ϊ�о��������Ʊ������ʣ�ijѧϰС������������̽�����
(1)ʵ����������������ʯ�����Ȼ���Ʊ�������д���÷�Ӧ�Ļ�ѧ����ʽ(ע����Ӧ����)___________________________________________________________��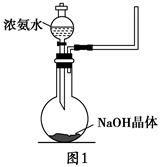
(2)ʵ�����л����Բ�����ͼ1װ��(��ȥ�˼г�װ��)��ҩƷ�����Ʊ�������
�ټ�ͬѧ�����÷������Ʊ�������ԭ��������ѧϰС���ڳ�Աչ�������ۣ��ó�������Ľ��ۣ����в���ȷ����________(����ĸ���)��
a����ˮ�е�NH3��H2O�ֽ�
b����ˮ�д��ڿ��淴Ӧ��NH3��H2O NH4+��OH����NaOH����ʹ�ÿ��淴Ӧ��ƽ�������ƶ�
NH4+��OH����NaOH����ʹ�ÿ��淴Ӧ��ƽ�������ƶ�
c����Ӧԭ������Ρ���ʯ���Ʊ������ķ�Ӧԭ����ͬ
d��NaOH����ˮ�ų�������ʹNH3��H2O�ֽ�
����ͬѧȡ��������Ϊ34%��0.89 g��mL��1��Ũ��ˮ10 mL���ù�����NaOH��֮��ϣ�Ȼ����500 mL����ƿ�ռ�������������������ռ�����״���µİ���________����ƿ��
(3)��ͬѧ���а�������Ȫʵ��̽����
���ռ�����ʱ��Ҫ�ø���������ͼ2��װ��B��ʢװ�ĸ������________��
�ڸ�ͬѧ����ͼ3װ�ã��ɹ����������Ȫʵ�顣�����Ҫ��д����ͬѧ��������ȷ������__________________________________________________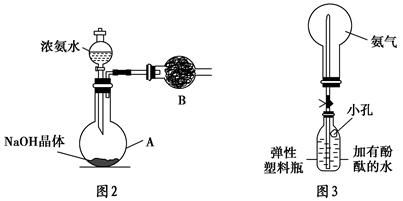
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com